Details of DPV and References
DPV NO: 322 December 1986
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Northern cereal mosaic virus | Acronym: NCMV
Northern cereal mosaic virus
S. Toriyama National Institute of Agrobiological Resources, Tsukuba Science City, Yatabe, Ibaraki 305, Japan
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Ito & Fukushi (1944a,
1944b;
1956).
Synonym
- Wheat rosette stunt virus (Anon., 1979)
-
A virus with enveloped bacilliform RNA-containing particles, c. 350 x 68 nm. It is not mechanically transmissible, but is transmitted by planthoppers in the persistent, circulative, manner. Its host range is limited to Gramineae and it causes chlorotic stripe and severe stunt diseases of oat, barley and wheat. The virus occurs in Japan, Korea and China.
Main Diseases
Disease caused by northern cereal mosaic virus is often economically important in north east Japan in oat, barley and wheat, but not in rye (Ito & Fukushi, 1944a, 1944b). Serious disease occurs on barley in Korea (Lee & Shikata, 1977) and on wheat in northern China (Anon., 1979; Liu & Chen, 1982). Primary symptoms of infection consist of chlorotic spots on the basal parts of elongating leaves. The chlorotic spots coalesce and develop into chlorotic stripe or mosaic symptoms. The mosaic symptoms differ slightly with plant species; they usually fade as the plant ages and temperatures rise. Leaves developing later are short and narrow and show chlorotic stripes or generalized yellowing. The growth of infected plants is much decreased and chlorotic tillers develop one by one, resulting in a stunted rosette appearance (Fig. 1, Fig. 2). Affected oat plants often develop up to 20-30 tillers (Ito & Fukushi, 1944a, 1944b). Field-infected wheat plants may be only 20-50% as high as uninfected ones. In wheat plants infected early, the panicles are either not formed or, if produced, are small and/or malformed and with low quality grain. In a wheat field in which more than 50% of the plants were diseased, the grain yield was decreased by 75% (Ogawa & Moichi, 1984). Annual bluegrass (Poa annua) is thought to be ecologically important as a carrier of the virus (Ito & Fukushi, 1944a, 1944b).
Geographical Distribution
The northern cereal mosaic disease was first recognized in 1910 on barley in northern Japan. Since then the incidence of the causal virus has increased gradually and it caused severe damage to oat in years around 1939 (Ito & Fukushi, 1944a, 1944b) and also in 1962. Recently the virus has been prevalent again on wheat and barley in northern Japan (Ogawa & Moichi, 1984); it also occurs in central Japan, but it is more localized and less important there (Shinkai, 1968; Toriyama & Yora, 1969). The virus also occurs in South Korea (Lee & Shikata, 1977) and northern China (Liu & Chen, 1982).
Host Range and Symptomatology
The virus is not transmitted by manual inoculation with sap, but is transmitted by planthoppers. Natural hosts are barley, Italian millet, oat, rye, Italian ryegrass, wheat, Digitaria violascens, Poa annua, Setaria pycrocoma and S. viridis. Experimentally, 31 species of 18 genera in the family Gramineae are susceptible (Ito & Fukushi, 1944a, 1944b; Shinkai, 1955; Shinkai et al., 1968; Mikoshiba et al., 1984). Bromus inermis, Dactylis glomerata, Oryza sativa, Poa pratensis and Zea mays are not infected (Ito & Fukushi, 1944a, 1944b). The host ranges of isolates of the wheat rosette and northern cereal mosaic strains were similar but not identical (Liu & Chen, 1982; Lin et al., 1983).
-
Diagnostic species
- Hordeum vulgare.
In young seedlings, chlorotic spots or dashes appear on the basal parts of developing leaves 4 to 5 days after inoculation by viruliferous planthoppers, later developing into mosaic or chlorotic stripes (Fig. 1). Leaves developing later show chlorotic stripes rather than mosaic symptoms. Elongation of the leaves is extremely suppressed and many fine (sometimes needle-like) chlorotic tillers develop, resulting in a stunted rosette. -
Avena sativa. The symptoms are similar to those in barley, and are frequently
accompanied by development of reddish-purple colour in the tips or margins of infected
leaves.
- Zea mays (cv. Golden Cross Bantam, Longfellow, Yellow Dent) and Poa pratensis are thought to be non-hosts.
-
Propagation species
- Virus cultures are best maintained in barley, oat, wheat, Lolium multiflorum or Poa annua by transmission by the planthopper Laodelphax striatellus. Systemically infected tissues of barley are a suitable source of virus for purification.
-
Assay species
- No local lesion assay is available. The virus may be assayed by determining the proportion of barley or oat seedlings that become infected following inoculation by viruliferous planthoppers.
Strains
Wheat rosette stunt virus (Anon., 1979) is serologically closely related to northern cereal mosaic virus (Lundsgaard et al., 1984; Gong et al., 1985; Milne et al., 1986). These two viruses also have similar host ranges (Liu & Chen, 1982) and particle morphology (Anon., 1979), suggesting that they are either identical or closely related strains (Liu & Chen, 1982; Lundsgaard et al., 1984).
Transmission by Vectors
The most important vector is the small brown planthopper, Laodelphax striatellus (Delphacodes striatella) (Ito & Fukushi, 1944a, 1944b); three other species are also known as vectors: Unkanodes albifascia (Ishii & Matsumoto, 1964), U. sapporona (Shinkai, 1966) and Muellerianella fairmairei (Ishii, 1967). In the field, the virus has been detected in hibernating nymphs of L. striatellus, but not of U. albifascia (Ishii, 1966). The latent period in L. striatellus is reported as 9 to 20 days (Ito & Fukushi, 1944a, 1944b) or 4 to 6 days (Shinkai, 1969). In U. albifascia, the latent period is about 2 weeks (Shinkai, 1969). When L. striatellus were reared on diseased plants for 1 or 10 days, the proportion that became viruliferous was 17% and 57% respectively (Ito & Fukushi, 1944a, 1944b). In contrast, Shinkai (1960) found that almost all the planthoppers reared on diseased plants for 10 days became viruliferous. The minimum inoculation feeding time is 5 to 10 min; most plants are infected after an inoculation feeding time of 2 h. Viruliferous planthoppers can transmit the virus daily or sporadically for up to about 60 days. The virus is reported not to be transmitted through eggs to the progeny of the vector (Ito & Fukushi, 1944a, 1944b; Toriyama & Yora, 1969).
Transmission through Seed
Not detected in Avena sativa, Hordeum vulgare or Triticum aestivum (Ito & Fukushi, 1944a, 1944b).
Serology
Preparations of intact virus particles are moderately immunogenic, giving antisera in rabbits with titres of 1/128 to 1/256 in the tube precipitin test (Toriyama, 1972) and 1/512 in the ring precipitin test (Kung & Zheng, 1985). On the other hand, preparations of the inner nucleocapsid are strongly immunogenic, giving antisera with titres of 1/1024 in gel diffusion tests (Shirako & Ehara, 1985). Agar gel diffusion tests are useful for serological comparisons (Lundsgaard, 1984). Enzyme-linked immunosorbent assay has proved useful for the detection of inner nucleocapsid antigen in infected plants and in viruliferous planthoppers (Shirako & Ehara, 1985). Immunosorbent electron microscopy has also been successful (Lundsgaard, 1984; Lundsgaard et al., 1984; Milne et al., 1985).
Relationships
Milne et al. (1986), using electron microscope serological tests (‘decoration’), found that northern cereal mosaic virus is closely related or identical to wheat rosette stunt virus but distantly related (SDI = 6) to barley yellow striate mosaic virus. Lundsgaard (1984) found no serological relationship between northern cereal mosaic virus and festuca leaf streak virus. For more information about the interrelationships of rhabdoviruses infecting cereals, see barley yellow striate mosaic virus. The fact that the particles of northern cereal mosaic virus mature in the cytoplasm indicates that it should be placed in sub-group 1 of the rhabdovirus group.
Stability in Sap
In extracts from infected plants or viruliferous planthoppers, assayed by the planthopper injection method, the thermal inactivation point (10 min) was 50 to 55°C, the dilution end-point was 10-2 to 10-3 (plant extracts) or 10-3 to 10-4 (insect extracts), and the longevity in vitro was 4 to 5 days at 5°C. Infectivity was retained for at least 1 year in diseased plants stored at -35°C (Murayama & Lu, 1967).
Purification
(Toriyama, 1972;
Toriyama & Peters, 1981).
Grind fresh infected barley
tissue (80 g) with 200 ml 0.1 M glycine (pH 7.2) and centrifuge the homogenate at
3000 g for 5 min. Mix the supernatant fluid with Celite (Hyflo Super-Cel)
and pass through the Celite filter in a Buchner funnel, until the filtrate becomes
pale green or brown. Centrifuge the filtrate at 30,000 g for 60 min.
Resuspend the pellet in 0.1 M glycine, pH 7.2, and centrifuge in 5-35% sucrose density
gradients at 60,000 g for 30 min. Collect the virus zone, layer on a
30-60% sucrose density gradient and centrifuge at 69,000 g for 120 min.
Recover the virus particles by centrifugation
(Fig. 3).
The virus preparations can be
further purified by zone electrophoresis
(Toriyama, 1972).
Preparations of the inner nucleocapsid may be obtained by the following method (Shirako & Ehara, 1985): Blend fresh or frozen tissue (100 g) with 250 ml 0.1 M ammonium citrate, pH 6.5, containing 0.1% Na-DIECA, 0.25% 2-mercaptoethanol and 1% polyvinyl pyrollidone. Mix the homogenate with Triton X-100 to 2% final concentration, layer the fluid over a 20% sucrose cushion and centrifuge at 66,000 g for 120 min. Resuspend the pellet in 0.1 M ammonium citrate, pH 6.5, and after low speed centrifugation, centrifuge the supernatant fluid through a 10-40% linear sucrose density gradient at 60,000 g for 120 min. The nucleocapsids form a zone at the centre of the tube and may be sedimented from fractions containing the zone by centrifugation. Purify further by equilibrium centrifugation in CsCl.
Particle Structure
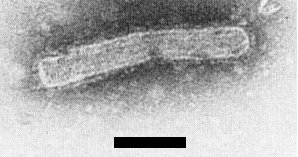
The virus has enveloped bacilliform particles (Fig. 3, Fig. 4, Fig. 5), measuring 300-350 x 60 nm, with a dense inner structure c. 40 nm wide in dip preparations (Shikata & Lu, 1967). In partially purified preparations, Lu et al. (1968) reported that the particles were 500-600 x 40 nm, but Toriyama (1972) found that purified enveloped particles, retaining high infectivity, were 330-380 nm (average 350 nm) x 68 nm; one end was round and the other slightly flattened (Fig. 3, Fig. 4). Occasionally, double-length particles are found. Distinct cross-striations are visible in particles purified with buffer containing EDTA and stained with uranyl acetate (Toriyama, 1972) (Fig. 4); they are less obvious in 2% phosphotungstate. The helical tubular inner nucleocapsid, obtained by removing the outer membrane with Triton X-100, is 28 nm wide and has a pitch reported as 5.5 nm (Shirako & Ehara, 1985) or 52 nm (Gong et al., 1985). The pitch of the cross striations formed by the G protein spikes of intact particles was about 4.5 nm (Gong et al., 1985).
Particle Composition
Nucleic acid: RNA
(Toriyama, 1972),
M. Wt c. 35 x 106,
estimated by electrophoresis in 1% agarose gel after formaldehyde denaturation
(Shirako & Ehara, 1985).
Molar percentage of nucleotides: G 16.3; A 30.3; C 16.7;
U 36.6
(Zheng et al., 1985).
Protein: The northern cereal mosaic strain has three major protein species,
of M. Wt 63 x 103 (G), 47 x 103 (N) and 19 x 103 (M),
and one minor species of M. Wt 88 x 103. Two species of polypeptide, of M.
Wt 47 x 103 and 45 x 103, were detected in preparations of
purified nucleocapsids; the smaller one is a partial degradation product of N protein
(Shirako & Ehara, 1985).
The wheat rosette stunt strain was found to have five
major protein species, of M. Wt (x 10-3) 140 (L), 66 (G), 46 (N), 40 (NS)
and 19 (M); protein N separates into two components, N1 and N2,
of M. Wt (x 10-3) 46 and 44 respectively, suggesting that N2
may be derived from N1
(Zheng et al., 1983;
Kung & Zheng, 1985;
Gong & Zhen, 1985;
Gong et al., 1985).
Lipid: Phospholipids (phosphatidyl ethanolamine and phosphatidyl serine) and sterols were detected in highly purified preparations (Toriyama, 1972). The percentage sterol composition of the virus particles (cholesterol : campesterol : stigmasterol : b-sitosterol = 10.7 : 12.9: 25.6: 50.8) was different from that of the barley host plant and its microsomal fraction (endoplasmic reticulum). The percentage of cholesterol in the virus particles was remarkably high (Toriyama, 1976a).
Relations with Cells and Tissues
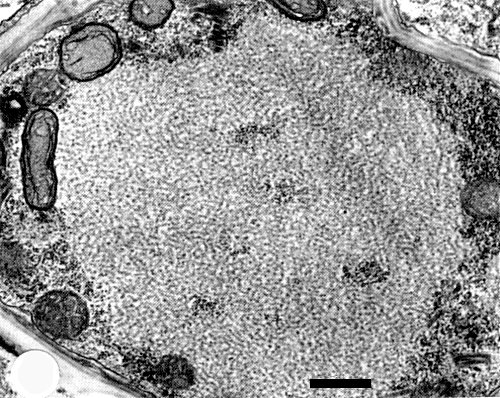
A single cytoplasmic round to ellipsoidal inclusion body (X-body), 5-9 x 3.5-6.5 µm, is observed in each cell of the young tiller (Ito & Fukushi, 1944a, 1944b) and in infected leaf epidermal cells (Shinkai, 1960). Virus particles accumulate in the cytoplasm within cisternae of the endoplasmic reticulum (Shikata & Lu, 1967; Toriyama, 1976b) (Fig. 6). Most cell types are infected: mesophyll cells, phloem parenchyma cells, companion cells, sieve elements, developing xylem cells, guard cells and epidermal cells. Prominent viroplasm-like structures which appear as an electron-dense matrix of granular or fibrous materials, are observed in the cytoplasm of infected cells. Fully formed particles appear around the viroplasm-like structure (Fig. 8). Membranous and filamentous inclusions also appear in the cytoplasm. Non-enveloped particles, c. 330 x 40 nm, were observed especially in membranes that protruded into central vacuoles (Toriyama, 1976b) (Fig. 7). Electron microscopical observations showed that the virus infects phloem cells first and then spreads to adjacent mesophyll cells (Toriyama, 1976b). Infection with the virus suppresses elongation of cells (Toriyama & Kuwata, 1973). The cell wall compositions of barley and wheat are altered by the infection: the proportion of a-cellulose (glucose) decreases and that of the hemicellulose fraction (xylose, arabinose) increases (Toriyama, 1977). Tests in which virus preparations were injected into viruliferous planthoppers (L. striatellus) showed that the virus multiplies in the vector and occurs in high concentration in the salivary glands, fat body and gut, even when cultured for 12 days after excision from planthoppers reared on infected plants (Yamada et al., 1967; Yamada & Shikata, 1969). Long tubular particles, 500-600 x 40 nm, were observed frequently in salivary glands but rarely in the fat body of viruliferous planthoppers, and no typical bacilliform particles were seen (Shikata & Lu, 1967).
Notes
Northern cereal mosaic disease was at first called ‘cereal stunt disease of Hokkaido’, but when the disease was proved to be a new one, the present name was adopted to avoid confusion with other soil-borne virus diseases, namely wheat yellow mosaic, barley yellow mosaic and soil-borne wheat mosaic, which were prevalent in the western and central part of Japan (Ito & Fukushi, 1944a, 1944b). The chlorotic stripe symptoms caused in wheat and barley in the early stages of infection by northern cereal mosaic virus are similar to those caused by rice stripe virus, but the early development of new tillers, characteristic of infection by northern cereal mosaic virus, is not seen with rice stripe virus.
Figures
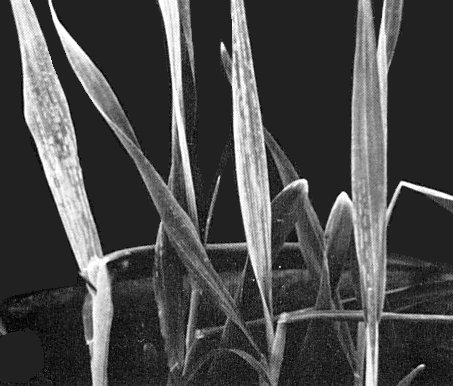
Initial symptoms on barley leaves. Note chlorotic spots and chlorotic stripes on young developing leaves. (Courtesy M. Kojima.)

Infected and stunted barley plants. The plants are one-fourth to one-fifth as high as healthy ones (not shown). Note the many chlorotic tillers in the plant on the left and the blighted lower leaves of the two plants to the right.

Bacilliform particles, extracted from infected barley plants and purified by sucrose density gradient centrifugation. Bar represents 500 nm.
Fine structure of virus particles: particles showing transverse striations, the outer membranous layer (slightly degraded) and the inner canal; bar represents 100 nm.
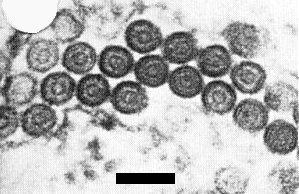
Fine structure of virus particles: cross-sections of virus particles in a wheat leaf section showing the lipid bilayer and the nucleocapsid; bar represents 100 nm.
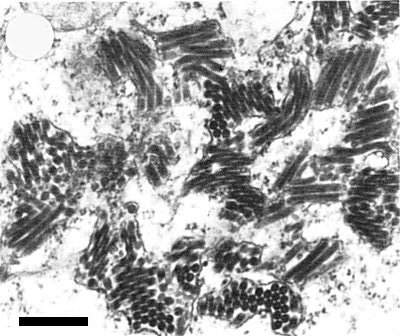
Accumulations of virus particles in the cisternae of the endoplasmic reticulum. Bar represents 500 nm.
References list for DPV: Northern cereal mosaic virus (322)
- Anon., Scientia sin. 22: 573, 1979.
- Gong & Zhen, Acta Biochim. Biophys. sin. 17: 650, 1985.
- Gong, Zhen, Peng, Tsao & Shikata, Chinese J. Virol. 1: 257, 1985.
- Ishii, Res. Bull. Hokkaido natn. agric. Exp. Stn 89: 49, 1966.
- Ishii, Jap. J. appl. Ent. Zool. 11: 191, 1967.
- Ishii & Matsumoto, Ann. phytopath. Soc. Japan 29: 280, 1964.
- Ito & Fukushi, J. Sapporo Soc. Agric. For. 36: 62, 1944a.
- Ito & Fukushi, J. Sapporo Soc. Agric. For. 36: 65, 1944b.
- Ito & Fukushi, Ann. phytopath. Soc. Japan 21: 3, 1956.
- Kung (= Gong) & Zheng (= Zhen), Acta Biochim. Biophys. sin. 17: 248, 1985.
- Lee & Shikata, Korean J. Pl. Prot. 16: 87, 1977.
- Lin, Ruan & Jin, Scientia agric. sin., 1983: 64, 1983.
- Liu & Chen, Scientia agric. sin., 1982: 8, 1982.
- Lu, Shikata & Murayama, Mem. Fac. Agric. Hokkaido Univ. 6: 335, 1968.
- Lundsgaard, Intervirology 22: 50, 1984.
- Lundsgaard, Tien & Toriyama, Phytopath. Z. 111: 232, 1984.
- Milne, Masenga & Conti, Intervirology 25: 83, 1986.
- Mikoshiba, Nemoto & Gomi, Ann. phytopath. Soc. Japan 50: 108, 1984.
- Murayama & Lu, J. Fac. Agric. Hokkaido Univ. 55: 182, 1967.
- Ogawa & Moichi, Ann. phytopath. Soc. Japan 50: 108, 1984.
- Shinkai, Ann. phytopath. Soc. Japan. 19: 40, 1955.
- Shinkai, in Plant Virus Diseases, p. 275, eds J. Hidaka, T. Hirai, D. Murayama & K. Yora, Tokyo: Asakura Pub. Co. Ltd., 400 pp., 1960.
- Shinkai, Ann. phytopath. Soc. Japan 32: 317, 1966.
- Shinkai, Shokubutsu Boeki 22: 445, 1968.
- Shinkai, Shokubutsu Boeki 23: 380, 1969.
- Shinkai, Kureha & Suzuki, Ann. phytopath. Soc. Japan 34: 376, 1968.
- Shikata & Lu, Proc. Japan Acad. 43: 918, 1967.
- Shirako & Ehara, Phytopathology 75: 453, 1985.
- Toriyama, Virus 22: 114, 1972.
- Toriyama, Ann. phytopath. Soc. Japan 42: 494, 1976a.
- Toriyama, Ann. phytopath. Soc. Japan 42: 563, 1976b.
- Toriyama, Ann. phytopath. Soc. Japan 43: 126, 1977.
- Toriyama & Kuwata, Ann. phytopath. Soc. Japan 39: 136, 1973.
- Toriyama & Peters, J. gen. Virol. 56: 59, 1981.
- Toriyama & Yora, Ann. phytopath. Soc. Japan 35: 360, 1969.
- Yamada & Shikata, J. Fac. Agric. Hokkaido Univ. 56: 91, 1969.
- Yamada, Shikata & Murayama, Ann. phytopath. Soc. Japan 33: 97, 1967.
- Zheng (=Zhen), Xü, Zhu, Shen & Kung (=Gong), Acta Biochim. Biophys. sin. 15: 561, 1983.