Details of DPV and References
DPV NO: 311 December 1986
Family: Tymoviridae
Genus: Unassigned Tymoviridae
Species: Poinsettia mosaic virus | Acronym: PnMV
Poinsettia mosaic virus
Renate Koenig Institut für Viruskrankheiten der Pflanzen, Biologische Bundesanstalt für Land- und Forswirtschaft, D-3300 Braunschweig, Germany
D.-E. Lesemann Institut für Viruskrankheiten der Pflanzen, Biologische Bundesanstalt für Land- und Forswirtschaft, D-3300 Braunschweig, Germany
R. W. Fulton Department of Plant Pathology, University of Wisconsin, Madison 53706, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Fulton & Fulton (1980),
Koenig & Lesemann (1980),
Pfannenstiel et al. (1982) and
Lesemann et al. (1983).
A virus with RNA-containing isometric particles c. 26 and 29 nm in diameter which sediment as two components. It is found in cultivated Euphorbia pulcherrima and E. fulgens. Its experimental host range is restricted to species of Euphorbia and Nicotiana. It resembles tymoviruses except that the membrane-bounded invaginations it induces in the choroplasts are bounded by single, not double, membranes. It is transmissible by inoculation with sap. No vector is known. The virus probably occurs world-wide.
Main Diseases
Euphorbia pulcherrima (poinsettia) and E. fulgens (Meyer et al., 1986; Paludan & Begtrup, 1986) are the only natural hosts known. Infected E. pulcherrima shows an angular mosaic (Fig. 1) or may be symptomless, especially at higher temperatures.
Geographical Distribution
Probably world-wide in cultivated poinsettias.
Host Range and Symptomatology
The experimental host range is restricted to species of Euphorbia and Nicotiana. The virus is readily transmissible by inoculation of sap from Nicotiana benthamiana to N. benthamiana and, with varying degrees of success, among Euphorbia spp.
-
Diagnostic species
- Nicotiana benthamiana.
Usually symptomless infection of inoculated leaves; irregular chlorosis and crinkling of systemically infected leaves. - Euphorbia cyathophora. Symptomless infection of inoculated leaves; systemic mosaic.
-
Propagation species
- Nicotiana benthamiana
and Euphorbia cyathophora.Assay species
- No local lesion host is known.
Strains
None reported.
Transmission by Vectors
Vector unknown.
Transmission through Seed
Not detected in Euphorbia cyathophora (Fulton & Fulton, 1980) or E. pulcherrima (R. W. Fulton, unpublished data).
Serology
The virus is a good immunogen. Antisera with titres of 1/256-1/2048 in gel diffusion tests have been prepared.
Relationships
The virus has many properties typical of tymoviruses including particle morphology, sedimentation behaviour, coat protein M. Wt, nucleic acid M. Wt and the occurrence of large amounts of ‘empty protein shells’ in nuclei. It differs from definitive tymoviruses in that the top component particles are smaller in diameter than those of the bottom component and are unstable in 2% neutral phosphotungstate or ammonium molybdate solutions. Also the vesicles induced in chloroplasts are bounded by a single membrane not the double one typical of tymoviruses (Lesemann et al., 1983). No serological relationships have been found to any of the definitive tymoviruses.
Stability in Sap
Sap from infected Euphorbia cyathophora was still infective after dilution to 10-4 and after heating for 10 min at 60° but not 65°C. Infectivity of sap stored at c. 24°C was lost after 8-10 days, but was retained for more than 1 year in finely diced leaves desiccated over CaCl2 at 2°C.
Purification
(Lesemann et al., 1983). Homogenize infected Nicotiana benthamiana or Euphorbia cyathophora leaves in 0.07 M phosphate buffer containing 0.1% mercaptoacetate at pH 7.2 (2 ml/g tissue), remove coarse plant debris by squeezing through cotton cloth, mix fluid gently with n-butanol to 8% (v/v). After 30 min, subject the mixture to one cycle of differential centrifugation (20 min at 12,000 g; 90 min at 65,000 g) and resuspend final pellets in 0.03 M phosphate buffer, pH 7.6. Separate and further purify the components by centrifugation for 4 h at 85,000 g on 10-40% (w/v) linear sucrose density gradient columns. Other methods have been described by Fulton & Fulton (1980) and Koenig & Lesemann (1980). Yields are c. 300 mg/kg E. cyathophora tissue.
Properties of Particles
The particles sediment as two components in sucrose gradients, a fast sedimenting (B) component consisting of infective nucleoprotein particles, and a slower sedimenting (T) component consisting of non-infective particles which are penetrated by negative staining agents. The two components are serologically indistinguishable.
Sedimentation coefficient (s20, w) at infinite dilution: 53 S (T), 115 S (B).
Isoelectric point: below pH 7.0.
Electrophoretic mobility: In 1% agarose containing 0.025 M phosphate buffer, pH 7, the virus migrates towards the anode.
A260 (0.1%, 1 cm): 8.4 for unfractionated virus.
A260/A280: 1.0 (T), 1.8 (B).
Buoyant density in CsCl (g/cm3): 1.29 (T), 1.41 (B).
Particle Structure
Particles of the bottom component are 29 nm in diameter, and those of the top component 26 nm in diameter, when negatively stained with uranyl acetate (Lesemann et al., 1983). Particles stained in phosphotungstate, or especially in ammonium molybdate, are slightly swollen or disrupted. The arrangement of morphological subunits seen with bottom component particles (Fig. 2, Fig. 3) closely resembles that of tymoviruses (Fulton & Fulton, 1980; Lesemann et al., 1983).
Particle Composition
Nucleic acid: Single-stranded RNA with a M. Wt of c. 2 x 106, estimated by gel electrophoresis under non-denaturing conditions, comprising c. 35% of the weight of B particles. Molar percentage of nucleotides (G:A:C:U) is 21:24:31:24 (Lesemann et al., 1983). T particles may possess a small amount of RNA (Pfannenstiel et al., 1982).
Protein: Single protein species with M. Wt c. 21 x 103.
Relations with Cells and Tissues
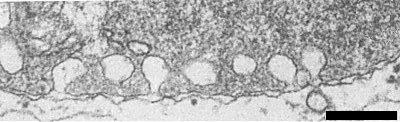
Particles of the virus occur in various tissues of infected leaves of poinsettia and N. benthamiana. The particle concentration is especially high in cytoplasm, vacuoles and nuclei of leaf parenchyma cells (Fig. 6). Protein shells that are apparently largely empty (Fig. 6) are found in the nuclei and in the vesicles of chloroplasts and, together with nucleoprotein particles, in the cytoplasm and vacuoles. In the central parts of the nuclei they form large accumulations (Fig. 6) which sometimes appear to have crystallized. Chloroplasts of affected cells often show an internal vesiculation (Fig. 4). These vesicles are bounded by a single membrane and apparently originate from invaginations of the inner membrane of the chloroplast envelope (Fig. 5). In contrast to this, all definitive tymoviruses studied induce the formation of vesicles bounded by a double membrane, formed by invagination of both membranes of the chloroplast envelope (Lesemann, 1977; Matthews, 1982).
Notes
Poinsettias usually contain poinsettia cryptic virus in addition to poinsettia mosaic virus. The former virus occurs in much lower concentration, can be distinguished from poinsettia mosaic virus by serology and causes no mosaic symptoms. Poinsettias free of poinsettia mosaic and poinsettia cryptic viruses may develop chlorotic spots of unknown etiology. These are readily distinguished from the angular mosaic seen in plants infected with poinsettia mosaic virus.
Acknowledgements
The senior author’s work was supported by the Deutsche Forschungsgemeinschaft.
Figures
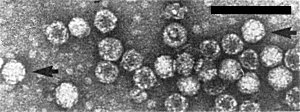
Particles from a partially purified preparation negatively stained with uranyl acetate. Particles showing morphological subunits are indicated by arrows. Bar represents 100 nm.

Particles from a purified preparation negatively stained with potassium phosphotungstate. Bar represents 100 nm.

Ultrathin section of a leaf parenchyma cell of poinsettia showing a chloroplast with internal vesicles. Bar represents 500 nm.
References list for DPV: Poinsettia mosaic virus (311)
- Fulton & Fulton, Phytopathology 70: 321, 1980.
- Koenig & Lesemann, Pl. Dis. 64: 782, 1980.
- Lesemann, Phytopath. Z. 90: 315, 1977.
- Lesemann, Koenig, Huth, Brunt, Phillips & Barton, Phytopath. Z. 107: 250, 1983.
- Matthews, Intervirology 17: 1, 1982.
- Meyer, Lesemann & Koenig, NachrBl. dt. PflSchutzdienst., Stuttg. 38: 163, 1986.
- Paludan & Begtrup, Tidsskr. PlAvl 90: 283, 1986.
- Pfannenstiel, Mintz & Fulton, Phytopathology 2: 252, 1982.