Details of DPV and References
DPV NO: 353 December 1989
Family: Closteroviridae
Genus: Closterovirus
Species: Citrus tristeza virus | Acronym: CTV
This is a revised version of DPV 33
Citrus tristeza virus
M. Bar-Joseph The S. Tolkowsky Laboratory, Department of Virology, The Volcani Centre, Bet Dagan, Israel
R. F. Lee University of Florida, Institute of Food and Agricultural Science, Citrus Research and Education Center, Lake Alfred, Florida 33850, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Disease described by Webber (1925) and
Toxopeus (1937); aphid and graft transmissibility reported by
Meneghini (1946) and
Fawcett & Wallace (1946).
Selected synonyms
- Citrus quick decline virus (Fawcett & Wallace, 1946)
- Podredumbre de las raicillas in Argentina (Zeman, 1931)
- Lime die-back virus (Hughes & Lister, 1949)
- Grapefruit stem pitting virus (Oberholzer et al., 1949)
- Hassaku dwarf virus (Tanaka et al., 1969)
- Podredumbre de las raicillas in Argentina (Zeman, 1931)
-
A virus with flexuous filamentous particles 2000 nm long and 10-11 nm in diameter containing single-stranded RNA. Infects a narrow range of hosts mostly restricted to the Rutaceae. Transmitted mechanically with difficulty and in a semi-persistent manner by a few aphid species.
Main Diseases
Tristeza, or quick decline (Fig. 1), of many commercial varieties of Citrus on sour orange (C. aurantium) rootstocks; the severity of symptoms depends on the virus strain, some strains causing almost no damage. Tristeza was first recognized as a scion/rootstock disease, no symptoms appearing in plants on tolerant rootstocks. Symptoms include rapid wilting of trees on sour orange rootstocks, and root damage; honeycombing often occurs immediately below the bud union on the sour orange stock. When trees decline rapidly, a yellow-brown stain is sometimes present at the bud union and on the inner surface of the bark patch (Fig. 2). Stunting of young trees propagated on sour orange rootstocks occurs in Florida. Stem-pitting and loss of plant vigour occurs on grapefruit (C. paradisi) scions in South Africa, Australia, and in the Far East (Fig. 3); and stem-pitting regardless of the rootstock occurs on certain sweet orange (C. sinensis) varieties, e.g. Pera in Brazil, California and the Far East. Probably the most devastating disease caused by the virus is dieback of limes (C. aurantifolia) irrespective of rootstock. Certain Passiflora species in the jungles of South America show serious wilt symptoms.
Geographical Distribution
Common in the Far East, Australia, Africa, India, South, Central and North America, and most other citrus growing areas. Only certain parts of the Mediterranean region are still free of the virus.
Host Range and Symptomatology
Most varieties of Citrus and a few species in certain other genera of the family Rutaceae, such as Aegle marmelos, Aeglopsis chevalieri, Afraegle paniculata, Citropsis gilletiana, Microcitrus australis, and Pamburus missionis, are hosts (Muller & Garnsey, 1984). A majority are tolerant, however, of commonly occurring strains of the virus. Certain Passiflora species such as P. gracilis (Muller et al., 1974), P. caerulea, P. incense and P. incarnata (Roistacher & Bar-Joseph, 1987b) are sensitive, whereas other species are tolerant or immune (Bar-Joseph et al., 1989).
-
Diagnostic species
- Citrus aurantifolia
(Key lime, Mexican lime, and various other names in different parts of the world) is the most commonly used indicator plant. It reacts to most strains of the virus by developing yellow flecks along leaf veins on the upper leaf surface and water-soaked spots on the lower leaf surface. Intensity of symptoms depends on the virus strain and the time elapsed after inoculation. Infected leaves are smaller, and may show cupping and chlorosis. Virulent strains induce vein corking on the upper leaf surface (Fig. 4) and may cause severe stem-pitting under the bark. -
C. medica (citron) (Fig. 5) and C. macrophylla (alemow) show symptoms similar to those in C. aurantifolia.
- C. sinensis (sweet orange): young plants on sour orange (C. aurantium) rootstocks show decline, the severity depending on the virus strain.
- C. aurantium (sour orange), lemon (C. limon) and grapefruit (C. paradisi) seedlings show stunting and severe yellows when infected with seedling yellows strains of tristeza virus.
- A battery of five indicator plants (Mexican lime seedlings, Navel budded onto sour orange, sour orange seedlings, Duncan grapefruit seedlings and Madam Vinous seedlings) has been proposed to index for tristeza infection severity (Garnsey et al., 1987).
- C. sinensis (sweet orange): young plants on sour orange (C. aurantium) rootstocks show decline, the severity depending on the virus strain.
-
Propagation species
- C. aurantifolia, C. excelsa, C. hystrix,
and C. macrophylla are good species for maintaining cultures for purification purposes. C. sinensis is the best species for maintaining virus cultures over long periods.
Strains
The virus probably has one of the most diverse ranges of strains with a wide range of biological activities. There are four major groups of strains. Mild strains also exist which are almost symptomless in sensitive indicator plants such as Mexican lime.
Seedling yellows strains. Cause severe stunting and yellowing of lemon, sour orange and grapefruit. Seedling yellows is principally a greenhouse disease, but lemons and grapefruit topworked on rootstocks carrying seedling yellows strains of tristeza virus can show field symptoms.
Grapefruit stem pitting strains. In Australia (Stubbs, 1964), South Africa (Oberholzer et al., 1949), and the Far East (Miyakawa, 1971), infected grapefruit trees are stunted, the fruit is small and misshapen, and yields are considerably reduced; the wood of the trunk and large limbs is pitted with longitudinal depressions; in some instances, the main scaffold branches are twisted and distorted.
Sweet orange stem pitting strains. In Brazil, Colombia, Hawaii and the Far East, severe isolates induce severe stem-pitting and decline in certain sweet orange varieties regardless of the rootstock combination. With the Capao Bonito strain in Brazil (Muller et al., 1968) and Hassaku dwarf strain in Japan (Miyakawa, 1971), fruit size and yield are considerably reduced.
Lime die-back strain (Hughes & Lister, 1949). Causes vein flecking on young leaves, severe pitting of wood of twigs and branches, stunting of trees, and die-back with eventual death.
Transmission by Vectors
Toxoptera citricida, the oriental citrus aphid, and Aphis gossypii are the most efficient vectors (Meneghini, 1946; Costa & Grant, 1951; Bar-Joseph & Loebenstein, 1973; Roistacher & Bar-Joseph, 1987a, 1987b). A. citricola and in a few cases T. aurantii also have been reported to transmit the virus (Dickson et al., 1951; Norman & Grant, 1956). The virus can be acquired and transmitted by aphids, usually after acquisition and inoculation feeding periods of at least 30 min; frequencies of transmission are positively correlated with the length of the acquisition period up to 24 h and the length of the inoculation period up to 6 h (Costa & Grant, 1951; Raccah et al., 1976). Transmission is in the semi-persistent manner with no latent period; infectivity is lost within 48 h of acquisition (Bar-Joseph et al., 1979a).
Transmission through Seed
None reported.
Transmission by Dodder
Reported to be transmitted by Cuscuta americana (Knorr & Price, 1954; Price, 1970), but this report is not confirmed.
Serology
Moderately immunogenic. Sodium dodecyl sulphate (SDS)-immunodiffusion (Gonsalves et al., 1978; Garnsey et al., 1979), enzyme-linked immunosorbent assay (ELISA) (Bar-Joseph et al., 1979b), radio-immunosorbent assay (Lee et al., 1981), immunofluorescent assays (Tsuchizaki et al., 1978; Sasaki et al., 1978; Brlansky et al., 1984), immunospecific electron microscopy (ISEM) (Garnsey et al., 1980; Brlansky et al., 1984), and western blotting (Lee et al., 1987) have been used for detecting the virus. Polyclonal antisera have been prepared in rabbits against unfixed virus particles, formaldehyde-fixed particles, and SDS-dissociated particles (Gonsalves et al., 1978). Multiple epitopes were suggested based on the types of serological assay in which antisera were effective (Brlansky et al., 1984). Polyclonal antisera also have been produced in chickens (Bar-Joseph & Malkinson, 1980). The double antibody sandwich (DAS) form of ELISA has been the most commonly used (Bar-Joseph et al., 1979b), but several modifications have been developed. Indirect ELISA systems use commercially available goat anti-rabbit, anti-chicken or anti-mouse conjugates. Avidin-biotin (Irey et al., 1988) and enzyme-amplification (Ben-Ze'ev et al., 1988) have been used to increase the sensitivity of ELISA for detecting citrus tristeza virus. Plate trapped antigen (PTA)-ELISA also has been applied, especially for use with monoclonal antibodies (Vela et al., 1986; Permar et al., 1990). All polyclonal antibodies so far produced against citrus tristeza virus appear to react with all strains tested (Bar-Joseph et al., 1989). Several monoclonal antibodies have been produced; most have reacted with all citrus tristeza virus strains tested (Vela et al., 1986; Gumpf et al., 1987), but one is capable of differentiating between severe and mild strains (Irey et al., 1988; Permar et al., 1990).
Relationships
The chemical and physical properties of the particles place the virus as a member of the closterovirus group. No serological cross-reactivity with other members of the closterovirus group has been reported.
Some mild strains, but not all, are capable of cross-protecting plants against severe strains (Fulton, 1986; Fig. 6). Mild strain cross-protection is used commercially to protect against stem-pitting of Pera sweet orange in Brazil (Costa & Muller, 1980) and against stem-pitting of grapefruit in South Africa (De Lange et al., 1981).
Stability in Sap
The virus is mechanically transmissible by slash inoculation of the bark of citron plants using extraction procedures that maintain virus integrity (Garnsey et al., 1977; Muller & Garnsey, 1984). This method of assay has been used to study the stability of the virus in crude sap extracts from infected bark; the thermal inactivation point (10 min) is about 50°C, and infectivity of sap persists for 24 h at room temperature, 5 days at 4°C and -60°C, and for 5 years in lyophilized preparations stored at -20°C. Infectivity is destroyed by RNase, but not by DNase. Infectivity persists after incubation in a solution of 0.005 M EDTA, 0.5 M NaCl and 0.005 M MgCl2. The virus retained infectivity when dialysed against pH 6.0 to 9.0 buffers, then back to pH 7.6 Tris-HCI buffer, but not when dialysed at pH values lower than 6.0 (Garnsey et al., 1981; R. F. Lee & R. H. Brlansky, unpublished data).
Purification
1. Bar-Joseph et al., (1985). Grind bark from young flushes of infected citron plants with a mortar and pestle in the presence of liquid nitrogen; thaw frozen powder in 0.1 M Tris-HCl, pH 7.8 (1 g bark powder/5 ml buffer), filter, re-extract pulp, combine filtrates, centrifuge at 4000 g for 10 min. Collect supernatant fluid and centrifuge at 8000 g for 5 min. Filter supernatant fluid through paper tissue; add polyethylene glycol, M. Wt 6000 (PEG) and NaCl to final concentrations of 6% and 1.1% (w/v), respectively. After 30-60 min at 4°C, centrifuge at 19,500 g for 15 min. Resuspend pellet in 25 ml 0.04 M sodium phosphate buffer, pH 8.2, (PB) for 1 h, centrifuge at 5900 g for 10 min. Float 8 ml samples of supernatant fluid on step gradients made by layering 1 ml each of 0, 15, 22.5 and 30% (w/v) Cs2SO4 dissolved in a solution of PB and 10% (w/v) sucrose. Centrifuge 2.5 h at 38,500 rev./min in a Beckman SW41 rotor at 8°C. Locate the virus-containing fractions by EM or ELISA. For further purification, dialyse against 0.05 M Tris-HCl, pH 7.8, and centrifuge in a 10-40% sucrose gradient for 3 h at 25,000 rev./min at 4°C in a Beckman SW27 rotor; concentrate the virus particles by ultracentrifugation and resuspend in 0.05 M Tris-HCl buffer, pH 7.8.
2. Lee et al. (1987). Collect bark tissue and leaf midribs from young flushes of citron, Mexican lime or C. excelsa, freeze on dry ice and pulverize in a mortar and pestle. Homogenize the frozen tissue powder in 0.1 M Tris-HCl buffer, pH 8.4, containing 0.1% (v/v) Triton X-100 (1 g tissue/5 ml buffer). Centrifuge at 10,000 g for 20 min, collect supernatant fluid and add PEG and NaCl to final concentrations of 4.0 and 0.8% (w/v), respectively. Stir 1 h at 4°C, centrifuge at 10,000 g for 20 min. Resuspend pellet in 0.04 M potassium phosphate buffer, pH 8.0 (1.3 ml/g tissue fresh wt). Stir suspension for 1 h at 4°C, then centrifuge at 5000 g for 10 min. Adjust supernatant fluid to a final concentration of 5% (w/v) PEG, 1% (w/v) NaCl, and stir 1 h at 4°C. Centrifuge mixture at 10,000 g for 15 min. Resuspend pellet in 30 ml 0.05 M Tris-HCl, pH 8.0, stir 1 h at 4°C. Centrifuge suspension at 5000 g for 10 min, float 5 ml supernatant fluid onto preformed step isopycnic (PSI) gradient made by overlayering 2.0, 2.0, 2.0 and 1.0 ml of 8.83, 17.66, 26.5 and 35.33% (all w/w), respectively, of Cs2SO4 dissolved in 0.05 M Tris-HCl buffer, pH 8.0. Centrifuge at 36,000 rev./min for 15 h at 4°C in a Beckman SW41 rotor. Identify the virus-containing fractions by ELISA and/or ISEM, dialyse overnight against 0.05 M Tris-HCl buffer, pH 8.0, and centrifuge at 10,000 g for 10 min. Adjust volume of supernatant fluid to 22 ml with 0.05 M Tris-HCl buffer, pH 8.0, mix with 17.0 ml of a 53% (w/w) solution of Cs2SO4 in 0.05 M Tris-HCl buffer, pH 8.0, place in VTi50 (or Type 60) rotor and centrifuge at 50,000 rev./min for 24 h at 4°C, then locate the virus-containing fractions by ELISA and/or ISEM. Yields of 0.4 to 5.1 A260 units per 100 g bark tissue are obtained, depending upon virus strain and purification host.
3. Lee et al. (1988). Strip bark from young flushes, chop into pieces about 1-2 mm wide, and add to extraction buffer containing 0.1 M sodium citrate, pH 6.0, with 2% Driselase enzyme, 10% sucrose, 0.5% 2-mercaptoethanol and 1 mM phenylmethylsulphonylfluoride (1 g tissue/4 ml buffer). Shake preparation at 6°C for 1-2 h, add 1/200 volume of 3.0 N NaOH to raise pH, and freeze at -20°C for at least 1 h. Thaw, stir with a glass rod, filter, and centrifuge at 9000 g for 10 min at 4°C. Add Triton X-100 to supernatant fluid to a final concentration of 0.2%. Overlay 27 ml of the virus preparation onto a step gradient formed by layering 5 ml 25% sucrose over 5 ml 60% sucrose, both made up in 0.05 M Tris-HCl, pH 8.0. Centrifuge at 19,000 rev./min for 15 h at 4°C in a Beckman SW28 rotor. Collect 1.5 ml fractions from the bottom of the tube, and locate the virus by ELISA; it is usually in the second and third fractions. Pool virus-containing fractions and layer onto a 1.5 x 7 cm column of Bio-gel A-15, 100-200 mesh; collect 1 ml fractions in tubes containing 0.25 ml 25% sucrose in 0.05 M Tris-HCl buffer, pH 8.0. The virus elutes in the void volume; pool virus-containing fractions and adjust to 22.5 ml with 0.05 M Tris-HCl buffer, pH 8.0, then mix with 17.5 ml of 3 molal Cs2SO4 made in 0.05 M Tris-HCl buffer, pH 8.0. Centrifuge at 50,000 rev./min for 18 h at 4°C in a Beckman VTi50 or Type 60 rotor. Locate virus-containing fractions by ELISA, combine and dialyse against 0.05 M Tris-HCl, pH 8.0, containing 10% sucrose, then centrifuge at 9000 g for 10 min at 4°C. Layer supernatant fluid onto a preformed step gradient made up of 2 ml each of 0.5, 1.0 and 1.5 molal Cs2SO4 in 0.05 M Tris-HCl buffer, pH 8.0. Centrifuge at 36,000 rev./min for 15 h at 4°C in a Beckman SW41 rotor. Fractionate tubes, combine virus-containing fractions, dialyse against 0.05 M Tris-HCl buffer, pH 8.0, containing 10% sucrose, and then centrifuge at 8000 g for 10 min at 4°C.
Other purification procedures have been reported by Bar-Joseph et al., (1970), Bar-Joseph et al., (1972), and Gonsalves et al., (1978).
Properties of Particles
Sedimentation coefficient (s20,w) determined in sucrose density gradients: 140 ± 10 S at zero depth (Bar-Joseph et al., 1970).
A260/A280: 1.21 (Bar-Joseph et al., 1972; Tsuchizaki et al., 1978); 1.12 ± 0.01 not corrected for light-scattering (Lee et al., 1987).
Buoyant density: 1.328 g/cm3 in CsCl after formaldehyde fixation (Bar-Joseph et al., 1972); 1.2570 g/cm3 unfixed in Cs2SO4 (Lee et al., 1987).
Particle Structure
Particles are very flexuous filaments 10-11 nm in diameter and 1900-2000 nm in length (Fig. 10), visible in leaf dip preparations, or in large numbers in bark dip preparations, mounted in phosphotungstate (Bar-Joseph et al., 1976). They are well preserved in ISEM in which antisera prepared against unfixed whole virus diluted 1/500 with 0.02 M Tris-HCl buffer, pH 8.0, are used to coat parlodion-coated grids. Grids for ISEM are positively stained for 1 min on drops of 5% uranyl acetate in 50% ethanol, rinsed for 20 sec in 95% ethanol, and air-dried (Lee et al., 1987).
Particle Composition
Nucleic acid: RNA, non-segmented, single-stranded, M. Wt about 6.5 x 106 or 20 kilobases (Bar-Joseph et al., 1979a; Bar-Joseph et al., 1985) and without a poly-A tail at the 3' end (M. Ballas & M. Bar-Joseph, unpublished data). The RNA in the particles is positive sense (Lee et al., 1988).
Protein: Two proteins are associated with purified preparations of Florida strains: a major coat protein (CP1) of 23,000 daltons and a minor coat protein (CP2) of 21,000 daltons, present in a ratio of 5/1 (CP1/CP2). The amino acid composition of CP1 for a severe Florida strain is reported as: (molar ratio) lys 17; his 4; arg 8; asp 22; thr 12; ser 16; glu 13; pro 5; gly 24; ala 16; cys 1; val 13; met 1; ile 8; leu 20; tyr 4; phe 4 (Lee et al., 1988). Recent analysis of the coat proteins of strains from Spain and Israel indicate M. Wt of 27,000-28,000 and 26,000 for CP1 and CP2, respectively (Dulieu & Bar-Joseph, 1990; Guerri et al., 1990).
Genome Properties
The RNA can be translated in vitro in rabbit reticulocyte lysates to produce a product of about 26,000 daltons which is precipitated by antiserum to virus particles (Lee et al., 1988).
Sequence homology between strains was compared using cDNA from a library obtained by random priming (Rosner et al., 1983). Six of nine strains hybridized with all the tested cDNA clones, the three other strains hybridized differentially (Rosner & Bar-Joseph, 1984); two of these cDNA clones were able to detect a specific virulent strain in Florida by differential hybridization (Rosner et al., 1986). cDNA libraries also have been prepared by oligo dT tailing; two clones from these libraries were selected that together represent 70% of the CTV genome with a 2200 bp overlap between the two clones (Calvert, 1987; Calvert et al., 1986, 1987).
A full length replicative form of 13.3 x 106 daltons (Fig. 7) has been found by extraction, purification, and polyacrylamide gel analysis of dsRNA (Dodds & Bar-Joseph, 1983). Also present are a number of ‘subgenomic’ dsRNA species whose size and relative amount depend on virus strain and host (Fig. 7) (Lee, 1984; Dodds et al., 1987). These dsRNA molecules have been denatured and translated in vitro in a rabbit reticulocyte lysate system. The translational product of a 0.8 kbp dsRNA co-electrophoresed with the viral coat protein and was immunoprecipitated by virus- specific antisera (Dulieu & Bar-Joseph, 1990).
Relations with Cells and Tissues
Flexuous filamentous particles occur in large numbers in phloem cells but not in other cells. Aggregates of virus particles, called chromatic cells (Schneider, 1959, 1973) or inclusion bodies (Christie & Edwardson, 1977), are often found in parenchyma or parenchyma-like cells adjacent to sieve tubes in the phloem. These inclusion bodies stain purple with Azure A (Fig. 8), appearing as cross-banded structures (Fig. 9). Fluorescent antibody staining techniques have been used to detect inclusion bodies in situ (Brlansky et al., 1988). Quantitative differences in number of inclusion bodies between mild and severe strains have been reported in certain hosts such as Mexican lime and C. hystrix (Brlansky, 1987). Necrosis occurs at the bud union as a result of destruction of phloem tissues in trees on sour orange rootstock which are undergoing quick decline.
Ecology and Control
Once introduced into a citrus-growing area, tristeza virus is spread naturally by aphids and becomes endemic. Introduction into new areas can be prevented by quarantine measures and budwood certification schemes. The development of rapid detection methods for tristeza virus has facilitated large-scale surveys and eradication schemes which have enabled virus incidence to be suppressed to low levels; survival of the virus in wild hosts has not appeared to present a problem. In some instances, such schemes have prolonged the use of sour orange as a rootstock. In areas where tree losses occur on sour orange rootstocks, replants are usually grown on tristeza-tolerant rootstocks such as rough lemon, Rangpur lime, trifoliate orange or trifoliate hybrids. Resistance to stem-pitting strains of tristeza virus is not available in commercially acceptable grapefruit, sweet oranges or limes, and mild strain cross-protection is presently the only method available for control of these strains.
Notes
Citrus blight, a disease of unknown aetiology, results in decline symptoms in trees on sour orange rootstocks similar to those induced by tristeza virus. Tristeza decline on sour orange rootstocks can be differentiated from blight by the syringe injection test (Lee et al., 1984); tristeza-affected trees take up water in the trunk wood whereas blight-affected trees do not.
Citrus greening, a disease caused by a fastidious, phloem-limited bacterium, is endemic in many of the citrus-growing areas where tristeza virus also is endemic, and its symptoms are often similar to those of tristeza. Greening can be diagnosed by grafting to seedlings of mandarin orange, a differential indicator which does not react to tristeza virus.
Figures

Sweet orange (C. sinensis) tree on sour orange (C. aurantium) rootstock showing quick decline caused by infection with a virulent strain of tristeza virus.

Yellow-brown stain as sometimes seen at the bud union of trees on sour orange rootstocks and showing tristeza quick decline.

Severe stem-pitting on the trunk of a grapefruit (C. paradisi) tree caused by a stem-pitting strain of tristeza virus.

Corking of the leaf veins on the upper leaf surface of a ‘Madam Vinous’ sweet orange indicator plant infected with a stem-pitting strain of tristeza virus.

(Above), Flecking along the veins of a citron (C. medica) leaf infected with tristeza virus. (Below), Healthy leaf.

Mild strain cross-protection in Eureka lemon (C. limon): A, non-inoculated; B, protected with a non-seedling yellows strain and challenge-inoculated with a virulent seedling yellows strain; C, inoculated with the virulent strain at the same time as plant B was challenge-inoculated.
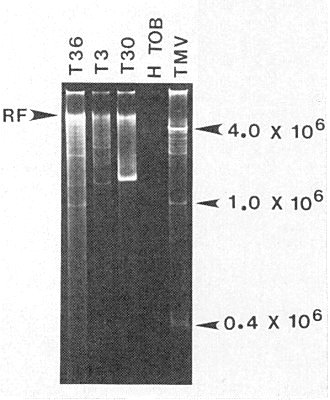
Double stranded (ds) RNA extracted from C. excelsa plants infected with virulent (T36 and T3) and mild (T30) Florida strains and electrophoresed on a polyacrylamide gel. The replicative form (RF) is indicated by an arrowhead. DsRNA preparations from healthy tobacco (H TOB) and from tobacco infected with tobacco mosaic virus (TMV) are included for size comparison.

Light micrograph of inclusion bodies (arrowheads) in phloem cells associated with tristeza virus infection. Stained with Azure A. Bar represents 10 µm. (Photo courtesy of R. H. Brlansky.)
References list for DPV: Citrus tristeza virus (353)
- Bar-Joseph & Loebenstein, Phytopathology 63: 716, 1973.
- Bar-Joseph & Malkinson, J. Virol. Meth. 1: 1, 1980.
- Bar-Joseph, Loebenstein & Cohen, Phytopathology 60: 75, 1970.
- Bar-Joseph, Loebenstein & Cohen, Virology 50: 821, 1972.
- Bar-Joseph, Loebenstein & Cohen, Proc. 7th Conf. int. Org. Citrus Virologists: 39, 1976.
- Bar-Joseph, Garnsey & Gonsalves, Adv. Virus Res. 25: 93, 1979a.
- Bar-Joseph, Garnsey, Gonsalves, Moscovitz, Purcifull, Clark & Loebenstein, Phytopathology 69: 190, 1979b.
- Bar-Joseph, Gumpf, Dodds, Rosner & Ginzburg, Phytopathology 75: 195, 1985.
- Bar-Joseph, Marcus & Lee, A. Rev. Phytopath. 27: 291, 1989.
- Ben-Ze'ev, Frank & Bar-Joseph, Phytoparasitica 16: 343, 1988.
- Brlansky, Phytophylactica 19: 211, 1987.
- Brlansky, Garnsey, Lee & Purcifull, Proc. 9th Conf. int. Org. Citrus Virologists: 337, 1984.
- Brlansky, Lee & Garnsey, Pl. Dis. 72: 1039, 1988.
- Calvert, Ph.D. Thesis, University of Florida, Gainesville, 60 pp., 1987.
- Calvert, Lee & Hiebert, Phytopathology 76: 1090, 1986.
- Calvert, Lee & Hiebert, Phytopathology 77: 1743, 1987.
- Christie & Edwardson, Monogr. Ser. Fla agric. Exp. Stn No. 9, 150 pp., 1977.
- Costa & Grant, Phytopathology 41: 105, 1951.
- Costa & Muller, Pl. Dis. 64: 538, 1980.
- De Lange, Van Vuuren & Bredell, Subtropica 2(5): 11, 1981.
- Dickson, Flock & Johnson, J. econ. Entomol. 44: 172, 1951.
- Dodds & Bar-Joseph, Phytopathology 73: 419, 1983.
- Dodds, Jordan, Roistacher & Jarupat, Intervirology 27: 177, 1987.
- Dulieu & Bar-Joseph, J. gen. Virol. 71: 443, 1990.
- Fawcett & Wallace, Calif. Citrogr. 32: 88, 1946.
- Fulton, A. Rev. Phytopath. 24: 67, 1986.
- Garnsey, Gonsalves & Purcifull, Phytopathology 67: 965, 1977.
- Garnsey, Gonsalves & Purcifull, Phytopathology 69: 88, 1979.
- Garnsey, Christie, Derrick & Bar-Joseph, Proc. 8th Conf. int. Org. Citrus Virologists: 9, 1980.
- Garnsey, Lee & Brlansky, Phytopathology 71: 218, 1981.
- Garnsey, Gumpf, Roistacher, Civerolo, Lee, Yokomi & Bar-Joseph, Phytophylactica 19: 151, 1987.
- Gonsalves, Purcifull & Garnsey, Phytopathology 68: 553, 1978.
- Guerri, Moreno & Lee, Phytopathology 80: 692, 1990.
- Gumpf, Zheng, Moreno & Diaz, Phytophylactica 19: 159, 1987.
- Hughes & Lister, Nature, Lond. 164: 880, 1949.
- Irey, Permar & Garnsey, Proc. Fla St. hort. Soc. 101: 73, 1988.
- Knorr & Price, Rep. Fla agric. Exp. Stn No. 195, 1954.
- Lee, Proc. Fla St. hort. Soc. 97: 53, 1984.
- Lee, Timmer, Purcifull & Garnsey, Phytopathology 71: 889, 1981.
- Lee, Marais, Timmer & Graham, Pl. Dis. 68: 511, 1984.
- Lee, Garnsey, Brlansky & Goheen, Phytopathology 77: 543, 1987.
- Lee, Calvert, Nagel & Hubbard, Phytopathology 78: 1221, 1988.
- Meneghini, Biologico 12: 285, 1946.
- Miyakawa, Bull. Tokushima hort. Exp. Stn 4: 1, 1971.
- Muller & Garnsey, Proc. 9th Conf. int. Org. Citrus Virologists: 33, 1984.
- Muller, Rodriguez & Costa, Proc. 4th Conf. int. Org. Citrus Virologists: 64, 1968.
- Muller, Costa, Kitajima & Camargo, Proc. 6th Conf. int. Org. Citrus Virologists: 75, 1974.
- Norman & Grant, Proc. Fla St. hort. Soc. 69: 38, 1956.
- Oberholzer, Mathews & Stiemie, Sci. Bull. Dep. Agric. S. Afr. No. 287, 17 pp., 1949.
- Permar, Garnsey, Gumpf & Lee, Phytopathology 80: 224, 1990.
- Price, CMI/AAB Descr. Pl. Viruses No. 33, 4 pp., 1970.
- Raccah, Loebenstein & Bar-Joseph, Phytopathology 66: 1102, 1976.
- Roistacher & Bar-Joseph, Phytophylactica 19: 103, 1987a.
- Roistacher & Bar-Joseph, Phytophylactica 19: 179, 1987b.
- Rosner & Bar-Joseph, Virology 139: 189, 1984.
- Rosner, Ginzburg & Bar-Joseph, J. gen. Virol. 64: 1757, 1983.
- Rosner, Lee & Bar-Joseph, Phytopathology 76: 820, 1986.
- Sasaki, Tsuchizaki & Saito, Ann. phytopath. Soc. Jpn 44: 205, 1978.
- Schneider, Citrus Virus Diseases, Univ. Calif. Press, Berkeley, 73, 1959.
- Schneider, A. Rev. Phytopath. 11: 119, 1973.
- Stubbs, Aust. J. agric. Res. 15: 752, 1964.
- Tanaka, Shikata & Sasaki, Proc. 1st int. Citrus Symp: 1445, 1969.
- Toxopeus, J. Pomol. hort. Sci. 14: 360, 1937.
- Tsuchizaki, Sasaki & Saito, Phytopathology 68: 139, 1978.
- Vela, Cambra, Cortes, Moreno, Miquet, Perez De San Roman & Sanz, J. gen.Virol. 67: 91, 1986.
- Webber, Sci. Bull. Dep. Agric. S. Afr. No. 6, 106 pp., 1925.
- Zeman, Physis, Augsburg 19: 410, 1931.

