Details of DPV and References
DPV NO: 343 December 1989
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Lettuce necrotic yellows virus | Acronym: LNYV
This is a revised version of DPV 26
Lettuce necrotic yellows virus
R. I. B. Francki Department ot Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064
J. W. Randles Department ot Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064
R. G. Dietzgen Department ot Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia, 5064
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Stubbs & Grogan (1963).
A virus with enveloped, RNA-containing, bacilliform particles about 227 x 68 nm. It has a narrow host range, is transmitted by inoculation of sap and by two species of aphid in the circulative, propagative manner. Reported from Australia and New Zealand.
Main Diseases
Causes a serious disease of lettuce in Australia and New Zealand (Fig. 1, Fig. 2) similar to those caused by tomato spotted wilt virus or by virulent isolates of lettuce mosaic virus. Sonchus oleraceus, the natural reservoir of both virus and vector, is infected symptomlessly (Stubbs & Grogan, 1963).
Geographical Distribution
Widespread in Australia and also reported in New Zealand (Fry et al., 1973). Its distribution appears to coincide with that of S. oleraceus though S. hydrophilus, S. kirkii, Reichardia tingitana and Embergeria megalocarpa are also natural hosts of both virus and vector (Randles & Carver, 1971; R. Beever & J. W. Randles, unpublished results) and are potentially important sources of the virus.
Host Range and Symptomatology
Narrow host range in the Chenopodiaceae, Compositae, Leguminosae, Liliaceae and
Solanaceae
(Stubbs & Grogan, 1963;
Behncken, 1983;
J. W. Randles & R. I. B.
Francki, unpublished results). Transmissible by sap inoculation with difficulty to
lettuce (Lactuca sativa) and sowthistle (Sonchus oleraceus), but easily
to Nicotiana glutinosa and N. clevelandii.
Diagnostic species
Faint interveinal chlorosis in systemically infected leaves
(Fig. 5).
Nicotiana glutinosa. Mild isolates induce slight stunting of the plant with downward curling and a faint mosaic in the systemically infected leaves (Fig. 3). Virulent isolates induce necrotic local lesions 6-8 days after mechanical inoculation; systemic symptoms are vein yellowing followed by chlorosis, leaf curling and cessation of growth in terminal leaves (Fig. 4).
Propagation species
Assay species
Strains
Variants isolated from the field have been distinguished by the severity of symptoms induced in Nicotiana glutinosa. The severe SE3 isolate (Fig. 4) (Stubbs & Grogan, 1963) has been used for most studies on the properties of the virus. No serological differences have been reported between strains.
Transmission by Vectors
Transmissible by the aphids Hyperomyzus lactucae (Stubbs & Grogan, 1963) and H. carduellinus (Randles & Carver, 1971). The virus is circulative and propagative in H. lactucae. Following acquisition feeds of 24 h, a temperature- dependent latent period is required of 18, 9 or 5 days at 15, 20 and 28°C, respectively. Not all aphids in a colony transmit the virus. The inoculation access threshold is 5-30 min on S. oleraceus and 1-5 min on lettuce. The virus is transmitted transovarially. The longevity of transmitting aphids is shorter than that of non-transmitting individuals (Boakye & Randles, 1974).
Epidemiological studies indicate that H. lactucae is the most important vector of the virus in lettuce crops in Australia (Randles & Crowley, 1970; Martin & Randles, 1981; Randles, 1983). S. oleraceus is the main field source of both the virus and the vector, H. lactucae. Lettuce, which is not colonised by the aphid, becomes infected when probed by migrating aphids. Disease outbreaks coincide with flights of H. lactucae and the distribution of infected lettuce plants also indicates that vectors bring the virus from outside the crop.
Transmission through Seed
No evidence of transmission through seed of lettuce varieties, Sonchus oleraceus or Nicotiana glutinosa (Stubbs & Grogan, 1963; R. I. B. Francki, unpublished results).
Serology
Polyclonal antiserum prepared in a rabbit by intramuscular and subcutaneous injections had titres of 1/256 and 1/64 in tube precipitin and gel-diffusion tests, respectively (McLean et al., 1971). Tube precipitin tests have been used (Harrison & Crowley, 1965) but can be complicated by formation of non-specific precipitates. Detergent-disrupted virus produced several precipitin lines in gel diffusion tests (McLean et al., 1971) and the antibodies recognised at least the three major viral structural proteins in immunoblots (Dietzgen & Francki, 1988). Plant extracts need to be concentrated to allow detection of the virus in gel diffusion tests (Randles & Carver, 1971). Double antibody sandwich enzyme-linked immunosorbent essay (DAS-ELISA) is at least 8000 and 600 times more sensitive than gel-diffusion and infectivity assays, respectively, for detecting virus in plant extracts (Chu & Francki, 1982). DAS-ELISA can also be used for detecting virus in individual aphids (Chu & Francki, 1982).
Monoclonal antibodies to the viral G and N proteins have been used in immunoblots and indirect ELISA to analyse the fine structure of the virus (Dietzgen & Francki, 1988).
Relationships
The particle morphology and composition place lettuce necrotic yellows virus in the family Rhabdoviridae (Matthews, 1982). In their possession of a single M protein, and of in vitro transcriptase activity, particles of the virus resemble those of members of the genus vesiculovirus infecting vertebrates (Matthews, 1982). By virtue of its particle composition and cytoplasmic site of replication, lettuce necrotic yellows virus is similar to broccoli necrotic yellows virus (Campbell & Lin, 1972), sonchus virus (Vega et al., 1976), American wheat striate mosaic virus (Sinha & Behki, 1972), barley yellow striate mosaic virus (Milne & Conti, 1986) and northern cereal mosaic virus (Toriyama, 1986) all of which are grouped in plant rhabdovirus sub-group 1 (Peters, 1981). No serological relationships of these viruses to lettuce necrotic yellows virus have been reported.
Stability in Sap
In Nicotiana glutinosa sap, the thermal inactivation point (10 min) is about 52°C, dilution end-point about 10-2, and infectivity is retained for 8-12 h (Stubbs & Grogan, 1963). Infectivity is stabilized by 0.1% bovine serum albumin but not by reducing agents (Crowley, 1967). Inactivation of virus is accelerated by some chelating agents such as diethyl dithiocarbamate (Atchison et al., 1969). The virus is inactivated by organic solvents such as butan-l-ol, chloroform and ether (Crowley et al., 1965; Harrison & Crowley, 1965). Maximum infectivity is obtained with inocula at pH 6 (R. I. B. Francki, unpublished results).
Purification
A simpler method than that used by McLean & Francki (1967) is as follows. Extract 50 g lots of leaf material, from experimentally-infected N. glutinosa or field-infected lettuce showing prominent symptoms, in 50 ml 0.2 M Na2HPO4. Strain the extract through cheesecloth and re-extract the fibre with another 25 ml 0.2 M Na2HPO4. Clarify the pooled extracts by centrifugation at 3000 g for 5 min and filter through a 10 mm pad of Celite in a Buchner flask, and repeat the filtration if the extract remains green. Concentrate the virus by centrifugation at 44,000 g for 20 min, resuspend the pellets in water and apply to a calcium phosphate gel column equilibrated with 10 mM phosphate buffer, pH 7.6 (McLean & Francki, 1967). Elute with the same buffer and collect opalescent fractions which contain the virus (calcium phosphate gel preparations vary widely in their properties and suitable batches must be selected by trial). Concentrate the virus by centrifugation at 44,000 g for 20 min and resuspend in 10 mM phosphate buffer, pH 7.6. At this stage the virus should be highly purified but to further purify (or check for its purity), centrifuge in 10-40% sucrose gradients at 37,000 rev/min for 30 min in a Beckman SW 41 rotor. Recover the single band of virus, dilute with 10 mM phosphate buffer, pH 7.6, and concentrate by centrifugation at 44,000 g for 30 min.
Properties of Particles
Sedimentation coefficient (s20,w): approximately 940 S.
Buoyant density in sucrose solution: 1.19-1.20 g/cm3 (Harrison & Crowley, 1965).
Particle Structure
Particles are bacilliform, about 227 nm long and 68 nm wide in negatively stained preparations (Harrison & Crowley, 1965) but 360 x 52 nm in tissue sections (Fig. 10, Fig. 11; Chambers et al., 1965). Bullet-shaped or pleomorphic particles (Fig. 7) seen in preparations negatively stained and dried for electron microscopy are artefacts (Wolanski et al., 1967; Wolanski & Francki, 1969; Francki & Randles, 1975).
The virus particle (Fig. 6, Fig. 8) consists of an outer lipid membrane with projections arranged in a hexagonal lattice enclosing a nucleocapsid of 260 S (Francki & Randles, 1975) coiled in a regular helix with 45 nm pitch (Wolanski et al., 1967). Dissociation of the viral envelope with a non-ionic detergent such as Nonidet P40 or Triton X-100 releases an infective nucleocapsid (Randles & Francki, 1972) whereas an ionic detergent such as sodium dodecyl sulphate dissociates both the envelope and nucleocapsid to release a non-infective RNA (Francki & Randles, 1973).
Particle Composition
Nucleic acid: RNA, single-stranded, negative sense, with M. Wt c. 4 x 106, as estimated from the sedimentation coefficient (s20,w) of c. 44 S (Francki & Randles, 1973), and c. 13,000 nucleotides, estimated from the electrophoretic mobility in agarose (Dietzgen et al., 1989).
Protein: Three major polypeptides of M. Wt c. 78 (G protein), c. 57 (N protein) and c. 19 (M protein) kilodaltons are resolved by polyacrylamide gel electrophoresis of dissociated virus particles (Fig. 9: Dale & Peters, 1981; Dietzgen & Francki, 1988). G is the envelope-associated glycoprotein, N is a protein associated with the RNA and M is the hydrophobic matrix protein (Francki & Randles, 1980; Dietzgen & Francki, 1988). In addition several minor bands have been observed (Fig. 9). Two of these, of 160-180 kilodaltons (L protein, Fig. 9) and c. 38 kilodaltons (NS protein, Fig. 9) respectively, may be associated with transcriptase activity as established for vesicular stomatitis virus (Banerjee, 1987). The others appear to be either multimers or degradation products of the G and N proteins (Dietzgen & Francki, 1988).
Carbohydrate: The G protein is glycosylated with a complex network of oligosaccharides containing N-acetylchitobiose, N-linked to asparagine residues. The carbohydrate binds to concanavalin A and can be removed by endoglycosidase F (Dietzgen & Francki, 1988).
Lipid: Contained in the lipoprotein envelope; essential for maintaining the integrity of the virus particle. No information on chemical composition or amount.
Enzyme activity: Purified virus preparations contain a RNA-dependent RNA polymerase (transcriptase). The enzyme is activated on dissociation of the viral envelope and is associated with the nucleocapsid (Francki & Randles, 1972, 1973; Randles & Francki, 1972; Toriyama & Peters, 1980).
Genome Properties
During replication of the virus, five distinct polyadenylated RNA species, of 6450, 2000, 1650, 1200 and 900 nucleotides, complementary to the viral genomic RNA (cRNA 1 to 5) are detected in infected N. glutinosa plants. From their size range and sequence complexity they are assumed to represent messenger (m)RNA molecules encoding the five viral structural proteins. The cRNA3 was identified as N protein mRNA (Dietzgen et al., 1989).
Relations with Cells and Tissues
Particles are seen in the cytoplasm of mesophyll, epidermal and hair cells as well as in immature xylem cells and in sieve tubes of infected plants (Chambers et al., 1965; Chambers & Francki, 1966; Francki et al., 1985). Cellular changes associated with infection appear first in the nucleus. At about the time of first symptom appearance in N. glutinosa, the outer nuclear membrane of infected cells develops ‘blisters’ containing small vesicles. This is accompanied by virus-specific RNA synthesis as measured by 3H-uridine incorporation (Wolanski & Chambers, 1971). Later, the virus-specific RNA synthesis is evident in the cytoplasm rather than the nuclei. During this time virus particles (Fig. 10, Fig. 11) are seen budding from vesicles of the endoplasmic reticulum (Fig. 10) and viroplasms consisting of threadlike structures can be seen (Wolanski & Chambers, 1971; Francki et al., 1985). During later stages of infection, nuclei, mitochondria and especially chloroplasts, show signs of degradation and there is a rapid loss of 70 S chloroplast ribosomes (Randles & Coleman, 1970). Reduced protein synthesis in leaves accompanies symptom development, together with a general increase in the content of soluble amino acids, amines and peptides (Randles & Coleman, 1972).
Particles without the outer coat are seen in the muscles, fat body, brain, mycetome, tracheae, epidermis, salivary glands, and cells of the alimentary canal of viruliferous Hyperomyzus lactucae (O’Loughlin & Chambers, 1967).
Notes
In lettuce, similar diseases can be caused by lettuce necrotic yellows, tomato spotted wilt and lettuce mosaic viruses. These viruses can be distinguished by serology and electron microscopy (Francki et al., 1981; Francki & Hatta, 1981; Chu & Francki, 1982). Tomato spotted wilt virus can also be identified by its characteristic symptoms on Nicotiana spp., Petunia hybrida, Cucumis sativus and Lycopersicon esculentum (Ie, 1970; Francki & Hatta, 1981), and lettuce mosaic virus by the reactions on Chenopodium amaranticolor, C. quinoa and Pisum sativum (Tomlinson, 1970).
Four other rhabdoviruses are known to infect lettuce and Sonchus oleraceus but are much less virulent in lettuce. Among these, lettuce necrotic yellows virus differs from sonchus yellow net virus (Jackson & Christie, 1979) in host range, aphid vectors, serology, cytopathology and protein composition; from sowthistle yellow vein virus (Peters, 1971; Jackson et al., 1987) in host range, serology, mechanical transmission, cytopathology and protein composition, and from gomphrena virus (Kitajima & Costa, 1966) in host range and cytopathology. However, the relationship between sonchus virus, described from Argentina (Vega et al., 1976), and lettuce necrotic yellows virus is not clear.
Figures

Infected lettuce (Lactuca sativa) plants in the field (two severely diseased plants are seen in the foreground).
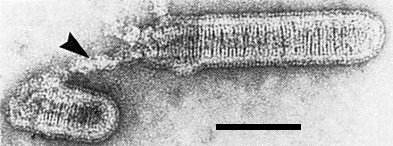
Broken virus particle stained with uranyl acetate with uncoiling nucleocapsid (arrow). Bar represents 100 nm.

Virus particles in preparation stained with phosphotungstic acid, pH 6.8, showing many bullet-shaped particles and a broken particle with extruded thread-like nucleocapsid (arrow). Bar represents 500 nm.

Model of virus particle showing the location of the viral RNA, the major structural proteins and lipid.
References list for DPV: Lettuce necrotic yellows virus (343)
- Atchison, Francki & Crowley, Virology 37: 396, 1969.
- Behncken, Australas. Pl. Path. 12: 64, 1983.
- Boakye & Randles, Aust. J. agric. Res. 25: 791, 1974.
- Banerjee, Microbiol. Rev. 51: 66, 1987.
- Campbell & Lin, CMI/AAB Descr. Pl. Viruses 85, 4 pp., 1972.
- Chambers & Francki, Virology 29: 673, 1966.
- Chambers, Crowley & Francki, Virology 27: 320, 1965.
- Chu & Francki, Ann. appl. Biol. 100: 149, 1982.
- Crowley, Virology 31: 107, 1967.
- Crowley, Harrison & Francki, Virology 26: 290, 1965.
- Dale & Peters, Intervirology 16: 86, 1981.
- Dietzgen & Francki, Virology 166: 486, 1988.
- Dietzgen, Hunter, Francki & Jackson, J. gen. Virol. 70: 2299, 1989.
- Francki & Hatta, In Handbook of Plant Virus Infections and Comparative Diagnosis, p. 491, ed. E. Kurstak, Elsevier/North-Holland, 1981.
- Francki & Randles, Virology 47: 270, 1972.
- Francki & Randles, Virology 54: 359, 1973.
- Francki & Randles, In Negative Strand Viruses, p. 223, ed. B. W. J. Mahy & R. D. Barry, Academic Press, London, 1975.
- Francki & Randles, In Rhabdoviruses Vol. III, p. 135, ed. D. H. L. Bishop, CRC Press Inc., Boca Raton, Fla, 1980.
- Francki, Kitajima & Peters, In Handbook of Plant Virus Infections and Comparative Diagnosis, p. 455, ed. E. Kurstak, Elsevier/North-Holland, 1981.
- Francki, Milne & Hatta, Atlas of Plant Viruses Vol. I, p. 73, CRC Press Inc., Boca Raton, Fla, 1985.
- Fry, Close, Procter & Sunde, N. Z. J. agric. Res. 16: 143, 1973.
- Harrison & Crowley, Virology 26: 297, 1965.
- Ie, CMI/AAB Descr. Pl. Viruses 39, 4 pp., 1970.
- Jackson & Christie, CMI/AAB Descr. Pl. Viruses 205, 5 pp., 1979.
- Jackson, Francki & Zuidema, In The Rhabdoviruses, p. 427, ed, R. R.Wagner, Plenum Press, New York, 1987.
- Kitajima & Costa, Virology 29: 523, 1966.
- Martin & Randles, In Pests, Pathogens and Vegetation, p. 479, ed. J. M. Thresh, Pitman, London, 1981.
- Matthews, Intervirology 17: 1, 1982.
- McLean & Francki, Virology 31: 585, 1967.
- McLean, Wolanski & Francki, Virology 43: 480, 1971.
- Milne & Conti, AAB Descr. Pl. Viruses 312, 5 pp., 1986.
- O'Loughlin & Chambers, Virology 33: 262, 1967.
- Peters, CMI/AAB Descr. Pl. Viruses 62, 3 pp., 1971.
- Peters, CMI/AAB Descr. Pl. Viruses 244, 6 pp., 1981.
- Randles, Current Topics in Vector Res. 1: 169, 1983.
- Randles & Carver, Aust. J. agric. Res. 22: 231, 1971.
- Randles & Coleman, Virology 41: 459, 1970.
- Randles & Coleman, Physiol. Pl. Path. 2: 247, 1972.
- Randles & Crowley, Aust. J. agric. Res. 21: 447, 1970.
- Randles & Francki, Virology 50: 297, 1972.
- Sinha & Behki, CMI/AAB Descr. Pl. Viruses 99, 4 pp., 1972.
- Stubbs & Grogan, Aust. J. agric. Res. 14: 439, 1963.
- Tomlinson, CMI/AAB Descr. Pl. Viruses 9, 4 pp., 1970.
- Toriyama, AAB Descr. Pl. Viruses 322, 5 pp., 1986.
- Toriyama & Peters, J. gen. Virol. 50: 125, 1980.
- Vega, Gracia, Rubio-Huertos & Feldman, Phytopath. Z. 85: 7, 1976.
- Wolanski & Chambers, Virology 44: 582, 1971.
- Wolanski & Francki, Virology 37: 437, 1969.
- Wolanski, Francki & Chambers, Virology 33: 287, 1967.






