Details of DPV and References
DPV NO: 329 September 1988
Family: Unallocated ssRNA+ viruses
Genus: Sobemovirus
Species: Subterranean clover mottle virus | Acronym: SCMoV
There is a more recent description of this virus: DPV 387
Subterranean clover mottle virus
R. I. B. Francki Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064, Australia
J. W. Randles Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064, Australia
D. J. Graddon Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064, Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Francki et al. (1983).
A virus with isometric particles c. 30 nm in diameter containing single-stranded RNA of M. Wt c. 1.5 x 106. Most naturally occurring isolates also contain one or both of two circular single-stranded RNA molecules of 332 and 388 bases (M. Wt c. 0.13 x 106) which appear to be satellites. The virus has been isolated from Trifolium subterraneum and T. globeratum in south-western Australia and infects a narrow range of experimental hosts. It is readily transmitted by mechanical inoculation.
Main Diseases
The virus causes severe stunting, pronounced mottle and leaf distortion in subterranean clover (Trifolium subterraneum) (Fig. 1, Fig. 2). It has also been isolated from club clover (T. globeratum) (Francki et al., 1983, 1985).
Geographical Distribution
The virus appears to be endemic to Australia. It has been recovered only from clover stands in some areas of south-western Australia (Francki et al., 1983) as well as New South Wales, Victoria, South Australia and Tasmania (K. Helms & P. Waterhouse, personal communication).
Host Range and Symptomatology
Transmitted readily by sap inoculation to a few species in the Leguminosae.
-
Diagnostic species
- Trifolium subterraneum.
Systemically infected leaves develop a mottle or mosaic and the plants are severely stunted (Fig. 1, Fig. 2). - Medicago truncatula. Systemically infected leaves develop mosaic
and plants are stunted.
- Pisum sativum. Inoculated leaves develop necrotic local lesions (Fig. 3).
-
Propagation species
- T. subterraneum
and M. truncatula are good sources of virus for purification and are useful for maintaining cultures.Assay species
- P. sativum
is suitable as a local lesion assay host (Fig. 3).
Strains
No strains characterised.
Transmission by Vectors
No vectors are known.
Transmission through Seed
Virus was detected by ELISA in up to 10% of seeds obtained from commercial sources and in up to 3% of the seedlings obtained from this seed.
Serology
Strongly immunogenic; rabbit antisera had homologous titres of 1/1024 in double diffusion tests in 0.75% agar buffered with 10 mM phosphate, pH 7 (Francki et al., 1983). ELISA and single antibody dot immunoassay are sensitive and specific methods for detecting virus in plant extracts (Graddon & Randles, 1986).
Relationships
The properties of the virus place it in the sobemovirus group and it especially resembles velvet tobacco mottle, solanum nodiflorum mottle and lucerne transient streak viruses in having associated satellite RNA molecules. It is serologically very distantly related to lucerne transient streak virus, with a serological differentiation index of c. 7, but is unrelated to any of the other sobemoviruses tested, namely southern bean mosaic, sowbane mosaic, velvet tobacco mottle and solanum nodiflorum mottle viruses (Francki et al., 1983; D.J. Graddon, unpublished data).
Stability in Sap
No information.
Purification
A suitable method (Francki et al., 1983; Gould et al., 1981) is to extract 100 g lots of leaf material from plants showing prominent symptoms in 200 ml 0.1 M phosphate buffer, pH 7.4, containing 0.1% thioglycollic acid. Strain the extract through cheesecloth and clarify the filtrate by centrifugation at 10,000 g for 10 min. Emulsify the supernatant fluid with an equal volume of a 1:1 mixture of chloroform and butan-1-ol and centrifuge at 10,000 g for 10 min. Concentrate the virus from the aqueous supernatant fluid by centrifugation at 78,000 g for 90 min and resuspend the pellets in a small volume of 20 mM phosphate buffer, pH 7.4; repeat the differential centrifugation twice more. When highly purified virus is required, centrifuge the preparation in 5-25% sucrose gradients at 26,000 rev./min for 2 h in a Beckman SW 27 rotor. Recover the single band of virus and concentrate by centrifugation at 230,000 g for 2 h.
Properties of Particles
Virus particle preparations contain a single component co-sedimenting with particles of southern bean mosaic virus, which have a sedimentation coefficient (s20,w) of 115 S (Tremaine & Hamilton, 1983).
Absorption coefficient (A260 (0.1%, 1 cm)): c. 5.
A260/A280: c. 1.5.
Particle Structure
Particles negatively stained in uranyl acetate are hexagonal in shape and c. 30 nm in diameter (Fig. 4).
Particle Composition
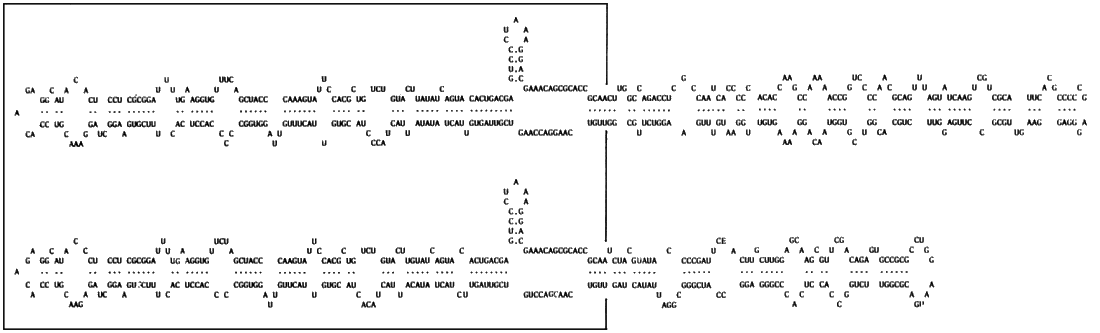
Nucleic acid. The particles contain ssRNA (RNA1) of M. Wt c. 1.5 x 106 (c. 4.5 kb). Most field isolates of the virus also contain one or both of two satellite RNA species of 332 or 388 nucleotides respectively, each of which exists as both circular (RNA2) and linear (RNA3) forms (Francki et al ., 1983; J. Haseloff, personal communication, 1987; Fig. 5, Fig. 6). The two satellite RNA species, whose nucleotide sequences (Fig. 8) display extensive regions with over 95% homology, can be present either singly or together in field isolates of the virus (Fig. 5). Isolates possessing only RNA1 have been found in the field (D. J. Graddon, unpublished data) indicating that the virus has a monopartite genome (RNA1) and that the other RNA species are satellites.
Protein: Dissociated virus particles yield polypeptides of M. Wt c. 29,000 (major band in polyacrylamide gels) and c. 26,000 (minor band). The significance of the minor band is unknown but it may be a degradation product of the major polypeptide similar to that found with viruses such as velvet tobacco mottle and solanum nodiflorum mottle (Chu & Francki, 1983).
Relations with Cells and Tissues
Virus particles, c. 22 nm in diameter, occur in cells of infected tissues that have been treated with ribonuclease to remove densely staining ribosomes (Hatta & Francki, 1981). They are distributed in nuclei, cytoplasm and cytoplasmic vacuoles of all types of cells. In the cytoplasm and nucleus they are always seen aggregated in distinct groups (Francki et al.,1983; Fig. 7). A similar distribution of virus particles is seen in cells of systemically infected subterranean clover plants and in local lesions in leaves of pea plants (Francki et al., 1985).
Notes
In the field, subterranean clover plants infected with the virus exhibit symptoms similar to those induced by the agent of subterranean clover stunt disease or by some isolates of bean yellow mosaic virus (Francki et al., 1983). However, subterranean clover mottle virus is readily distinguished from these by its particle morphology and serological specificity.
Figures

Stunting of manually inoculated Trifolium subterraneum plant (left) compared to uninfected plant (right).

Mottled and distorted leaf from an infected T. subterraneum plant (above) compared to one from an uninfected plant (below).
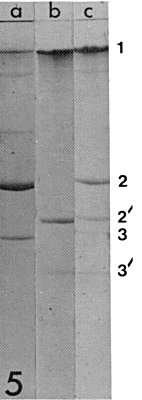
Separation of RNA1 (1) and the two types of RNA species 2 (2, 2') and 3 (3, 3') on a denaturing polyacrylamide gel. Tracks a and b are preparations from virus isolates carrying the 388-base or 332-base satellite RNA species respectively, and track c is a preparation from virus carrying both types of satellite RNA.
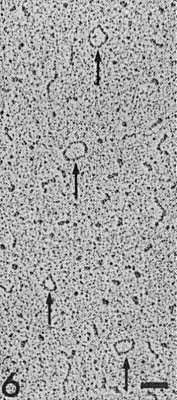
Rotary-shadowed RNA extracted from a preparation of subterranean clover mottle virus showing circular molecules (arrows) after spreading under denaturing conditions. Bar represents 100 nm.
References list for DPV: Subterranean clover mottle virus (329)
- Chu & Francki, Virology 129: 350, 1983.
- Francki, Randles, Hatta, Davies & Chu, Pl. Path. 32: 47, 1983.
- Francki, Randles, Chu, Rohozinski & Hatta, in Subviral Pathogens of Plants and Animals:Viroids and Prions, pp. 265-297, ed. K. Maramorosch & J.J. McKelvey, New York: Academic Press, 1985.
- Gould, Francki, Hatta & Hollings, Virology 108: 499, 1981.
- Graddon & Randles, J. Virol. Meth. 13: 63, 1986.
- Hatta & Francki, J. Ultrastruct. Res. 74: 1, 1981.
- Tremaine & Hamilton, CMI/AAB Descr. Pl. Viruses 274, 6 pp., 1983.