Details of DPV and References
DPV NO: 327 September 1988
Family: Caulimoviridae
Genus: Soymovirus
Species: Blueberry red ringspot virus | Acronym: BRRV
Blueberry red ringspot virus
J. M. Gillett Dept. of Botany and Plant Pathology, Michigan State University, East Lansing, Michigan 48824, USA
D. C. Ramsdell Dept. of Botany and Plant Pathology, Michigan State University, East Lansing, Michigan 48824, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease described by Hutchinson (1950) and Hutchinson & Varney (1954).
A virus with spherical particles 42-46 nm in diameter containing double-stranded DNA. The particles occur embedded in cytoplasmic inclusion bodies and are also found in the nucleus. Found in nature only in Vaccinium spp. Graft-transmissible from Vaccinium to Vaccinium. Vector unknown.
Main Diseases
Causes red spots and rings 2-6 mm in diameter on leaves of highbush blueberry, Vaccinium corymbosum and V. australe (Fig. 1, Fig. 2, Fig. 3, Fig. 4). The symptoms appear on older leaves in mid to late summer and progress to younger leaves later in the growing season. The spots may coalesce, especially on older leaves. Stems (canes) one year old or more may also exhibit red spots, rings, and blotches (Fig. 2) (Stretch & Varney, 1970; Ramsdell et al., 1987). Fruit from diseased bushes is usually smaller and ripens 1 week earlier than fruit from uninfected bushes in the same field (Gillett, 1988). The disease can also significantly decrease yield (Gillett, 1988). Fruit from infected bushes may have circular light areas (Fig. 3). A powdery mildew (Microsphaera alni var. vaccinii) may cause similar leaf symptoms, but the red spots can be seen on both sides of the leaf and the lower leaf surface usually has a water-soaked appearance.
Geographical Distribution
Reported only from USA. The disease is economically most important in New Jersey and is widespread in plantings in Arkansas (Kim et al., 1981). The disease has also been reported from Michigan, Connecticut, Massachusetts, New York, North Carolina, and most recently in Oregon (Converse & Ramsdell, 1982).
Host Range and Symptomatology
Vaccinium spp. are the only known hosts (Kim et al., 1981). The disease has been observed in many cultivars, including Blueray, Bluetta, Burlington, Cabot, Coville, Darrow, Earliblue and Rubel. The cultivar Jersey is apparently field-immune and the cultivar Bluecrop is field-resistant. The virus can be transmitted by chip-budding and whip-grafting (Stretch & Scott, 1977). Blueberry red ringspot disease symptoms are similar to those of ringspot disease in cranberry (V. macrocarpon). Electron microscope studies of affected cranberry show inclusion bodies similar to those found in blueberry tissue infected with red ringspot virus (K. S. Kim & A. W. Stretch, unpublished data). These two diseases may be caused by the same virus.
Strains
None reported.
Transmission by Vectors
None reported. However, aphid and/or mealybug transmission is suspected (J. M. Gillett & D. C. Ramsdell, unpublished data).
Transmission through Seed
None reported.
Serology
No antiserum was produced when purified virus emulsified in Freund’s adjuvant was injected into a rabbit (Kim et al., 1981). However the virus is moderately immunogenic in rabbits if it is fixed in 0.5% (v/v) formaldehyde prior to emulsification and injection (Gillett & Ramsdell, 1984). After four intramuscular injections of fixed, emulsified virus, antiserum was obtained with a titre of 1/128 against 100 µg/ml purified virus in agar gel double diffusion tests. Crude sap from diseased plants does not react in agar gel double diffusion tests but reacts in ELISA (Gillett & Ramsdell, 1984; Hepp & Converse, 1987).
Relationships
Purified particles of blueberry red ringspot virus at 100 µg/ml gave no reaction in agar gel double diffusion tests with antisera to cauliflower mosaic (Kim et al., 1981), carnation etched ring, dahlia mosaic or figwort mosaic viruses (Gillett, 1988). In the same tests, purified cauliflower mosaic virus particles gave no reaction with an antiserum to blueberry red ringspot virus.
Stability in Sap
No information.
Purification
Gillett & Ramsdell (1984) developed the following purification procedure, which is a modification of the methods for cauliflower mosaic virus (Hull et al., 1976) and carnation etched ring virus (Lawson & Civerolo, 1978). Homogenize each 100 g frozen symptom-bearing blueberry leaves in 600 ml 0.1 M sodium phosphate buffer, pH 7.2, containing 0.01 M 2-mercaptoethanol and 0.005 M thioglycollic acid. Add butan-1-ol to 8% (v/v), urea to 6% (w/v) and Triton X-100 to 2.5% (v/v). Stir overnight at 4°C. Squeeze through cheesecloth. In the resulting extract, dissolve polyethylene glycol (M. Wt 6000) to 8% (w/v) and sodium chloride to 1% (w/v). Centrifuge at 10,000 g for 30 min and resuspend the pellet overnight in 60 ml 0.01 M sodium phosphate, pH 7.2. Follow with one cycle of differential centrifugation, resuspending the pellet as before. Layer on 0-30% linear-log sucrose gradients and centrifuge in a Beckman SW41 rotor at 38,000 rev./min for 60 min. Further purify the virus in a CsCl step gradient (1 ml each of r = 1.55, 1.45, 1.40 and 1.35 g/cm3), centrifuging for 15 h in a Beckman SW 50.1 rotor at 35,000 rev./min.
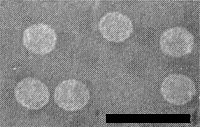
Properties of Particles
The particles sediment as two peaks in sucrose density gradients; however, no morphological differences were observed between populations of particles from the two peaks (Kim et al., 1981).
Sedimentation coefficients (s°20,w) = 212> S ± 5% and 275 S (by extrapolation).
Isoelectric point: pH 4.0.
Electrophoretic mobility: 24.5 x 10-6 cm2 sec-1 volt-1 at pH 5.0 in 0.02 M Na2HPO4, 0.02 M Tris, titrated with citric acid (determinations were made in 0.7% agarose gels).
A 0.1%, 1cm, 260nm: c. 7.0.
Buoyant density in CsCl: 1.30 and 1.40 g/cm3.
Particle Structure
Isometric particles 42-46 nm in diameter (Fig. 5). No obvious surface structure is revealed in ammonium molybdate negative stain.
Particle Composition
Nucleic acid: Double-stranded DNA, Tm = 86.0°C in 0.15 M NaCl, 0.015 M citrate, pH 7.0, with hyperchromicity of 28.5% (Gillett, 1988).
Protein: Gel electrophoresis of purified virus boiled 3 min in the presence of sodium dodecyl sulphate reveals one major protein band of M. Wt 44,000. Sometimes a faint protein band of M. Wt c. 101,000 (by extrapolation) is present (Gillett, 1988).
Relations with Cells and Tissues
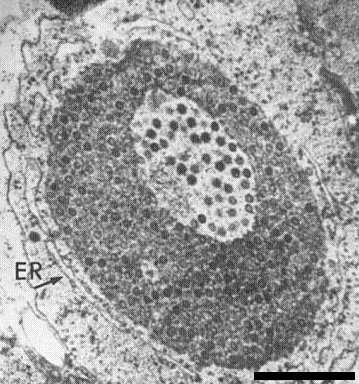
Cytoplasmic inclusion bodies have been observed in blueberry leaf samples expressing red ringspot symptoms (Fig. 6, Fig. 7) (Kim et al., 1981). They are well-defined circular bodies ranging from 0.5 to 2.5 µm in diameter and consist of a finely granular, electron-dense matrix in which homogeneous spherical virus particles are embedded. One to several electron-lucent areas containing well-defined virus particles may occur in each inclusion. All the inclusions observed in red ringspot-diseased leaves are enclosed in a layer of rough endoplasmic reticulum. Although most virus particles are found in cytoplasmic inclusion bodies, individual particles are occasionally observed in nuclei, scattered randomly, or grouped together near the nucleoli, or closely associated with lipid globules. Inclusion bodies have not been observed in nuclei.
Notes
Blueberry red ringspot virus resembles viruses in the caulimovirus group in most characteristics, notably nucleic acid type and strandedness, particle size and shape, and inclusion body type. However, when the virus particles are centrifuged in sucrose or CsCl gradients, two components are observed as opposed to one with typical caulimoviruses. Also, although the inclusion bodies induced by members of the caulimovirus group are often associated with ribosomes, none have been reported to be surrounded by a rough endoplasmic reticulum like that observed around the inclusions induced by blueberry red ringspot virus (Kim et al., 1981).
Figures

Shoot from blueberry (Vaccinium corymbosum) cv. Cabot infected with blueberry red ringspot virus and showing rings and spots on the leaves, spots on the stem, and decrease in leaf symptoms apically. (Courtesy of A. W. Stretch.)

Blueberry leaves (left to right): uninfected, and three infected leaves showing small circular, large circular, and coalesced lesions respectively.
References list for DPV: Blueberry red ringspot virus (327)
- Converse & Ramsdell, Pl. Dis. 66: 710, 1982.
- Gillett, M.Sc. Thesis, Michigan State Univ., 104 pp., 1988.
- Gillett & Ramsdell, Phytopathology 74: 862, 1984.
- Hepp & Converse, Pl. Dis. 71: 536, 1987.
- Hull, Shepherd & Harvey, J. gen. Virol. 31: 93, 1976.
- Hutchinson, Proc. 19th Ann. Blueberry Open House 19: 9, 1950.
- Hutchinson & Varney, Pl. Dis. Reptr 38: 260, 1954.
- Kim, Ramsdell, Gillett & Fulton, Phytopathology 71: 673, 1981.
- Lawson & Civerolo, Phytopathology 68: 181, 1978.
- Ramsdell, Kim & Fulton, in: Virus Diseases of Small Fruits, USDA Agriculture Handbook No. 631: 121, 1987.
- Stretch & Scott, Pl. Dis. Reptr 61: 605, 1977.
- Stretch & Varney, in: Virus Diseases of Small Fruits and Grapevines, Univ. California Press, p. 121, 1970.