Details of DPV and References
DPV NO: 346 December 1989
Family: Virgaviridae
Genus: Tobravirus
Species: Tobacco rattle virus | Acronym: TRV
This is a revised version of DPV 12
Tobacco rattle virus
D. J. Robinson Scottish Crop Research Institute, Invergowrie, Dundee, DD2 5DA, UK
B. D. Harrison Scottish Crop Research Institute, Invergowrie, Dundee, DD2 5DA, UK
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Mauche disease of tobacco was described by
Behrens (1899), and the virus by
Quanjer (1943).
Selected synonyms:
(Most of the names listed below refer to isolates that differ to some extent from the type culture). - Aster ringspot virus (Rev. appl. Mycol. 33: 675)
- Belladonna mosaic virus (Rev. appl. Mycol. 22: 451)
- Potato corky ringspot virus (Rev. appl. Mycol. 26: 76)
- Potato stem mottle virus (stengelbont virus) (Rev. appl. Mycol. 27: 33)
- Ratel virus (Rev. appl. Mycol. 25: 83)
- Tabakmauche Virus (Behrens, 1899 and Rev. appl. Mycol. 37: 402)
- Tabak Streifen und Kräuselkrankheit Virus (Rev. appl. Mycol. 7: 546)
- Belladonna mosaic virus (Rev. appl. Mycol. 22: 451)
-
A virus with straight tubular particles of two predominant lengths, the longer about 190 nm and the shorter 50 to 115 nm, depending on the isolate. Normal particle-producing isolates (called M-type) have two species of genomic RNA, RNA-1 and RNA-2, are readily transmitted by inoculation with sap, and by nematodes in the family Trichodoridae. Other isolates (called NM-type) have only RNA-1, do not produce particles, are transmitted with difficulty by inoculation with sap, and are probably not transmitted by nematodes. The virus has a wide host range including many cultivated species, and occurs in several parts of the world.
Main Diseases
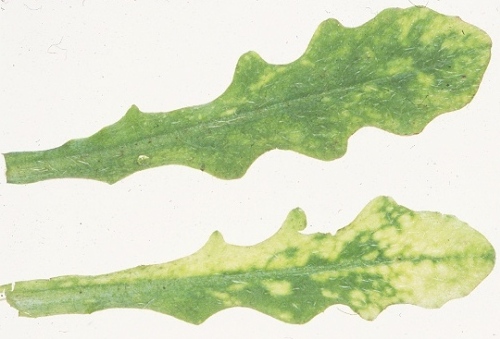
Causes one type of necrotic arcing (known as spraing, corky ringspot, Pfropfenbildung, kringerigheid) in the tuber flesh (Fig. 1), and stem-mottle (distortion, stunting and mottling, typically confined to one or a few of the shoots produced from an infected tuber) and figure aucuba (Fig. 2) in the foliage of potato (Harrison & Robinson, 1981). Systemic infection of potato is rarely complete; thus many spraing-affected tubers give rise to virus-free progeny plants. Other diseases caused include notched leaf in gladiolus (Cremer & Schenk, 1967); malaria (necrotic spots in the bulb flesh) in hyacinth (Van Slogteren, 1958); ringspot in aster (Anderson, 1954); yellow blotch in sugar beet (Gibbs & Harrison, 1964); yellow mottle in spinach (Bailiss & Okonkwo, 1979); rattle (systemic necrotic line patterns, and death or stunting of shoots, also known as Mauche, ratel, Streifen und Kräuselkrankheit) in tobacco; and unnamed diseases in lettuce, hydrangea, narcissus, tulip and several other ornamental species. Many weed species (Fig. 5) become infected in nature, especially in their roots, but several are invaded systemically and some of these, such as Stellaria media, may show no obvious symptoms.
Geographical Distribution
Europe and USSR, Japan, New Zealand, North America.
Host Range and Symptomatology
Has the widest known host range of any plant virus. More than 400 species in more than 50 dicotyledonous and monocotyledonous families can be infected experimentally; in many instances the infection does not become systemic (Uschdraweit & Valentin, 1956; Noordam, 1956; Schmelzer, 1957).
-
Diagnostic species
- Chenopodium amaranticolor.
Necrotic local lesions (Fig. 3), some tending to spread, develop in 3-5 days; most isolates do not become systemic. - Cucumis sativus (cucumber). Chlorotic or necrotic local lesions; not systemic.
- Nicotiana tabacum cv. Samsun NN (tobacco). Characteristic symptoms include necrotic spots or rings in inoculated leaves, and sporadic systemic distortion and/or necrosis. However, symptoms depend on the environmental conditions, and some virus strains produce other symptoms, or none at all.
- Phaseolus vulgaris (French bean). Pin-point, necrotic local lesions develop in 2-4 days (Fig. 4); not systemic.
- Pisum sativum (pea) and Vicia faba (broad bean). Small necrotic local lesions; not systemic.
- Nicotiana tabacum cv. Samsun NN (tobacco). Characteristic symptoms include necrotic spots or rings in inoculated leaves, and sporadic systemic distortion and/or necrosis. However, symptoms depend on the environmental conditions, and some virus strains produce other symptoms, or none at all.
-
Propagation species
- Nicotiana clevelandii,
which becomes infected systemically, is the best host for maintaining cultures and as a source of virus for purification.Assay species
- Chenopodium amaranticolor,
and primary leaves of Phaseolus vulgaris, are suitable for local lesion assay. Petunia hybrida, Nicotiana tabacum ‘White Burley’ and Cucumis sativus are useful bait plants for testing transmission by vectors; infection of the bait plants is best determined by assaying the infectivity of root extracts.
Strains
Many strains are described but few can be reliably distinguished by symptoms in test plants. Indeed, symptomatologically distinguishable variants can often be isolated from a bulk culture (Cadman & Harrison, 1959). Some of the best characterized strains are:
PRN (Cadman & Harrison, 1959; Harrison & Nixon, 1959). Originally obtained from potato in Scotland. Now used as the type strain. The short particles are 78 nm long.
SYM (Kurppa et al., 1981). Originally obtained from spinach in England. Causes systemic distortion and necrosis in Chenopodium amaranticolor unless dilute inocula are used. The short particles are 101 nm long.
Oregon strains. Originally obtained from potato in Oregon. Separated and characterized by Lister & Bracker (1969). One variant induces yellow ringspots and line patterns in N. clevelandii and N. glutinosa. The short particles are 81, 90 or 100 nm long.
Italian No. 6. (Van Hoof et al., 1966). Originally obtained from tobacco bait seedlings planted in soil from Italy. Serologically related to and classified as pea early-browning virus (English serotype) by Harrison (1973), but re-assigned to tobacco rattle virus by Robinson et al. (1987). The short particles are 105 nm long.
N5. Originally obtained from narcissus in Scotland (Harrison et al., 1983). Causes severe necrosis in N. clevelandii, and kills many of the plants. Serologically related to the Dutch serotype of pea early-browning virus (Robinson et al., 1987). The short particles are 75 nm long.
PSG and TCM. Originally obtained from potato and tulip, respectively, in the Netherlands (Cornelissen et al., 1986). TCM is serologically related to the Dutch serotype of pea early-browning virus.
Isolates unable to produce nucleoprotein particles (NM-type isolates) can be obtained from any of the above strains by using inocula containing only long particles. They are poorly transmissible by mechanical inoculation with sap but more easily transmitted by using nucleic acid inocula made with the aid of phenol (Sanger & Brandenburg, 1961). Many of them cause more necrosis in plants than do their parent M-type cultures (Fig. 9) and they are slow to become systemic (Cadman, 1962). Similar isolates are found in naturally infected plants.
Transmission by Vectors
At least seven nematode species in the genera Paratrichodorus and Trichodorus (Trichodoridae) are natural vectors in Europe, N. America or Japan (Brown et al., 1989). There is some evidence of specificity between virus strain and vector species (Van Hoof, 1968; Robinson et al., 1988). Adults and juveniles can transmit, but virus is probably not retained through the moult. The virus can be acquired by P. allius in 1 h, inoculated in 1 h (Ayala & Allen, 1968) and retained for many months by non-feeding nematodes (Van Hoof, 1970). The virus particles become attached to the oesophageal wall of the nematodes (Taylor & Robertson, 1970, Fig. 7) and are thought to be egested with saliva into root cells when the nematodes feed. There is no evidence for multiplication of the virus in the vector and it is probably not transmitted through nematode eggs (Ayala & Allen, 1968).
Transmission through Seed
Up to 10% in Viola arvensis, and lower frequencies in some other weeds (Lister & Murant, 1967; Cooper & Harrison, 1973).
Transmission by Dodder
At least six Cuscuta spp. can transmit. The virus infects the dodder (Schmelzer, 1956).
Serology
Several strains are poorly immunogenic but antisera with tube precipitin titres of 1/1000 have been made against others. In tube precipitin tests, the precipitates are intermediate between somatic and flagellar in type. Immunosorbent electron microscopy and enzyme-linked immunosorbent assay (Harrison et al., 1983) are now the most used tests. Double diffusion tests in agar or agarose gel are insensitive.
Relationships
Tobacco rattle virus has similar particle and genome properties to other tobraviruses. Indeed, distant serological relationships to pea early-browning virus (Maat, 1963) and pepper ringspot virus (Harrison & Woods, 1966; Kurppa et al., 1981) have been reported, but these viruses do not react with tobacco rattle virus RNA-1 probes in hybridization tests (Robinson & Harrison, 1985a; Robinson et al., 1987), and do not form pseudo-recombinants with tobacco rattle virus (Frost et al., 1967; Lister, 1968; Robinson & Harrison, 1985b). Many serotypes of tobacco rattle virus exist (Harrison & Woods, 1966; Robinson & Harrison, 1985a), and members of different serotypes are only distantly serologically related; there is also evidence for serological variation within serotypes. However, all isolates cross-react in nucleic acid hybridization tests with probes for RNA-l (Robinson & Harrison, 1985a; Robinson et al., 1987), and pseudo-recombinant isolates are readily produced with all combinations of RNA-1 and RNA-2 (Harrison & Robinson, 1986). Some strains, such as Italian No. 6 and N5, are closely serologically related to one or other of two serotypes of pea early-browning virus, but are classified as tobacco rattle virus on the basis of nucleic acid hybridization tests with RNA-1 probes and pseudo-recombination tests (Robinson et al., 1987).
Stability in Sap
In N. clevelandii sap, the thermal inactivation point (10 min) of M-type isolates is 80-85°C, dilution end-point 10-5 to 10-6, and infectivity is retained at 20°C for more than 6 weeks and at -20°C for many years. By contrast, NM-type isolates lose infectivity when sap is heated for 10 min at 50°C, diluted to 10-1, kept at 20°C for 1 h or frozen at -20°C (Cadman & Harrison, 1959; Cadman, 1962).
Purification
Systemically infected N. clevelandii leaves yield 10-100 mg virus per kg leaf.
1. (Harrison & Nixon, 1959;
Robinson, 1983).
Store sap at -20°C. Thaw, clarify by low-speed
centrifugation and precipitate virus particles with 10% (w/v) polyethylene glycol
M. Wt 6000 and 2%
(w/v) NaCl. Allow pellets to resuspend in 70 mM phosphate buffer
(pH 7.5) for 16 h at 4°C, and
further purify virus particles by two cycles of differential centrifugation.
2. (Lister & Bracker, 1969). Grind cooled leaves in 10 mM citric acid + phosphate buffer (pH 7.4), containing 0.1% sodium thioglycollate. Clarify extract by blending with 0.5 vol. of a 1:1 mixture of butan-1-ol and chloroform and freezing aqueous layer overnight. Thaw and purify virus particles by differential centrifugation, resuspending them in 10 mM phosphate buffer (pH 7.0).
Properties of Particles
Tubular particles of two predominant lengths (Fig. 6), the longer ones (L) 185-196 nm and the shorter ones (S) ranging from 50 to 115 nm, depending on the isolate (Harrison & Robinson, 1978). L and S particles contain RNA-1 and RNA-2 respectively and can be separated by sedimentation in sucrose density gradients (Fig. 8). L particles are infective alone and induce synthesis of RNA-1 but not of virus particles. S particles are non-infective alone but carry the gene for the particle protein. L and S particles are produced when the inoculum contains them both (Lister, 1966; Frost et al., 1967; Lister & Bracker, 1969; Sänger, 1969). Many strains produce in addition small amounts of shorter particles that contain subgenomic RNA species (Robinson et al., 1983).
Sedimentation coefficients (s20,w; svedbergs): 296-306 (L), 155-245 (S) (Harrison & Woods, 1966; Semancik, 1966).
Buoyant density (g/cm3) in CsCl: 1.324 (Cooper & Mayo, 1972).
Particle weight (daltons x 10-6): 48-50 (L) and 11-29 (S) (Harrison & Klug, 1966).
Electrophoretic mobility (cm2 sec-1 V-1): -1.7 x 10-5 (strain PRN, I = 0.15, pH 7.0) (Harrison & Nixon, 1959); -1.1 x 10-5 (strains B and C, I = 0.1, pH 7.0) (Semancik, 1966).
Extinction coefficient (A260nm, 0.1%, 1cm): about 3.0 (Lister & Bracker, 1969).
A260/A280: 1.10-1.15 (Harrison & Nixon, 1959).
Particle Structure
Particles are tubular with helical symmetry, 23 nm in diameter with a central canal of diameter about 5 nm (Nixon & Harrison, 1959; Cooper & Mayo, 1972). Number of subunits per turn = n + 1/3. Alternative values proposed for n are 25 (Finch, 1965), or about 32 (Roberts & Mayo, 1980). Pitch = 2.5 nm (Finch, 1965).
Particle Composition
Nucleic acid: RNA, single-stranded, linear; two genomic species, RNA-1 and RNA-2, packaged separately in L and S particles respectively, and comprising 5% of the particle weight. RNA-l of strain SYM comprises 6791 nucleotides (M. Wt 22 x 106) composed of 25.6% G, 29.3% A, 16.6% C, 28.5% U (Hamilton et al., 1987); RNA-1 of other strains is of similar size and composition. M. Wt of RNA-2 ranges from 0.6 to 1.4 x 106, depending on the strain; for strain PSG, it comprises 1905 nucleotides composed of 22.9% G, 29.4% A, 17.7% C, 30.0% U (Cornelissen et al., 1986). Two subgenomic RNA species are packaged in separate particles: RNA-1a, derived from RNA-1, M. Wt 0.54 x 106 in several strains; RNA-2a, derived from RNA-2, M. Wt 0.6 x 106 in strain SYM, but of different size in other strains (Robinson et al., 1983, 1987). Subgenomic RNA-1b, derived from the 3' terminal region of RNA-1, does not become packaged in virus particles (Boccara et al., 1986).
Protein. 95% of particle weight. One species, of M. Wt 22,856 (strain PSG) (Cornelissen et al., 1986) or 22,405 (strain TCM) (Angenent et al., 1986), and of similar size in other strains (Mayo & Robinson, 1975). Amino acid compositions for other strains are given by Offord & Harris (1965), Semancik (1966), Miki & Okada (1970) and Ghabrial & Lister (1973). On storage of virus particles in the absence of antimicrobial agents, the coat protein is partially degraded to a lower M. Wt form (Mayo & Cooper, 1973).
Genome Properties
Nucleotide sequences have been determined for RNA-1 of strain SYM (Hamilton et al., 1987), RNA-2 of strain PSG (Cornelissen et al., 1986) and RNA-2 of strain TCM (Angenent et al., 1986). 5' termini are probably capped (Pelham, 1979), and 3' termini have tRNA-like secondary structures including ‘pseudoknots’ (Van Belkum et al., 1987), although no amino acid acceptor activity has been detected. There is no genome-linked protein or polyadenylate sequence.
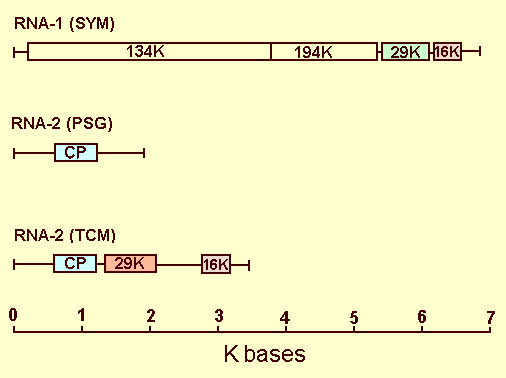
RNA-1 is capable of independent replication and systemic spread in plants. It carries genetic determinants for lesion type and for systemic infection in Nicotiana spp. (Cadman & Harrison, 1959; Sänger, 1969). It contains four open reading frames (Fig. 10), coding for (from the 5' end): a 134 K protein terminated by an opal stop codon; a 194 K protein, produced by read-through of this stop codon, which has regions of homology with the putative replicase of tobacco mosaic virus; a 29 K protein which has homology with the putative 30 K transport protein of tobacco mosaic virus (Boccara et al., 1986); and a 16 K protein of unknown function.
RNA-2 carries genetic determinants for ability to cause systemic symptoms in Chenopodium amaranticolor (Kurppa et al., 1981) and yellow mosaic in tobacco (Lister & Bracker, 1969), and for serological specificity (Sänger, 1969). It codes for the coat protein. There is sequence homology between RNA-1 and RNA-2 at both ends, but the extent of this homology varies between strains; in some strains the homologous region at the 3' end is large enough to include some or all of the 16 K and 29 K open reading frames of RNA-1 (Cornelissen et al., 1986; Robinson et al., 1987). RNA-2 of strain TCM contains an additional open reading frame, between the coat protein gene and the 3' terminal homologous region, coding for a 29 K protein that is not homologous to that coded for by RNA-1 (Angenent et al., 1986) (Fig. 10).
In vitro, and presumably in vivo, the 134 K and 194 K proteins are translated from RNA-1 (Fritsch et al., 1977), whereas the 29 K, 16 K and coat proteins are translated from subgenomic RNA species 1a, 1b and 2a, respectively (Robinson et al., 1983; Boccara et al., 1986; Cornelissen et al., 1986).
Relations with Cells and Tissues
Most tissues of systemically invaded plants become infected. In leaf hair cells of N. clevelandii, ‘X-bodies’ largely composed of abnormal mitochondria can be induced by strain PRN, but persist for only a few days. Small aggregates of virus particles occur in the X-bodies and elsewhere in the cytoplasm (Harrison et al., 1970).
Ecology and Control
Tobacco rattle virus is essentially a virus of weeds and other wild plants (Noordam, 1956; Cooper & Harrison, 1973; Fig. 5), and its distribution in soil reflects that of its vectors, which prefer light sandy or peaty soils (Cooper, 1971). It may be patchily distributed within a field. Control measures include the use of nematicidal and nematostatic chemicals, and the planting of resistant cultivars or virus-free stock (Harrison & Robinson, 1986).
Notes
Tobacco rattle virus is distinguished from viruses in other virus groups by its characteristic particles, together with its symptoms in diagnostic hosts and ability to produce NM isolates. Strain CAM and other South American isolates, previously classified as tobacco rattle virus serotype III, are now considered to be isolates of pepper ringspot virus (Robinson & Harrison, 1985a). Pepper ringspot virus is at best only distantly serologically related to tobacco rattle virus and there is no cross-reaction in nucleic acid hybridization tests with RNA-1 probes (Robinson & Harrison, 1985a). Tobacco rattle virus can be distinguished from pea early-browning virus by failure to infect pea or Phaseolus vulgaris systemically, by production of pinpoint lesions in P. vulgaris, and by nucleic acid hybridization tests using RNA-1 probes. M isolates of tobacco rattle virus can sometimes be identified by serological tests but the great antigenic differences between strains make negative results in such tests inconclusive. However, all M isolates can be detected by nucleic acid hybridization tests with RNA-1 probes (Robinson & Harrison, 1985a; Robinson et al., 1987). NM isolates can also be identified in hybridization tests for RNA-1, or by adding S particles or RNA-2 to the culture to generate an M isolate (Harrison & Robinson, 1982).
Acknowledgements
Photographs courtesy of the Scottish Crop Research Institute. Fig. 2 from Harrison, 1971; Fig. 7 from Taylor & Robertson, 1970; Fig. 9 from Harrison & Robinson, 1981
Figures

Tuber of naturally infected potato (Solanum tuberosum cv. Pentland Dell) cut to show necrotic arcs in the flesh.

Virus particles in longitudinal chordal section of oesophagus of Paratrichodorus pachydermus. Arrows indicate the oesophageal walls. Bar represents 200 nm.

Separation of L and S particles by rate zonal sedimentation in a sucrose density gradient. M marks the meniscus.
References list for DPV: Tobacco rattle virus (346)
- Anderson, Phytopathology 44: 87, 1954.
- Angenent, Linthorst, Van Belkum, Cornelissen & Bol, Nucleic Acids Res. 14: 4673, 1986.
- Ayala & Allen, J. agric. Univ. P. Rico 52: 101, 1968.
- Bailiss & Okonkwo, J. hort. Sci. 54: 289, 1979.
- Behrens, Landwn VersStnen 52: 422, 1899.
- Boccara, Hamilton & Baulcombe, EMBO J. 5: 223, 1986.
- Brown, Ploeg & Robinson, Revue Nématol. 12: 235, 1989.
- Cadman, Nature, Lond. 193: 49, 1962.
- Cadman & Harrison, Ann. appl. Biol. 47: 542, 1959.
- Cooper, Pl. Path. 20: 51, 1971.
- Cooper & Harrison, Ann. appl. Biol. 73: 53, 1973.
- Cooper & Mayo, J. gen. Virol. 16: 285, 1972.
- Cornelissen, Linthorst, Brederode & Bol, Nucleic Acids Res. 14: 2157, 1986.
- Cremer & Schenk, Neth. J. Pl. Path. 73: 33, 1967.
- Finch, J. molec. Biol. 12: 612, 1965.
- Fritsch, Mayo & Hirth, Virology 77: 722, 1977.
- Frost, Harrison & Woods, J. gen. Virol. 1: 57, 1967.
- Ghabrial & Lister, Virology 52: 1, 1973.
- Gibbs & Harrison, Pl. Path. 13: 144, 1964.
- Hamilton, Boccara, Robinson & Baulcombe, J. gen. Virol. 68: 2563, 1987.
- Harrison, in Diseases of Crop Plants, p. 123, ed. J. H. Western, London: Macmillan, 404 pp.,1971.
- Harrison, CMI/AAB Descr. Pl. Viruses 120, 4 pp., 1973.
- Harrison & Klug, Virology 30: 738, 1966.
- Harrison & Nixon, J. gen. Microbiol. 21: 569, 1959.
- Harrison & Robinson, Adv. Virus Res. 23: 25, 1978.
- Harrison & Robinson, in Handbook of Plant Virus Infections and Comparative Diagnosis, p.515, ed. E. Kurstak, Amsterdam: Elsevier/North-Holland, 943 pp., 1981.
- Harrison & Robinson, J. virol. Methods 5: 255, 1982.
- Harrison & Robinson, in The Plant Viruses, vol. 2, p. 339, eds M.H.V. Van Regenmortel & H. Fraenkel-Conrat, New York: Plenum, 424 pp., 1986.
- Harrison & Woods, Virology 28: 610, 1966.
- Harrison, Stefanac & Roberts, J. gen. Virol. 6: 127, 1970.
- Harrison, Robinson, Mowat & Duncan, Ann. appl. Biol. 102: 331, 1983.
- Kurppa, Jones, Harrison & Bailiss, Ann. appl. Biol. 98: 243, 1981.
- Lister, Virology 28: 350, 1966.
- Lister, J. gen. Virol. 2: 43, 1968.
- Lister & Bracker, Virology 37: 262, 1969.
- Lister & Murant, Ann. appl. Biol. 59: 49, 1967.
- Maat, Neth. J. Pl. Path. 69: 287, 1963.
- Mayo & Cooper, J. gen. Virol. 18: 281, 1973.
- Mayo & Robinson, Intervirology 5: 313, 1975.
- Miki & Okada, Virology 42: 993, 1970.
- Nixon & Harrison, J. gen. Microbiol. 21: 582, 1959.
- Noordam, Tijdschr. PlZiekt. 62: 219, 1956.
- Offord & Harris, Proceedings of the 2nd FEBS Meeting, p. 216, 1965.
- Pelham, Virology 97: 256, 1979.
- Quanjer, Tijdschr. PlZiekt. 49: 37, 1943.
- Roberts & Mayo, J. Ultrastruct. Res. 71: 49, 1980.
- Robinson, J. gen. Virol. 64: 657, 1983.
- Robinson & Harrison, J. gen. Virol. 66: 171, 1985a.
- Robinson & Harrison, J. gen. Virol. 66: 2003, 1985b.
- Robinson, Mayo, Fritsch, Jones & Raschké, J. gen. Virol. 64: 1591, 1983.
- Robinson, Hamilton, Harrison & Baulcombe, J. gen. Virol. 68: 2551, 1987.
- Robinson, Brown & Ploeg, Rep. Scott. Crop Res. Inst., 1987: 189, 1988.
- Sänger, J. Virol. 3: 304, 1969.
- Sänger & Brandenburg, Naturwissenschaften 48: 391, 1961.
- Schmelzer, Phytopath. Z. 28: 1, 1956.
- Schmelzer, Phytopath. Z. 30: 281, 1957.
- Semancik, Phytopathology 56: 1190, 1966.
- Taylor & Robertson, J. gen. Virol. 6: 179, 1970.
- Uschdraweit & Valentin, NachrBl. dt. PflSchutzdienst., Stuttg. 8: 132, 1956.
- Van Belkum, Cornelissen, Linthorst, Bol, Pleij & Bosch, Nucleic Acids Res. 15: 2837, 1987.
- Van Hoof, Nematologica 14: 20, 1968.
- Van Hoof, Neth. J. Pl. Path. 76: 329, 1970.
- Van Hoof, Maat & Seinhorst, Neth. J. Pl. Path. 72: 253, 1966.
- Van Slogteren, Tijdschr. PlZiekt. 64: 452, 1958.