Details of DPV and References
DPV NO: 313 December 1986
Family: Unallocated ssRNA- viruses
Genus: Varicosavirus
Species: Tobacco stunt virus | Acronym: TStV
Tobacco stunt virus
S. Kuwata Central Research Institute, Japan Tobacco Inc., Yokohama 227, Japan
S. Kubo Central Research Institute, Japan Tobacco Inc., Yokohama 227, Japan
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease described by Hidaka (1950) and Hidaka et al. (1956); virus particles characterized by Kuwata & Kubo (1981).
A virus with a genome of double-stranded RNA and rod-shaped particles 18 nm wide, the commonest lengths being 300-340 nm. Transmitted in soils by the fungus Olpidium brassicae and readily by inoculation of sap to a moderate range of hosts. Found in Japan.
Main Diseases
Causes severe stunting and necrosis in tobacco (Nicotiana tabacum) (Fig. 1). A characteristic necrotic banding occurs in the stem at ground level. The field symptoms vary with tobacco cultivar and with environmental conditions.
Geographical Distribution
Reported only from Japan.
Host Range and Symptomatology
Restricted in nature to tobacco but transmitted, experimentally, to 35 species in 13 families by inoculation with viruliferous Olpidium brassicae (Hiruki, 1967) and to 41 species in 9 dicotyledonous families by sap inoculation (Hiruki, 1975).
-
Diagnostic species
- Nicotiana tabacum
(tobacco). Necrotic lesions 3-6 mm in diameter in inoculated leaves 4-7 days after manual inoculation (Fig. 2). Systemic infection occurs in the glasshouse below 20°C. The first systemic symptom is clearing of the veins, followed by vein necrosis, necrotic ring-like patterns, mottling and stunting. The young leaves are crinkled and deformed. Systemic symptoms are masked almost completely at constant temperatures above 25°C (Hidaka et al., 1956). -
Tetragonia expansa. Chlorotic or necrotic local lesions in inoculated
leaves; no systemic infection.
- Chenopodium amaranticolor. Necrotic local lesions in inoculated leaves; no systemic infection.
-
Propagation species
- Nicotiana tabacum
is useful for maintaining cultures and as a source of virus for purification.Assay species
- Nicotiana tabacum
is the most reliable local lesion host (Fig. 2). N. tabacum cv. Bright Yellow is a convenient ‘bait plant’ in fungus transmission experiments (Fig. 3).
Strains
None distinguished, although there are minor differences in symptoms induced by different isolates (S. Kuwata, unpublished results).
Transmission by Vectors
The virus is transmitted by the chytrid fungus, Olpidium brassicae. The virus is carried, apparently internally, in the resting spores, in which it persists for at least 10 years. Viruliferous O. brassicae loses the virus after serial transfer in cowpea (Hiruki, 1965) or lettuce (Hidaka et al., 1975). Virus-free O. brassicae can acquire transmissible virus when cultured on plants that were infected by manual inoculation of sap or by grafting (Hidaka & Tagawa, 1965; Hiruki, 1965).
Transmission through Seed
None found in tobacco (Hidaka et al., 1956).
Serology
Moderately immunogenic in rabbits. An antiserum with a titre of 1/512 in microprecipitin tests has been prepared (Kuwata & Kubo, 1983). The virus precipitates on exposure to 0.15 M NaCl and therefore Ouchterlony double diffusion tests are done in agarose gel containing 0.1 M K2HPO4. In double diffusion tests the antiserum reacted with partially purified virus, but not with crude extracts of infected tobacco leaves, to produce a single line of precipitate. Enzyme-linked immunosorbent assay and immunosorbent electron microscopy are useful for detecting the virus (Kuwata & Kubo, 1983).
Relationships
The particles are morphologically similar to the rod-shaped particles found by Kuwata et al. (1983) in lettuce with big vein disease; in double diffusion tests, the two kinds of particle reacted with homologous and heterologous antisera to produce fused lines of precipitate. No serological relationship was detected between tobacco stunt virus and tobacco mosaic, soil-borne wheat mosaic or beet necrotic yellow vein viruses (Kuwata & Kubo, 1983).
Stability in Sap
Infectivity of sap is stabilized by adding chelating agents such as 1-phenylthiosemicarbazide and sodium diethyldithiocarbamate (Hiruki, 1964), or reducing agents such as sodium sulphite and dithiothreitol (Kuwata & Kubo, 1981). Transmission by inoculation with sap is difficult without these additives. In tobacco sap, the thermal inactivation point (10 min) is 40-50°C and the dilution end-point between 10-3 and 10-4. In the presence of 0.01 M sodium sulphite, the virus in sap at 0°C loses most of its infectivity in 8 h but retains a little for 1 day (S. Kuwata, unpublished results).
Purification
The virus has not been highly purified without loss of infectivity; the particles have a strong tendency to aggregate under conditions in which infectivity is retained. The following method yields partially purified virus with low infectivity. Blend each 1 g infected leaf tissue with 2 ml 0.3 M phosphate buffer, pH 7.4, containing 0.01 M sodium sulphite, add Triton X-100 to 1% (v/v), and stir for 30 min. Centrifuge at 6000 g for 20 min. Concentrate the virus by centrifuging at 70,000 g for 75 min through a 30% sucrose cushion in 0.3 M phosphate buffer, pH 7.4, containing 1% (v/v) Triton X-100 and 0.01 M sodium sulphite. Resuspend the pellet in the same buffer for 30 min. Clarify and concentrate by centrifugation (6000 g for 20 min; 100,000 g for 45 min) through a 30% sucrose cushion. Resuspend the pellet in 0.1 M phosphate buffer, pH 7.0. Purify the virus particles further by isopycnic centrifugation in caesium sulphate: the virus particles form a single light-scattering band, but infectivity is abolished (Kuwata & Kubo, 1982).
Properties of Particles
Buoyant density in Cs2SO4 is c. 1.27 g/cm3 (Kuwata & Kubo, 1982).
Particle Structure
The particles are straight rods, helically constructed with a pitch of 5.0 nm and a hollow core. The particles are 18 nm wide and usually 300-340 nm long (Fig. 4, Fig. 5); they are completely degraded in 1% neutral potassium phosphotungstate or ammonium molybdate unless previously fixed with glutaraldehyde (Kuwata & Kubo, 1981).
Particle Composition
Nucleic acid. Purified virus yields two double-stranded RNA species with M. Wt of 4.5 x 106 and 4.0 x 106, estimated by electrophoresis in 3% polyacrylamide gels (Fig. 6; Kuwata & Kubo, 1984).
Protein: A single species, of M. Wt 48,000, estimated by electrophoresis in 10% polyacrylamide gels containing sodium dodecyl sulphate (Kuwata & Kubo, 1984).
Relations with Cells and Tissues
The virus particles aggregate in the cytoplasm of infected tobacco mesophyll cells. Some aggregates are in paracrystalline arrays, and the particles are around 300 nm long (Fig. 7; Kuwata & Kubo, 1981).
Notes
Tobacco stunt virus and lettuce big vein virus (Kuwata et al., 1983) share several characteristics. Both have similar rod-shaped particles containing double-stranded RNA, and are transmitted in the same manner by the chytrid fungus, Olpidium brassicae. Moreover, there is a close serological relationship between the two viruses. They differ markedly, however, in host range; tobacco stunt virus infects tobacco but not lettuce, whereas lettuce big vein virus infects lettuce but not tobacco. Therefore they should be considered as distinct viruses rather than as strains of one virus.
The properties of these two viruses strongly suggest that they form a new plant virus group.
Figures

Systemic symptoms in N. tabacum cv. Bright Yellow seedlings inoculated by means of viruliferous Olpidium brassicae.
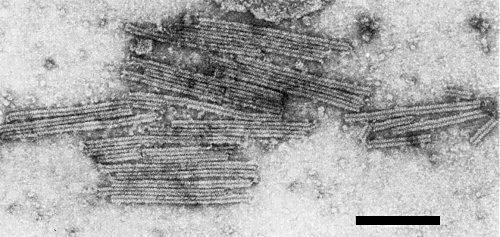
Virus particles in sap from an infected tobacco leaf, stained with 2% uranyl acetate. Bar represents 200 nm.

Virus particles from a partially purified preparation, stained with 2% potassium phosphotungstate after fixation with 5% glutaraldehyde for 15 min. Bar represents 100 nm.
References list for DPV: Tobacco stunt virus (313)
- Hidaka, Ann. phytopath. Soc. Japan 15: 40, 1950.
- Hidaka, Hiruki, Nakano, Shimizu & Uozumi, Bull. Hatano Tobacco Exp. Stn 40: 1, 1956.
- Hidaka & Tagawa, Ann. phytopath. Soc. Japan 31: 369, 1965.
- Hidaka, Tagawa & Hidaka, Proc. 1st intersect. Cong. IAMS, Tokyo, 1974, 3: 287, 1975.
- Hiruki, Virology 23: 288, 1964.
- Hiruki, Virology 25: 541, 1965.
- Hiruki, Virology 33: 131, 1967.
- Hiruki, Can. J. Bot. 53: 2425, 1975.
- Kuwata & Kubo, Ann. phytopath. Soc. Japan 47: 264, 1981.
- Kuwata & Kubo, Ann. phytopath. Soc. Japan 48: 392, 1982.
- Kuwata & Kubo, Ann. phytopath. Soc. Japan 49: 433, 1983.
- Kuwata & Kubo, Abstr. 6th int. Congr. Virol. Sendai, Abstr. No. P45-2, 1984.
- Kuwata, Kubo, Yamashita & Doi, Ann. phytopath. Soc. Japan 49: 246, 1983.



