Details of DPV and References
DPV NO: 334 September 1988
Family: Bromoviridae
Genus: Bromovirus
Species: Cassia yellow blotch virus | Acronym: CYBV
Cassia yellow blotch virus
J. L. Dale Agricultural Research Laboratories, Department of Primary Industries, Indooroopilly, Queensland 4068, Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by Dale et al. (1984).
A virus with isometric particles c. 26 nm in diameter with four species of single-stranded RNA, the three largest of which are required for infectivity. It is mechanically transmissible by inoculation with sap but has a narrow experimental host range. Reported only from one western district of Queensland, Australia.
Main Diseases
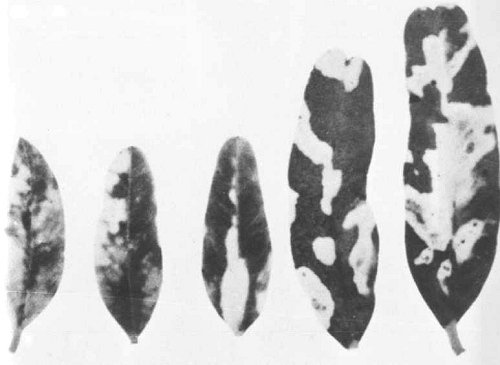
Cassia pleurocarpa (Leguminosae) is the only recorded natural host. The virus causes a vivid yellow blotch symptom on the leaves (Fig. 1) (Dale et al., 1984).
Geographical Distribution
Reported from only one western district of Queensland, Australia (Dale et al., 1984).
Host Range and Symptomatology
In Cassia pleurocarpa, the only known natural host, the virus causes vivid yellow blotches on the leaflets (Fig. 1). The experimental host range (determined by inoculation with sap) is narrow: the virus systemically infected only nine species from two families of the 44 species from seven families tested (Dale et al., 1984).
-
Diagnostic species
- Cassia occidentalis.
Systemic chlorotic mottle develops 14-21 days after inoculation. - Chenopodium amaranticolor. Fine discrete chlorotic local lesions develop 4-5 days after
inoculation (Fig. 3). Does not become systemically infected.
- Nicotiana clevelandii. Systemic green mottle develops 10-16 days after inoculation (Fig. 2). This is the only non-leguminous species that is reported to be systemically infected.
-
Propagation species
- Nicotiana clevelandii
is suitable as a source of virus for purification. Cassia occidentalis is suitable for maintaining cultures.Assay species
- Chenopodium amaranticolor
is a suitable local lesion host.
Strains
No strains reported.
Transmission by Vectors
No information.
Transmission through Seed
No information.
Serology
The virus particles are very poorly immunogenic. In attempts to produce antisera by injecting rabbits with either untreated or formaldehyde-treated virus particles, the best antiserum obtained had a titre of only 1/8. In agarose gel double diffusion tests, this antiserum reacted with formaldehyde-treated homologous virus particles to produce a single line of precipitation (Dale et al., 1984).
Relationships
In its particle morphology and composition, cassia yellow blotch virus is more closely related to the bromoviruses (Lane, 1981) than to viruses of any other group. However, no serological relationship has been found to brome mosaic, broad bean mottle or cowpea chlorotic mottle viruses. In the amino acid composition of its coat protein, cassia yellow blotch virus is more closely related to other bromoviruses than to cucumoviruses or alfalfa mosaic virus but less closely related to other bromoviruses than they are to each other (Dale et al., 1984).
Stability in Sap
Not known.
Purification
This virus can be purified by a method adapted from that of Bancroft et al. (1967). Extract sap from each 100 g N. clevelandii leaf, harvested 10 to 14 days after inoculation, in 200 ml 200 mM sodium acetate buffer, pH 4.8, containing 1 mM EDTA. Express the homogenate through cheesecloth, incubate at 4°C for 4 h and then centrifuge at 8000 g for 15 min. Retain the supernatant fluid and add polyethylene glycol (M. Wt 6000) and sodium chloride to 10% and 1% (w/v) respectively. Incubate at 4°C for 1 h and then centrifuge at 10,000 g for 15 min. Resuspend the pellets in 100 mM sodium acetate buffer, pH 5.0, containing 1 mM EDTA and centrifuge at 10,000 g for 15 min. Further purify the virus from the resulting supernatant fluid by two cycles of differential centrifugation (164,000 g for 90 min and 10,000 g for 10 min), resuspending each time in 100 mM sodium acetate buffer, pH 5.0. This procedure results in yields of up to 400 mg virus/kg infected leaf tissue.
Properties of Particles
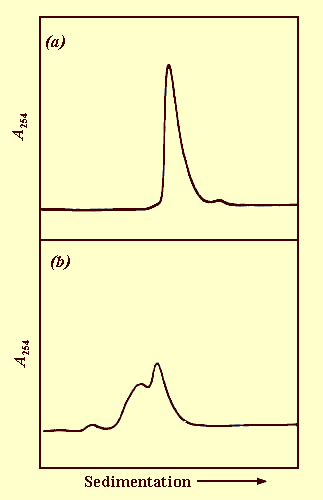
At pH 5.0, the virus particles sediment as a single component whereas at pH 7.0 they sediment as three components, one slightly slower than the single component at pH 5.0 and two considerably more slowly (Fig. 5). This probably results from the swelling of the particles at pH 7.0.
Sedimentation coefficient at pH 5.0 (s°20, w): 85.1 S.
Isoelectric point: pH 3.6.
Particle Structure
The particles are rounded to slightly angular and have a diameter of 25-27 nm (Fig. 4). In uranyl acetate or sodium phosphotungstate, the particles may collapse on the grid to produce a central cavity that contains a pool of stain.
Particle Composition
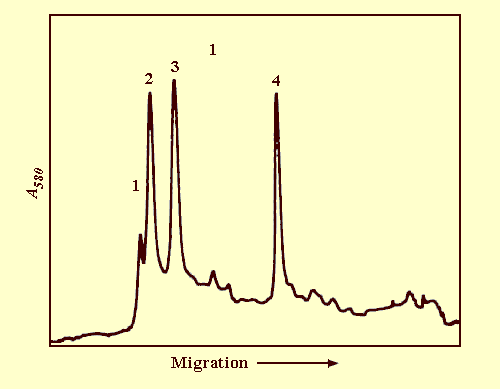
Nucleic acid: RNA, single-stranded. Under non-denaturing conditions (Peacock & Dingman, 1968), the RNA migrated as four major bands (Fig. 6) with apparent M. Wt (x 10-6) of 1.10 (RNA-1), 0.96 (RNA-2), 0.78 (RNA-3) and 0.31 (RNA-4). Only the three largest RNA molecules are required for infectivity. The percentage base composition of the RNA was guanine 24.4, adenine 24.9, cytosine 22.2 and uracil 28.5 (Dale et al., 1984).
Protein: A single protein species which under denaturing conditions (Laemmli, 1970) has an apparent M. Wt of 20,800 ± 1%. Amino acid composition indicates that the coat protein contains 196 amino acid residues (Dale et al., 1984).
Relations with Cells and Tissues
No information.
Notes
The virus is difficult to confuse with others. It has been found only in western Queensland in an Australian native legume, Cassia pleurocarpa. No other viruses have been reported to infect this species, but bean yellow mosaic virus has been recorded in a number of other Cassia spp. in Australia.
Figures

Particles in a purified preparation negatively stained with uranyl acetate. Bar marker represents 100 nm.
References list for DPV: Cassia yellow blotch virus (334)
- Bancroft, Hills & Markham, Virology 32: 354, 1967.
- Dale, Gibbs & Behncken, J. gen. Virol. 65: 281, 1984.
- Laemmli, Nature, Lond. 277: 680, 1970.
- Lane, in Handbook of Plant Virus Infections and Comparative Diagnosis, p. 333, ed. E. Kurstak, 943 pp., New York: Elsevier/North Holland, 1981.
- Peacock & Dingman, Biochemistry 7: 668, 1968.