Details of DPV and References
DPV NO: 332 September 1988
Family: Partitiviridae
Genus: Betacryptovirus
Species: White clover cryptic virus 2 | Acronym: WCCV-2
White clover cryptic virus 2
E. Luisoni Istituto di Fitovirologia applicata del C.N.R., Via O. Vigliani 104, I-10135 Torino, Italy
R. G. Milne Istituto di Fitovirologia applicata del C.N.R., Via O. Vigliani 104, I-10135 Torino, Italy
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Boccardo et al. (1985).
Synonym
- None, but the name white clover temperate virus was used to indicate a mixture of white clover cryptic viruses 1, 2 and 3 (Natsuaki et al., 1984, 1986).
-
A virus with isometric particles about 38 nm in diameter, containing two size classes of double-stranded (ds) RNA. Occurs symptomlessly and in low concentration in white clover. Not transmissible by sap inoculation or by grafting but transmitted to a high degree through the seed.
Main Diseases
Causes no symptoms in white clover (Trifolium repens), the only host known (Boccardo et al., 1983, 1985).
Geographical Distribution
Found in white clover grown from seed from Europe, Japan, New Zealand and the USA. Probably very widespread, but in a seed collection held at the Waite Agricultural Research Institute, South Australia, only one carrier plant was detected among 173 seedlings from five cultivars. However, this one seed may have come by accident from other seed lines being handled in our laboratory at the time (Boccardo et al., 1985; Natsuaki et al., 1986).
Host Range and Symptomatology
The virus was found in about half the white clover cultivars tested (Boccardo et al., 1985; Natsuaki et al., 1986). The experimental host range cannot be determined because the virus is not transmissible except through seed.
-
Diagnostic species
- No diagnostic species known.
-
Assay species
- No assay species known.
-
Propagation species
- Any carrier white clover plant, vegetatively propagated, provided that no other viruses, in particular other white clover cryptic viruses, are present. The presence of the virus in vegetatively propagated carrier clones must be confirmed periodically because virus-free stolons sometimes arise (see Notes).
Strains
All samples so far isolated show no serological differences in double antibody sandwich (DAS) ELISA with antiserum to one isolate.
Transmission by Vectors
Not tested.
Transmission through Seed
Boccardo et al. (1985) detected the virus in 15-41% of the tested seedlings of six white clover cultivars (carrier or non-carrier condition of parents not known). Of plants obtained from seeds produced by carrier or non-carrier mother plants pollinated by natural means in the open, about 65% and 4%, respectively, were found to be carriers (V. Lisa & E. Luisoni, unpublished information).
Transmission by Grafting
Not transmitted from white clover to white clover by grafting carrier scions to non-carrier stocks, even when the grafts took well and survived up to 4 months. Moreover, no transmission resulted when carrier scions were grafted to non-carrier stocks obtained from virus-free stolons of plants that had originally been carriers (see Notes); failure of graft transmission is thus unlikely to have been due to any genetic resistance of the grafted stock plant. When alfalfa mosaic virus was present, along with white clover cryptic virus 2, in the carrier scions, it was transmitted to about 50% of grafted stock plants, whereas white clover cryptic virus 2 was not. Nor was the latter virus graft-transmitted in the presence of white clover cryptic virus 1 (Boccardo et al., 1985; Boccardo et al., 1987; V. Lisa, G. Dellavalle & E. Luisoni, unpublished information).
Transmission by Dodder
No transmission obtained (V. Lisa & E. Luisoni, unpublished information).
Serology
The virus is a good immunogen and antigen. Antisera with titres up to 1/2048 in agar gel double diffusion tests have been obtained in rabbits by intramuscular injection of about 0.7 mg purified virus (E. Luisoni, unpublished information). Owing to the low virus content in carrier plants, concentrated sap is required to obtain reliable results with this test. DAS-ELISA and immunoelectron microscopy (immunosorbent electron microscopy (ISEM) plus decoration) easily detected the virus in crude plant sap (Boccardo et al., 1985; Natsuaki et al., 1986).
Nucleic Acid Hybridization
Complementary DNA (cDNA) probes have been obtained that can detect the homologous RNA species of white clover cryptic virus 2 in hybridization experiments on nitrocellulose membranes, but not the RNA species of white clover cryptic viruses 1 and 3. Similarly, cDNA probes made to the RNA species of white clover cryptic viruses 1 and 3 did not hybridize detectably to the RNA species of white clover cryptic virus 2 (G. Boccardo & P. Palukaitis, unpublished information).
Relationships
No serological relationship was found to members of sub-group A of the proposed cryptovirus group (see Notes). Among the viruses of sub-group B, white clover cryptic virus 2 is serologically related to a morphologically similar cryptic virus in red clover (Fig. 3), and more distantly related to a second similar virus in hop trefoil (Fig. 4) (Boccardo et al., 1987; Luisoni et al., 1987; E. Luisoni & R. G. Milne, unpublished information). White clover cryptic virus 2 shares some properties with mycoviruses (Partitiviridae), especially those of sub-group D (Buck et al., 1984), but it did not react with any of 10 antisera to 15 mycoviruses, including examples from sub-group D (Boccardo et al., 1985).
Stability in Sap
Thermal inactivation, longevity in vitro and dilution end-point have not been determined because infectivity tests are not feasible. The virus can react in agar double diffusion tests giving a sharp precipitin band, and appears morphologically intact by electron microscopy, after over 1 year of storage at 4°C (E. Luisoni & R. G. Milne, unpublished information).
Purification
(Boccardo et al., 1985). Perform all operations near 4°C. Homogenize 1 kg leaf and stem tissue of carrier plants with 2-3 litres 0.05 M phosphate buffer, pH 7, containing 20 mM Na2SO3, 10 mM DIECA and 5 mM EDTA. Good results are obtained by using a high-speed centrifugal grinding mill and mixing frozen and crushed buffer with the plant material. Emulsify the slurry with an equal volume of CHCl3 and, after separation of the emulsion, add polyethylene glycol (M. Wt 6000) to 10% (w/v) and NaCl to 1% (w/v), and dissolve in the aqueous phase. After stirring for 2.5 h, centrifuge the mixture at low speed and resuspend the pellets in 0.5 M phosphate buffer, pH 7, containing 20 mM Na2SO3. Clarify the preparation again with CHCl3 as above, and recover the virus particles from the aqueous phase by ultracentrifugation. Resuspend the pellets in 0.05 M phosphate buffer, pH 7; layer the suspension, clarified by low-speed centrifugation, on to linear 20-45% (w/v) Cs2SO4 gradients, prepared in the same buffer just before use, and ultracentrifuge for 90 min. Collect the light-scattering band (Fig. 1) and free it from Cs2SO4 by overnight dialysis or by ultracentrifugation. Yields are about 250 µg per kg tissue. For isopycnic centrifugation, suspend the purified virus in CsCl solution (made up in 0.01 M phosphate buffer, pH 7) of initial density about 1.38 g/ml, and ultracentrifuge for up to 70 h.
Properties of Particles
After isopycnic centrifugation in CsCl, the virus forms one band with a buoyant density of 1.375 g/ml.
A260/A280: 1.31; Amax/Amin: 1.08 (both values uncorrected for light-scattering) (Boccardo et al., 1985).
Particle Structure
The particles (Fig. 2), when negatively stained in uranyl acetate, appear about 38 nm in diameter, rounded in profile, and with prominent morphological subunits; sometimes they exhibit a ‘core’, probably an artifact of staining.
Particle Composition
Nucleic acid: Two size classes of dsRNA, of M. Wt 1.49 x 106 and 1.38 x 106, as estimated in 5% polyacrylamide gels buffered in tris-borate-EDTA (Fig. 6). The particles contain about 24% RNA by weight, calculated from the particle buoyant density (method of Sehgal et al., 1970). In the electron microscope both dsRNA species appear linear under non-denaturing conditions, with modal lengths of 689 nm and 653 nm. The RNA has a buoyant density in Cs2SO4 solutions of 1.605 g/ml. RNA preparations react with a serum containing antibodies to dsRNA (Fig. 5) (Boccardo et al., 1985).
The RNA molecules have been translated in vitro and appear to act as monocistronic genes, one of which codes for the coat protein (D. D. Dunigan & G. Boccardo, unpublished information; and see below).
Protein: No information available, because of difficulty in solubilizing the coat protein (Boccardo et al., 1987). A product that could be precipitated with antiserum to the virus was made in rabbit reticulocyte and wheat germ in vitro translation systems primed with viral RNA (D. D. Dunigan & G. Boccardo, unpublished information).
Relations with Cells and Tissues
After clearing ribosomes from tissues by treatment with RNase, Boccardo et al. (1985) found small numbers of virus-like particles in the cytoplasm of a few of the parenchyma cell profiles examined from plants carrying white clover cryptic viruses 1 and 2 (Fig. 7) but whether any of the particles were those of white clover cryptic virus 2 is uncertain. No other pathological changes were detected in cells of carrier plants. The virus was readily detected by ISEM in ovaries, stamens, petals, leaves, roots, epidermal strips from stolons, and stolon segments with epidermis removed (Boccardo et al., 1985).
Notes
Boccardo et al. (1987) suggested that the as yet unofficial ‘cryptovirus group’ could be divided into sub-groups A and B, based on differences in particle morphology. Sub-group A would contain viruses similar to carnation cryptic virus (Lisa et al., 1986). White clover cryptic virus 2 could constitute the type virus for sub-group B. Particles of sub-group B morphology have been noted in alfalfa (Medicago sativa), hop trefoil (M. lupulina) and red clover (Trifolium pratense) (see Relationships).
Two other cryptic viruses, white clover cryptic viruses 1 and 3, also occur in white clovers but these have sub-group A morphology (Boccardo et al., 1985; Natsuaki et al., 1986). There is no correlation between their presence and that of white clover cryptic virus 2, in individual plants. Analysis of dsRNA from whole plants has shown that two further pairs of dsRNA species can occur, with M. Wt similar to those of cryptic viruses. Thus the presence of two further cryptic viruses of white clover is suspected (G. P. Accotto, unpublished information).
When plants carrying the virus are propagated vegetatively, stolons occasionally arise that are virus-free, judged by ELISA, ISEM (Boccardo et al., 1987) and dsRNA analysis (G. P. Accotto, unpublished information). Some stolons were found that produced virus-containing leaves on one side and virus- free leaves on the other. This behaviour is similar to genetic sectoring, and probably results from an inability of the virus to pass from cell to cell, although it can multiply within cells. During many cell divisions, occasional virus-free meristems would arise, that could not be reinfected. The inability to pass from cell to cell has not been confirmed, but is predicted from the observed lack of graft-transmissibility (Boccardo et al., 1987).
Figures
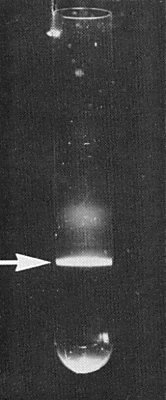
Light-scattering band (arrow) formed by the virus after 90 min centrifugation at 35,000 rev./min in a 20-45% preformed caesium sulphate density gradient.

Particles of the virus negatively stained in uranyl acetate; one particle shows a ‘core’ (large arrow). Small arrows indicate two particles of tomato bushy stunt virus added as an internal size standard (diameter 34 nm). Bar represents 100 nm.
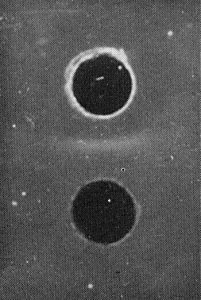
Reaction of the virus (upper well) with an antiserum to a mixture of cryptic viruses from red clover.

Reaction of a sub-group B virus from hop trefoil (upper well) with an antiserum to white clover cryptic virus 2.
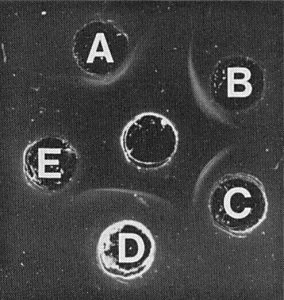
Reactions of a maize rough dwarf virus antiserum containing antibodies to dsRNA (Luisoni et al., 1975) (central well) with RNA preparations from rice gall dwarf virus (A), white clover cryptic viruses 1 (B), 2 (C) and 3 (D). Well (E) contained single-stranded RNA from tobacco mosaic virus, which gave no reaction.

Electrophoretic separation in a 5% polyacrylamide slab gel of dsRNA from white clover cryptic viruses 1 (A), 1 and 2 (B), 2 alone (C), maize rough dwarf virus (D) and cytoplasmic polyhedrosis virus (E) (gift of C. C. Payne; photo courtesy of G. Boccardo). Stained with silver. In addition to the two more rapidly migrating bands of white clover cryptic virus 2 RNA, lane C also contains two slowly migrating bands, sometimes present in extracts of virus particle preparations still contaminated with host materials (Boccardo et al., 1987).
References list for DPV: White clover cryptic virus 2 (332)
- Boccardo, Lisa & Milne, in Double-Stranded RNA Viruses, p. 425, eds R. W. Compans & D. H. L. Bishop, Elsevier, New York, 1983.
- Boccardo, Milne, Luisoni, Lisa & Accotto, Virology 147: 29, 1985.
- Boccardo, Lisa, Luisoni & Milne, Adv. Virus Res. 32: 171, 1987.
- Buck, Ackermann, Bozarth, Bruenn, Koltin, Rawlinson, Ushiyama & Wood, Intervirology 22: 17, 1984.
- Lisa, Luisoni & Milne, AAB Descr. Pl. Viruses 315, 4 pp., 1986.
- Luisoni, Milne & Boccardo, Virology 68: 86, 1975.
- Luisoni, Milne, Accotto & Boccardo, Intervirology 28: 144, 1987.
- Natsuaki, Natsuaki, Okuda, Teranaka, Yamashita & Doi, J. agric. Sci., Tokyo Nogyo Daigaku 29: 49, 1984.
- Natsuaki, Natsuaki, Okuda, Teranaka, Milne, Boccardo & Luisoni, Intervirology 25: 69, 1986.
- Sehgal, Jean, Bhalla, Soong & Krause, Phytopathology 60: 1778, 1970.
