Details of DPV and References
DPV NO: 33 October 1970
Family: Closteroviridae
Genus: Closterovirus
Species: Citrus tristeza virus | Acronym: CTV
There is a more recent description of this virus: DPV 353
Citrus tristeza virus
W. C. Price Guinobatan Experiment Station, Guinobatan, Albay, Phillippines
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Transmitted by
Meneghini (1946) and
Fawcett & Wallace (1946),
and described
by Kitajima et al. (1964).
Synonym
- Citrus quick decline virus (Fawcett & Wallace, 1946)
-
A virus with highly flexuous filamentous particles, c. 2000 x 12 nm, restricted to the family Rutaceae. Causes a severe disease in only a few species. Common wherever citrus is grown. Transmitted by aphids, not by sap inoculation. Aphids retain virus up to 48 h when starved.
Main Diseases
Tristeza, or quick decline, of commercial varieties of citrus on sour orange root-stock; stem pitting, or stunt bush, of grapefruit; die-back of lime; seedling yellows of sour orange; and decline of Ellendale mandarin and Pera orange.
Geographical Distribution
World-wide in tropical and semi-tropical regions where citrus is grown.
Host Range and Symptomatology
Most varieties of Citrus and a few species in certain other genera of the family Rutaceae, such as Aeglopsis, Afraegle, and Pamburus, are susceptible to infection. A majority are, however, tolerant of commonly occurring strains of the virus. Tristeza is principally a disease of sweet orange or other varieties when grown on sour orange rootstock, and of grapefruit, lime, and calamondin.
-
Diagnostic species
- Citrus aurantifolia
(West Indian lime, Key lime, Mexican lime, Galego lime, Kagzi lime). Young leaves on infected seedlings are small, develop yellow flecks along their veins, and become cupped or canoe-shaped; stems are pitted under the bark (Wallace & Drake, 1951). - C. sinensis (sweet orange) plants on C. aurantium (sour orange) rootstock suddenly wilt, decline, and die (quick decline; Fig. 1), or develop a characteristic overgrowth at the bud-union and ‘honeycombing’ in the sour orange stock just below the union (Fig. 4).
-
Propagation species
- C. aurantifolia
is perhaps the best species for maintaining cultures because it displays diagnostic symptoms.
Strains
Many strains, differing in virulence, are found in nature. A few have been isolated experimentally (Grant & Higgins, 1957). The following are probably the most common naturally occurring strains of tristeza virus:
Citrus seedling yellows virus (Fraser, 1952). Causes severe yellowing and stunting of seedlings of Eureka lemon, Seville orange, grapefruit, and citron. (Some authors regard seedling yellows virus as being distinct from tristeza virus, but occurring with it.)
Grapefruit stem pitting virus (Oberholzer, Mathews & Stiemie, 1949); grapefruit stunt bush virus (Anon., 1950). In Australia, S. Africa and S. America, infected trees are stunted and bushy; leaves are sparse, small, rounded, and develop interveinal chlorosis; fruit is small, misshapen, and has thick rind; wood of trunk and large limbs is pitted with longitudinal depressions; main scaffold branches are twisted and distorted. This strain, or a similar one, is responsible for the decline of calamondin trees in the Philippines (Fig. 5).
Lime die-back virus (Hughes & Lister, 1949). Causes vein flecking on young leaves, severe pitting of wood of twigs and branches, stunting of trees, and die-back with eventual death.
Ellendale mandarin decline virus (Stubbs, 1952). This strain may be the same as, or similar to, the citrus seedling yellows strain.
Transmission by Vectors
Transmissible most efficiently by the tropical citrus aphid, Toxoptera citricidus (Meneghini, 1946; Costa & Grant, 1951); less efficiently by Aphis gossypii, A. spiraecola, and T. aurantii (Dickson, Flock & Johnson, 1951; Norman & Grant, 1956). Virus can be acquired in a few seconds and transmitted in a few seconds’ feeding. Latent period unknown. T. citricidus can retain the virus for as long as 24 h when not allowed to feed. It has been suggested (Price, 1968) that tristeza virus is a circulative, non-propagative virus. Recent work (Retuerma & Price, unpublished) suggests that it may be both stylet-borne and circulative.
Transmission through Seed
None reported.
Transmission by Dodder
Reported to be transmitted by Cuscuta americana (Knorr & Price, 1954), but not confirmed.
Serology
No antisera have been prepared.
Relationships
Several authors have found that infection of test plants with mild strains of tristeza virus protects them, at least in some cases, from more severe strains. Cross-protection tests have given evidence for relationship of seedling yellows, stem pitting, and Ellendale mandarin decline viruses to the tristeza virus complex (Stubbs, 1964).
Stability in Sap
No information.
Purification
A few flexuous filaments thought to be tristeza virus particles were found in preparations obtained by high-speed centrifugation of sap from leaves (Kitajima et al., 1964; Price, 1966). More recently Bar-Joseph, Loebenstein & Cohen (1970) found large numbers of such particles in preparations made from peeled bark of diseased West Indian lime seedlings. The method was as follows. Grind frozen tissue with mortar and pestle in liquid air. Thaw and extract twice with 0.05 M Tris/HCl buffer pH 7.4 (1 g/12.5 ml). Centrifuge 10 min at 4000 g. then 5 min at 8000 g. To every 100 ml of the supernatant add 4 g polyethylene glycol (M. Wt 6000) and 4 ml 20% NaCl. Centrifuge 15 min at 16,000 g. Suspend pellets in 0.04 M phosphate buffer pH 8.2. Clarify by centrifuging 10 min at 5000 g. Sediment at 29,000 rpm in Spinco No. 30 rotor for 90 min. Suspend in water and centrifuge 10 min at 5000 g.
Properties of Particles
Sedimentation coefficient (s20,w) determined in sucrose density gradients: 140 ± 10 S at zero depth (Bar-Joseph et al., 1970).
Particle Structure
Particles are very flexuous filaments c. 2µm long and 10-11 nm in diameter (Fig. 3). Visible in leaf dip preparations, or in large numbers in bark dip preparations, mounted in phosphotungstate.
Particle Composition
No information.
Relations with Cells and Tissues
Flexuous filamentous particles occur in large numbers in phloem cells (Fig. 2) but not in other cells. Destruction of phloem tissues in sour orange rootstock is responsible for quick decline of orchard trees.
Figures

Sweet orange on sour orange rootstock in quick decline phase ot tristeza. (Photo courtesy J. M. Wallace.)
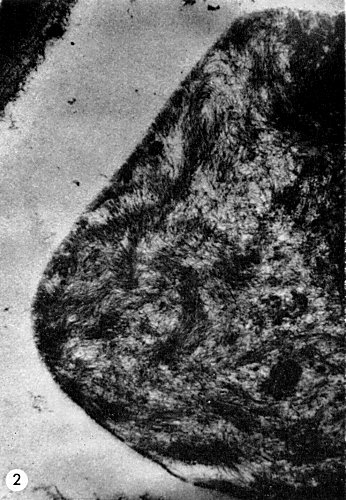
Electron micrograph of filamentous particles in a portion of a phloem cell of a West Indian lime seedling infected with tristeza virus.

Two filamentous particles, c. 2000 x 12 nm in an electron micrograph of a dip preparation from a West Indian lime leaf infected with tristeza virus.
References list for DPV: Citrus tristeza virus (33)
- Anon., Agr. Gaz. N.S.W. 61: 365, 1950.
- Bar-Joseph, Loebenstein & Cohen, Phytopathology 60: 75, 1970.
- Costa & Grant, Phytopathology 41: 105, 1951.
- Dickson, Flock & Johnson, Calif. Citrogr. 36: 135, 1951.
- Fawcett & Wallace, Calif. Citrogr. 32: 50, 1946.
- Fraser, Agr. Gaz. N.S.W. 63: 125, 1952.
- Grant & Higgins, Phytopathology 47: 272, 1957.
- Hughes & Lister, Nature, Lond. 164: 880, 1949.
- Kitajima, Silva, Oliveira, Muller & Costa, Nature, Lond. 201: 1011, 1964.
- Knorr & Price, Rep. Fla agric. Exp. Stn 195, 1954.
- Meneghini, Biologico 12: 285, 1946.
- Norman & Grant, Proc. Fla St. hort. Soc. 69: 38, 1956.
- Oberholzer, Mathews & Stiemie, Sci. Bull. Dep. Agric. S. Afr. 287, 17 pp., 1949.
- Price, Virology 29: 285, 1966.
- Price, Indian Phytopath. 21: 159, 1968.
- Stubbs, J. Dep. Agric. Vict. 50: 124, 1952.
- Stubbs, Aust. J. agric. Res. 15: 752, 1964.
- Wallace & Drake, Calif Citrogr. 36: 136, 1951.

