Details of DPV and References
DPV NO: 328 September 1988
Family: Unallocated ssRNA+ viruses
Genus: Sobemovirus
Species: Ginger chlorotic fleck virus | Acronym: GCFV
Ginger chlorotic fleck virus
J. E. Thomas Agricultural Research Laboratories, Department of Primary Industries, Indooroopilly, Queensland 4068, Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Thomas (1986).
A virus with isometric particles c. 30 nm in diameter which sediment as a single component and contain one major species of ssRNA. The virus is mechanically transmitted to ginger, the only known host. No vector is known. Detected in Australia in some imported lines of ginger.
Main Diseases
Causes chlorotic flecks on the leaves of infected ginger (Zingiber officinale: Zingiberaceae). No obvious symptoms occur on the rhizomes. The effect on yield has not been studied. The virus is transmitted through the rhizome of infected plants.
Geographical Distribution
Detected in ginger introduced to Australia from India, Malaysia, Mauritius and Thailand. The imported material was grown in isolation in experimental plots and has since been destroyed. The virus has not been found in Australia in surveys of commercial ginger plantings (Thomas, 1986).
Host Range and Symptomatology
Transmitted by mechanical inoculation to ginger, the only known host (Thomas, 1986).
-
Diagnostic species
- Zingiber officinale
(ginger). The leaves of infected plants show chlorotic flecks, 1-10 mm long, parallel to and usually centred on the veins (Fig. 1). Symptoms first appear 3-4 weeks after inoculation in young leaves and persist in these and all subsequently formed leaves.Propagation and assay species
- Zingiber officinale;
no local lesion assay host is known.
Strains
All isolates studied appear serologically identical.
Transmission by Vectors
Vector not known. Not transmitted by the aphids Myzus persicae, Pentalonia nigronervosa, Rhopalosiphum maidis or R. padi (Thomas, 1986).
Transmission through Seed
No information.
Serology
Moderately immunogenic; an antiserum prepared in a rabbit had a gel diffusion titre of 1/512 (Thomas, 1986). Single lines of precipitate are obtained in gel diffusion tests against either purified virus or infective sap. Immunosorbent electron microscopy and a plate-trapped antigen form of ELISA are useful for virus detection.
Relationships
Ginger chlorotic fleck virus has many properties in common with viruses in the sobemovirus group (Matthews, 1982) including 30 nm-diameter particles which sediment as a single component (Fig. 4), a predominant ssRNA species of M. Wt 1.5 x 106, salt-labile particles and a limited host range. However some properties, including banding behaviour in CsCl and Cs2SO4 gradients, instability in potassium phosphotungstate and migration as two electrophoretic components, are atypical of this group. The virus is serologically unrelated to several members and possible members of the sobemovirus group, including cocksfoot mottle, lucerne transient streak, solanum nodiflorum mottle, southern bean mosaic, sowbane mosaic, turnip rosette and velvet tobacco mottle viruses. Additionally, no serological reactions were detected with antiserum to the following viruses with isometric particles; carnation mottle, cucumber fruit streak, cucumber mosaic, cymbidium ringspot, elm latent, galinsoga mosaic, glycine mottle, hibiscus chlorotic ringspot, narcissus tip necrosis, pelargonium flower-break, pelargonium ring pattern, pelargonium vein netting, phleum mottle, red clover necrotic mosaic, saguaro cactus, tobacco necrosis and turnip crinkle (Thomas, 1986).
Stability in Sap
No information.
Purification
Homogenize each 100 g fresh ginger leaves in 400-500 ml 0.5 M sodium citrate buffer, pH 6.5, containing 0.5% (w/v) Na2SO3. Filter the extract through cheesecloth, then stir with 25 ml chloroform for 10 min. Break the emulsion by centrifugation at 8000 g for 10 min. Collect the aqueous phase and subject to two cycles of differential centrifugation (235,000 g for 60 min and 8000 g for 10 min, resuspending the pellets each time in 50 mM sodium phosphate buffer, pH 7.0, containing 1 mM EDTA. Purify further by centrifugation in 10-40% linear sucrose density gradients. Yields are usually 50-90 mg/kg tissue (Thomas, 1986).
Similar yields are obtained when leaf tissue is homogenized in 0.25 M sodium citrate buffer, pH 6.5, containing 0.25% (w/v) Na2SO3, and clarified by adding butan-1-ol to 5% (v/v), leaving all other steps as previously described. Yields are greatly reduced when leaves are frozen at -20°C before use.
Properties of Particles
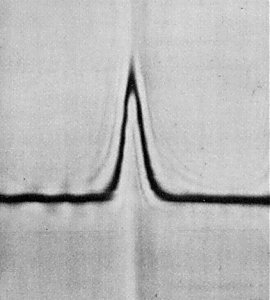
The particles sediment as a single component (Fig. 4).
Sedimentation coefficient, (s20,w): 111 S.
Electrophoretic mobility: migrates towards the anode as two approximately equimolar components.
Absorbance at 260 nm (A260 (0.1%, 1 cm)): 5.6.
A260/A280: 1.58 (not corrected for light-scattering).
Buoyant density in CsCl (g/cm3): 1.355 (particles fixed in glutaraldehyde).
Buoyant density in Cs2SO4 (g/cm3): 1.297 (at pH 4.9) or 1.293 (at pH 6.9).
Stability: particles are stable in 10 mM Tris, 10 mM NaCl, pH 8.25, but unstable in 10 mM Tris, 10 mM EDTA, 1 M NaCl, pH 8.25. Neutral potassium phosphotungstate and caesium chloride solutions also cause disruption of the particles (Thomas, 1986).
Particle Structure
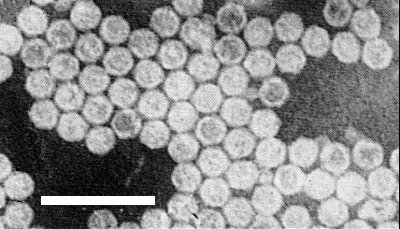
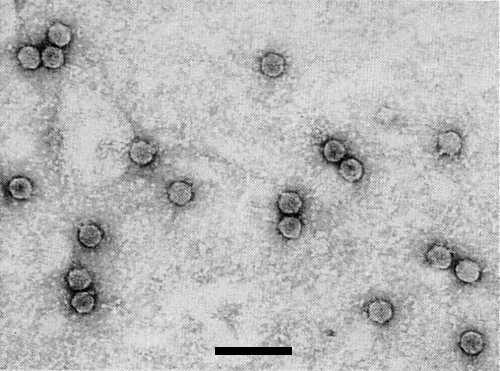
Particles are disrupted by mounting in neutral potassium phosphotungstate. In ammonium molybdate (Fig. 2) or uranyl acetate (Fig. 3) they are isometric, about 30 nm in diameter and have obvious hexagonal outlines. A proportion are penetrated by the negative stain (Thomas, 1986).
Particle Composition
Nucleic acid: RNA, single-stranded, comprising c. 20% of particle weight (estimated from the buoyant density in CsCl). Polyacrylamide gel electrophoresis under non-denaturing conditions indicates a M. Wt for the main component of 1.50 x 106. Minor components of M. Wt 0.59 x 106 and 0.14 x 106 are also detected in varying amounts (Thomas, 1986).
Protein: A predominant protein species of M. Wt 29.0 x 103, estimated by SDS-polyacrylamide gel electrophoresis. A minor species of M. Wt 27.3 x 103 is sometimes present (Thomas, 1986).
Relations with Cells and Tissues
Randomly distributed virus-like particles and membrane-bound vesicles have been observed in the cytoplasm of mesophyll cells from infected ginger (J. E. Thomas & D. H. Gowanlock, unpublished data).
Notes
Few virus diseases of ginger have been reported. Wheat mosaic streak virus described from India (Ganguly & Raychaudhuri, 1971; Basu & Raychaudhuri, 1972) differs in infecting wheat ( Triticum aestivum) and in being transmitted by the aphids Rhopalosiphum maidis and R. padi. Cucumber mosaic virus (‘ginger mosaic virus’; So, 1980) differs in particle properties, host range and aphid transmissibility, and is serologically distinct. A mosaic disease of ginger from India, described by Nambiar & Sarma (1974), has similar leaf symptoms but has not been sufficiently characterized to allow a detailed comparison.
Ginger chlorotic fleck virus is readily identified by serological tests.
Acknowledgements
Figs 1, 2 and 4 from Thomas (1986).
Figures
References list for DPV: Ginger chlorotic fleck virus (328)
- Basu & Raychaudhuri, Indian J. Ent. 34: 115, 1972.
- Ganguly & Raychaudhuri, Phytopath. Z. 70: 11, 1971.
- Matthews, Intervirology 17: 1, 1982.
- Nambiar & Sarma, Arecanut and Spices Bull. 6: 3, 1974.
- So, Korean J. Pl. Prot. 19: 67, 1980.
- Thomas, Ann. appl. Biol. 108: 43, 1986.