Details of DPV and References
DPV NO: 320 December 1986
Family: Unallocated ssRNA- viruses
Genus: Tenuivirus
Species: Rice grassy stunt virus | Acronym: RGSV
Rice grassy stunt virus
H. Hibino International Rice Research Institute, P.O. Box 933, Manila, Philippines
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Disease described by Rivera et al. (1966) and
Bergonia et al. (1966).
Virus characterized by Hibino et al. (1985b).
Synonym
- Rice rosette virus (Bergonia et al., 1966)
-
A virus with filamentous particles, many of them circular, with a modal contour length of 950-1350 x 6-8 nm. It is transmitted in a persistent manner by planthoppers, especially Nilaparvata lugens. The host range is confined to Oryza spp. and Nilaparvata spp. It is widely distributed in rice-growing countries in Asia, and economically important in the tropical and subtropical regions.
Main Diseases
Rice plants (Oryza sativa) infected at the seedling stage are severely stunted, with excessive tillering and erect leaves (Fig. 1, Fig. 2). The leaves are short, narrow, and pale green or pale yellow, and often have numerous small irregular dark brown spots of various shapes and sizes (Rivera et al., 1966; Abeygunawardena, 1969; Ling, 1972; Tantera et al., 1973). Newly developed leaves may be mottled or striped. Leaves may remain green when supplied with adequate nitrogenous fertilizer (Rivera et al., 1966; Ling, 1972). Infected plants produce either no panicles or only a few panicles bearing dark brown and unfilled grains. Late infection causes leaf yellowing, brown coloured panicles and abortive kernels (Rivera et al., 1966; Iwasaki et al., 1980b). Depending on the variety, yield reductions in greenhouse experiments were 70-100% when the plants were infected at 1-1.5 months, and 0-80% when they were infected at 2 months (Iwasaki et al., 1980b; Palomar & Ling, 1968). The yield loss in the field is difficult to estimate, because damage caused by the virus is difficult to distinguish from that caused by the brown planthopper vector and by the planthopper-borne rice ragged stunt virus (Palmer et al., 1978; Hibino, 1979). In 1974-1977, a total of 1.2 million ha in Indonesia was affected by the vector and the virus, and yield losses from both causes combined were in excess of 3 million tons of paddy rice (Palmer et al., 1978).
Geographical Distribution
Reported from India, Indonesia, Japan, Malaysia, Philippines, Sri Lanka, Taiwan and Thailand (Rivera et al., 1966; Raychaudhuri et al., 1967; Abeygunawardena, 1969; Ou & Rivera, 1969; Wathanakul & Weerapat, 1969; Hsieh & Chiu, 1970; Anjaneyulu, 1974; Iwasaki & Shinkai, 1979).
Host Range and Symptomatology
Only rice is infected naturally. The virus is not transmitted mechanically. All Oryza spp. so far tested experimentally (by exposure to virus-carrying vectors) were infected (Ling et al., 1970; Ling, 1972; Chen, 1984).
-
Diagnostic species
- Oryza sativa
cv. Taichung Native 1. Symptoms described above (Fig. 1, Fig. 2). -
O. sativa cv. Shan-san-sa-san. Conspicuous yellowish-white striping
(Ling et al., 1970)
(Fig. 4).
- O. sativa cv. Reiho. Severe leaf necrosis and eventual wilting (Iwasaki & Shinkai, 1979).
-
Propagation species
- Rice cultivar Taichung Native 1 is commonly used. The virus can be grown in Oryza spp. inoculated by virus-carrying vectors.
-
Assay species
- No local lesion host is known. Seedlings of rice cultivars that are not resistant to the vector or to the virus can be used.
Strains
Three symptom variants of the virus were isolated in Taiwan (Fig. 2) (Chen & Chiu, 1982). Isolates designated ‘wilted stunt’ cause severe stunting and leaf yellowing in rice, and are often lethal. ‘Grassy stunt B’ isolates cause conspicuous tillering and narrow, short and pale green leaves. ‘Grassy-stunt Y’ isolates cause less stunting, less prominent proliferation of tillers and pale green to normal-coloured leaves of normal width. The incubation period in N. lugens was 3-14 days (average 6.3) for wilted stunt isolates, 4-23 days (average 8.7) for grassy stunt B isolates and 4-17 days (average 7.8) for grassy stunt Y isolates.
In the Philippines a new strain (strain 2), which occurred in rice crops during 1982-1983, caused yellow to orange discoloration of leaves and premature death of plants in addition to the symptoms described for an ‘ordinary’ strain (strain 1) (Fig. 1) (Cabauatan & Hibino, 1983; Hibino & Cabauatan, 1983; Cabauatan et al., 1985; Hibino et al., 1985a). Strains 1 and 2 are serologically related (Fig. 7), and are similar in morphology and vector relations (Cabauatan & Hibino, 1983; Hibino et al., 1983c; Cabauatan et al., 1985; Hibino et al, 1985a). Strain 2 is pathogenic to rice cultivars with a resistance gene derived from O. nivara (Cabauatan & Hibino, 1983; Cabauatan et al., 1985; Hibino et al., 1985a). Isolates similar to strain 2 in symptomatology were reported in Thailand (Disthaporn et al., 1983; Chettanachit et al., 1985) and India (Mariappan et al., 1984). Wilted stunt isolates have some similarities in symptomatology to those of strain 2 (Chen & Chiu, 1982; Cabauatan & Hibino, 1983; Cabauatan et al., 1985). Severe, mild and ordinary strains, which are serologically related, were reported in Japan (Iwasaki et al., 1985a).
Serologically, a Japanese isolate was indistinguishable from strains 1 and 2 (Hibino & Cabauatan, 1983; Hibino et al., 1983c; Cabauatan et al., 1985; Hibino et al., 1985a) and closely related to severe strains from India (Mariappan et al., 1984) and Thailand (Chettanachit et al., 1985), and to grassy stunt B and wilted stunt isolates from Taiwan (C. C. Chen, personal communication).
Transmission by Vectors
The planthoppers Nilaparvata lugens (Fig. 3) (Rivera et al., 1966), N. bakeri and N. muiri (Iwasaki et al., 1980a) transmit the virus. The proportion of active transmitters depends on the colony, ranging from 5 to 60% for N. lugens (Agueiro & Ling, 1978; Chen, 1983; Chen & Chiu, 1982; Ghosh et al., 1979; Iwasaki et al., 1980a, 1984; Ling et al., 1969, 1970; Rivera et al., 1966; Senboku & Shikata, 1980; Tantera et al., 1973). The proportion of transmitters can be increased by selective breeding of transmitting insects but decreases gradually when the selection is discontinued (Ling & Aguiero, 1967; Iwasaki et al., 1982). With N. lugens, the proportion of transmitters does not differ obviously between male and female adults, between dark brown and light brown insects between macropterous and brachypterous insects (Ling et al., 1969), or between biotypes 1, 2 and 3 (Aguiera & Ling, 1978). However, N. lugens nymphs are more efficient than adults in transmitting the virus, which also has a shorter incubation period in nymphs than in adults (Chen, 1983).
In N. lugens, the minimum acquisition access period is 1 h, the latent period averages 8 days (range 3-28 days), and the minimum inoculation period is 9 min (Ling et al., 1969; Iwasaki & Shinkai, 1979; Senboku et al., 1980; Chen & Chiu, 1982; Hibino et al., 1985a). In serial daily transmissions, N. lugens transmits the virus intermittently until death. No infective N. lugens were obtained from the eggs of transmitting females (Ling, 1972). The average life span of transmitting N. lugens (15.4 days) is significantly shorter than that of non-transmitting insects (17.5 days) (Ling, 1972). The virus can be detected in extracts of individual N. lugens by the latex agglutination test and by ELISA (Omura et al., 1984; Hibino et al., 1985b; Iwasaki et al., 1958b); 40-50% of individual N. lugens that were found to contain the virus by ELISA transmitted it to test plants (Omura et al., 1984; Iwasaki et al.,1985b.
Transmission through Seed
None found in rice (Ling, 1972).
Serology
The virus is a good immunogen. Antisera with titres of 1/256-1/2048 in ring precipitin tests were obtained from rabbits (Hibino et al., 1983b, 1983c, 1985a, 1985b; Iwasaki et al., 1985b). In gel diffusion tests in 0.8% agar with or without SDS, a single band of precipitate was formed (Hibino et al., 1983a, 1985a, 1985b; Iwasaki et al., 1985b). The latex agglutination test detected the virus in extracts of infected rice leaves or of vector insects diluted up to 1/5120 and 1/1024-1/2400, respectively (Hibino & Cabauatan, 1983; Hibino et al., 1985b; Omura et al., 1984). In ELISA, the virus was detected in leaf extracts diluted up to 1/100,000 and in insect extracts diluted up to 1/5120 (Hibino et al., 1985b; Iwasaki et al., 1985b; Toriyama & Kojima, 1985). ELISA has been used to assess virus incidence in N. lugens (Iwasaki et al., 1985b; Flores et al., 1986) and in diagnosis of the disease.
Relationships
In SDS-immunodiffusion tests with antiserum to rice stripe virus, rice grassy stunt virus formed a single band of precipitate, and a spur formed between this band and that produced by rice stripe virus (Hibino et al., 1983a, 1985b) (Fig. 7). Antiserum to rice grassy stunt virus did not react with rice stripe virus. In the ring interface test, antiserum to rice stripe virus reacted with rice grassy stunt and maize stripe viruses but not with rice hoja blanca virus; antiserum to rice grassy stunt virus reacted weakly with rice stripe virus and healthy antigen, but not with maize stripe or rice hoja blanca viruses (Hibino et al., 1983a, 1985b). Toriyama & Kojima (1985) confirmed the serological reaction between rice grassy stunt virus and antiserum to rice stripe virus with the ring interface test but not with ELISA. Rice stripe, maize stripe and rice hoja blanca viruses are similar to rice grassy stunt virus in morphology and vector relations (Koganezawa et al., 1975; Gingery et al., 1981; Toriyama, 1982; Gingery et al., 1983; Morales & Niessen, 1983; Hibino et al., 1985b), and can be classified in the same group.
Stability in Sap
Assayed by determining the frequency of virus transmission by vector insects that had been injected with treated sap or insect extracts (Senboku & Shikata, 1980; Shikata et al., 1980; Hibino et al., 1982; Chen & Wang, 1984). Dilution end-point: 10-2-10-3 in extracts from infected rice leaves and 10-3-10-6 in extracts from viruliferous insects (Senboku & Shikata, 1980; Hibino et al., 1982; Chen & Wang, 1984). Thermal inactivation point (10 min): 50°C (Chen & Wang, 1984). Longevity in leaf extracts: less than 12 h at room temperature (Hibino et al., 1982), more than 6 days at 4°C (Chen & Wang, 1984). Infectivity in plant extracts was retained after freezing and thawing, and after clarification with organic solvents (Hibino et al., 1982; Chen & Wang, 1984). In another report, infectivity was reduced by freezing and by treatment with polyethylene glycol (PEG; M. Wt 6000), chloroform, n-butanol or a 1:1 chloroform : n-butanol mixture (Senboku et al., 1980).
Purification
The virus was purified from infected rice plants by clarification with Mg-bentonite and carbon tetrachloride, treatment with PEG and Triton X-100, and sucrose density gradient centrifugation (Hibino et al., 1983b, 1985b). The virus was also purified following a similar but slightly modified method (Hibino et al., 1985a). The yield of virus was 0.5-1.5 mg per 300 g fresh leaf tissue. Iwasaki et al. (1985b) purified the virus by filtration through a Celite layer, treatment with chloroform and Triton X-100, differential centrifugation and sucrose density gradient centrifugation.
The method recommended (H. Hibino & T. Omura, unpublished data) is as follows. Homogenize fresh infected plants with 0.1 M phosphate buffer, pH 7.0 (PB) containing 0.01 M ascorbate and 0.01 M sodium diethyldithiocarbamate. Shake 4 vol homogenate with 1 vol chloroform for 2 min and collect the aqueous layer after centrifugation at 8000 g for 15 min. To the aqueous solution add PEG (M. Wt 6000) to 4% (w/v), NaCl to 0.2 M, and Triton X-100 to 0.1% (v/v). Centrifuge at 8000 g for 15 min and suspend the pellet in 0.1 M PB. Treat the suspension with 10% carbon tetrachloride for 10 min and collect the aqueous layer. Repeat this treatment once more. Centrifuge the clarified solution at 130,000 g for 2 h. Suspend the pellet in 0.01 M PB and centrifuge the suspension at 8000 g for 15 min. Float the supernatant fluid on a 10-45% sucrose density gradient in 0.01 M PB and centrifuge at 70,000 g for 3 h. The preparation forms three bands. The material from each band may be concentrated by centrifugation at 130,000 g for 2 h. There is three times as much virus in the middle and bottom bands together as in the top band.
Properties of Particles
A260/A280 of purified sucrose gradient fractions: 1.16-1.29 (Hibino et al., 1985a, 1985b). Purified sucrose gradient fractions were not infective when assayed by injection into N. lugens (Hibino et al., 1983a, 1985b). Infectivity in clarified extracts of infected leaves was neutralized by incubating them with immunoglobulin to the virus before injection into the vector (Iwasaki et al., 1985b).
Particle Structure
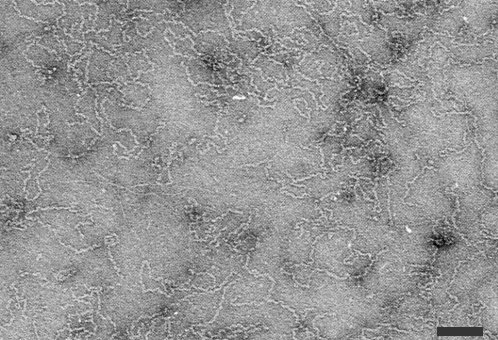
Purified virus particles are long threads up to 2 µm long and 6-8 nm wide in uranyl acetate (Fig. 8) (Cabauatan et al., 1985; Hibino et al., 1985a, 1985b; Iwasaki et al., 1985b). Many of the particles are circular (Fig. 9) with a contour length of 200 to 2400 nm but mainly of 950-1350 nm (Fig. 6) (Hibino et al., 1985a, 1985b). The particles are about 4 nm wide in phosphotungstate.
Particle Composition
Nucleic acid: RNA, four species, of M. Wt (x 10-6) 1.45, 1.3, 1.2 and 1.15 (Toriyama, 1985). Of the three bands obtained (Toriyama, 1985) when purified virus was centrifuged in 20-50% sucrose density gradients, the middle component had little or none of the 1.45 x 106 M. Wt RNA and the top component contained only a small amount of RNA.
Protein: One species, of M. Wt 3.1 x 104, was reported by Hibino et al. (1985b), but Toriyama (1985) reported two proteins of M. Wt 3.4 x 104 and 3.15 x 104.
Relations with Cells and Tissues
Bundles of fibrils were observed in chlorenchyma cells, either in the nucleus and cytoplasm (Fig. 5) or in membrane-bound bodies in the cytoplasm (Pellegrini & Bassi, 1978; Chen et al., 1979). However, in addition, tubular structures associated with 25 nm-diameter particles were found in sieve elements and phloem cells of infected rice leaves (Pellegrino & Bassi, 1978; Shikata et al., 1980). Similar 25 nm particles were found in crystalline arrays in fat bodies and tracheae of viruliferous planthoppers (Shikata et al., 1980).
Ecology and Control
The disease was successfully controlled in Indonesia, Philippines and Vietnam by planting cultivars having the gene for virus resistance derived from O. nivara (Ling et al., 1970; Khush & Ling, 1974; Nuque et al., 1982). In the early 1980s, however, a new strain (strain 2), which can infect O. nivara and cultivars with the resistance gene, appeared in the Philippines (Cabauatan & Hibino, 1983; Cabauatan et al., 1985; Hibino et al., 1985a). The wilted stunt strain too can infect O. nivara (Chen & Chiu, 1982). Moreover, grassy stunt isolates from India can infect O. nivara or cultivars with the O. nivara gene (Ghosh et al., 1979; Mariappan et al., 1984). In the field, cultivars that are resistant to the vector show decreased infection with the virus. Insecticide application to decrease vector populations is not always successful, especially in regions where rice is grown throughout the year. The vector is known to make long distance migratory flights (Kisimoto, 1971) to colonize rice in parts of China, Japan and Korea where the vector cannot overwinter. In Japan, about 0.1% of vector insects migrating across the ocean carry the virus (Hirao et al., 1984; Iwasaki et al., 1985b). In the Philippines, 0.5-1.2% of vectors caught in light traps carried the virus (Flores et al., 1986).
Notes
Small spherical virus-like particles, 18-25 nm, were reported in rice and vector planthoppers infected with the grassy stunt virus (Pellegrini & Bassi, 1978; Shikata et al., 1980; Hibino et al., 1982; Hibino et al., 1985b). Shikata et al. (1980) reported that the infectivity of a fraction containing numerous 20 nm particles was high and concluded that these particles were most probably the causal agent of grassy stunt. The spherical particles were not further characterized and their relation to grassy stunt disease is unknown.
Figures

Rice plants cv. Taichung Native 1, 6 weeks after infection with strain 1 (left) or strain 2 (centre); healthy plant on right.

Rice plants cv. Taiching Native, 1,90 days after infection with wilted stunt (GSW), Y (GSY), or B (GSB) strains; healthy plant at right (CK) (Courtesy of C. C. Chen.)

Nilaparvata lugens, a vector of grassy stunt virus; macropterous male (above), brachypterous female (below).

Bundles of fibrils in the nucleus (N) and cytoplasm of a rice grassy stunt virus-infected leaf cell. Bar represents 500 nm.
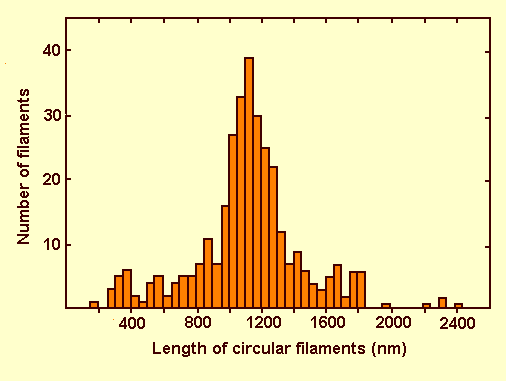
Contour length distribution of circular rice grassy stunt virus particles stained with 1% uranyl acetate.

Immunodiffusion tests: (above), antiserum to strain 2 of rice grassy stunt virus (centre well), strain 1 (1), strain 2 (2), clarified healthy sap (H), and buffer (B); (below), antiserum to rice stripe virus (centre well), grassy stunt virus (G), rice stripe virus (R), healthy sap (H), and buffer (B).
References list for DPV: Rice grassy stunt virus (320)
- Abeygunawardena, in Virus Diseases of the Rice Plant, p. 53, Baltimore: Johns Hopkins, 354 pp., 1969.
- Aguiero & Ling, Philipp. Phytopath. 14: 89, 1978.
- Anjaneyulu, Curr. Sci. 43: 416, 1974.
- Bergonia, Capule, Novero & Calica, Philipp. J. Pl. Ind. 31: 47, 1966.
- Cabauatan & Hibino, Int. Rice Res. Newsl. 8(2): 12, 1983.
- Cabauatan, Hibino, Lapis, Omura & Tsuchizaki, Int. Rice Res. Pap. Ser. 106: 1, 1985.
- Chen, Pl. Prot. Bull. Taiwan 25: 245, 1983.
- Chen, Pl. Prot. Bull. Taiwan 26: 315, 1984.
- Chen & Chiu, Pl. Dis. 66: 15, 1982.
- Chen & Wang, Bull. Taichung Dist. agric. Improv. Stn 8: 9, 1984.
- Chen, Ko & Chiu, Pl. Prot. Bull. Taiwan 21: 368, 1979.
- Chettanachit, Putta, Belaveang, Hongkajorn & Disthaporn, Int. Rice Res. Newsl. 10(2): 10, 1985.
- Disthaporn, Chettanachit & Putta, Int. Rice Res. Newsl. 8(6): 12, 1983.
- Flores, Hibino & Perfect, Int. Rice Res. Newsl. 11(4): 26, 1986.
- Gingery, Nault & Bradfute, Virology 172: 99, 1981.
- Gingery, Nault & Yamashita, J. gen. Virol. 64: 1765, 1983.
- Ghosh, John & Rao, Pl. Dis. Reptr 63: 523, 1979.
- Hibino, Rev. Pl. Prot. Res. 12: 98, 1979.
- Hibino & Cabauatan, Int. Rice Res. Newsl. 8(2): 12, 1983.
- Hibino, Iwasaki & Izumi, Ann. phytopath. Soc. Japan 43: 388, 1982.
- Hibino, Usugi, Omura & Shohara, Int. Rice Res. Newsl. 8(1): 9, 1983a.
- Hibino, Usugi, Tsuchizaki, Iwasaki & Izumi, Int. Rice Res. Newsl. 8(2): 11, 1983b.
- Hibino, Cabauatan, Omura & Tsuchizaki, Int. Rice Res. Newsl. 8(6): 11, 1983c.
- Hibino, Cabauatan, Omura & Tsuchizaki, Pl. Dis. 69: 538, 1985a.
- Hibino, Usugi, Omura, Tsuchizaki, Shohara & Iwasaki, Phytopathology 75: 894, 1985b.
- Hirao, Inoue & Oya, Appl. Ent. Zool. 19: 257, 1984.
- Hsieh & Chiu, Pl. Prot. Bull. Taiwan 12: 136, 1970.
- Iwasaki & Shinkai, Ann. phytopath. Soc. Japan 45: 741, 1979.
- Iwasaki, Nakano & Shinkai, Ann. phytopath. Soc. Japan 46: 411, 1980a.
- Iwasaki, Nakano & Shinkai, Proc. Assoc. Pl. Prot. Kyushu 26: 7, 1980b.
- Iwasaki, Nakano & Shinkai, Proc. Assoc. Pl. Prot. Kyushu 28: 1, 1982.
- Iwasaki, Nakano & Shinkai, Proc. Assoc. Pl. Prot. Kyushu 30: 1, 1984.
- Iwasaki, Nakano & Shinkai, Ann. phytopath. Soc. Japan 51: 351, 1985a.
- Iwasaki, Nakano & Shinkai, Ann. phytopath. Soc. Japan 51: 450, 1985b.
- Khush & Ling, J. Hered. 65: 134, 1974.
- Kisimoto, in Proc. Symp. on Rice Insects, p. 201, Tropical Agriculture Research Center, Tokyo, 332 pp., 1971.
- Koganezawa, Doi & Yora, Ann. phytopath. Soc. Japan 41: 148, 1975.
- Ling, in Rice Virus Diseases, p. 142, International Rice Research Institute, Los Banos, Philippines, 142 pp., 1972.
- Ling & Aguiero, Int. Rice Res. Newsl. 3: 6, 1967.
- Ling, Lee & Aguiero, Philipp. Phytopath. 5: 8, 1969.
- Ling, Aguiero & Lee, Pl. Dis. Reptr 54: 565, 1970.
- Mariappan, Hibino & Shanmugam, Int. Rice Res. Newsl. 9(6): 9, 1984.
- Morales & Niessen, Phytopathology 73: 971, 1983.
- Nuque, Aguiero & Ou, Pl. Dis. 66: 63, 1982.
- Omura, Hibino, Usugi, Inoue, Morinaka, Tsurumachi, Ong, Putta, Tsuchizaki & Saito, Pl. Dis. 68: 374, 1984.
- Ou & Rivera, in The Virus Diseases of the Rice Plant, p. 23, Baltimore: Johns Hopkins, 354 pp., 1969.
- Palmer, Soepriaman & Kartaatmadja, Pl. Dis. Reptr 62: 962, 1978.
- Palomar & Ling, Philipp. Phytopath. 4: 14, 1968.
- Pellegrini & Bassi, Phytopath. Z. 92: 247, 1978.
- Raychaudhuri, Mishra & Ghosh, Pl. Dis. Reptr 51: 300, 1967.
- Rivera, Ou & lida, Pl. Dis. Reptr 50: 453, 1966.
- Senboku & Shikata, Ann. phytopath. Soc. Japan 46: 487, 1980.
- Senboku, Ishimizu, Shikata, Tiongco & Ling, Ann. phytopath. Soc. Japan 46: 411, 1980.
- Shikata, Senboku & Ishimizu, Proc. Japan Acad. Ser. B 56: 89, 1980.
- Tantera, Satomi & Roechan, Contr. Central Res. Inst. Bogor, Indonesia, No. 2, p. 8, 1973.
- Toriyama, J. gen. Virol. 61: 187, 1982.
- Toriyama, Ann. phytopath. Soc. Japan 51: 59, 1985.
- Toriyama & Kojima, Ann. phytopath. Soc. Japan 51: 358, 1985.
- Wathanakul & Weerapat, in The Virus Diseases of the Rice Plant, p. 79, Baltimore: Johns Hopkins, 354 pp., 1969.