Details of DPV and References
DPV NO: 356 September 1998
Family: Potyviridae
Genus: Bymovirus
Species: Barley mild mosaic virus | Acronym: BaMMV
Barley mild mosaic virus
S. Kashiwazaki National Agriculture Research Center, Tsukuba, Ibaraki 305, Japan
W. Huth Federal Biological Research Center, Institute of Biochemistry and Plant Virology, Messeweg 11-12, D-38104 Braunschweig, Germany
D.-E. Lesemann Federal Biological Research Center, Institute of Biochemistry and Plant Virology, Messeweg 11-12, D-38104 Braunschweig, Germany
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Huth et al. (1984) and
Huth & Adams (1990).
Selected synonyms
- Originally called the M or Streatley isolate of
barley yellow mosaic virus,
but now known to be distinct from this virus - see Notes
-
A virus with slightly flexuous filamentous particles with a diameter of c. 12 nm and two modal lengths of 270 and 568 nm, containing two species of ssRNA. Host range is restricted to species in the Gramineae. Transmitted by the plasmodiophorid fungus Polymyxa graminis and by mechanical inoculation. Induces the formation of pinwheel inclusions in the cytoplasm of infected cells.
Main Diseases
Barley mild mosaic virus is one of two agents (the other is barley yellow mosaic virus) responsible for yellow mosaic disease of barley. Both viruses are transmitted in soil by the plasmodiophorid fungus Polymyxa graminis. The viruses occur, either separately or together, in autumn-sown barley and cause similar symptoms. Affected fields show yellow patches or appear entirely yellow, depending on the distribution of infested soil (Fig. 1). The initial symptoms are irregularly distributed chlorotic streaks of varying size on the youngest emerging leaves, often associated with upward rolling of leaf margins. The streaks are most distinct on the youngest leaves, and develop into a mosaic pattern (Fig. 2). The mosaic symptoms are occasionally associated with necrotic patches, yellow discoloration, and/or rapid death of the older leaves. The symptoms on leaves appear in early spring but tend to disappear as the weather becomes warmer. At temperatures above 20°C, new leaves are free from symptoms. The growth of infected plants is greatly diminished by temperatures in the range 5-10°C, especially when this happens under high soil moisture conditions. When such weather lasts until April, infected plants remain stunted and, although the dwarfed plants respond quickly as spring temperatures increase, yield can be lessened by up to 80% (Hill & Evans, 1980; Huth, 1988a).
Geographical Distribution
Widespread in Germany (Huth, 1990; Proeseler et al., 1991), France (Hariri et al., 1990), Great Britain (Adams, 1991) and other western European countries (Huth, 1988b). The most southerly European record is from Greece (Katis et al., 1997), the most northerly one Denmark (U. Junga, personal communication), and the most easterly one Ukraine (Fantakhun et al., 1987). Also reported from Japan (Kashiwazaki et al., 1990), China (Chen et al., 1992) and South Korea (Lee et al., 1996).
Host Range and Symptomatology
The virus has a narrow host range, limited
to the Gramineae. The only known natural host is Hordeum vulgare (barley).
The virus is transmissible by mechanical inoculation to Hordeum spp.,
Aegilops spp., Eremopyrum hirsutum, Lagurus ovatus
(Proeseler, 1988), Triticosecale (triticale)
(Kegler et al., 1985),
Secale cereale (rye)
(Ordon et al., 1992) and Triticum durum
(Proeseler, 1993), but not to Triticum aestivum (wheat) or Avena sativum
(oat)
(Ordon et al., 1992). Vector transmission of the virus has been possible
only to barley.
Diagnostic species
- Hordeum vulgare
(barley). Cultivars differ in susceptibility to infection, both by mechanical and vector inoculation, and severity of symptoms. For mechanical transmission, barley seedlings at the two- to three-leaf stage are inoculated with infective sap and then incubated at 10-17°C. The air-brush (spray-gun) method of inoculation is more efficient than leaf rubbing (Adams et al., 1986). Leaf streaks usually appear in 2-3 weeks. Symptoms in mechanically inoculated plants are similar to those in field-infected plants. For vector transmission, barley seedlings are planted in a mixture of viruliferous resting spores of Polymyxa graminis and sterilized silver sand, grown in a glasshouse at 10-15°C, and irrigated with nutrient solution as described in detail by Adams et al. (1986). The symptoms appear in 3-15 weeks.
- Secale cereale (rye). The cv. Somro shows severe systemic mosaic and
stunting of plants after mechanical inoculation
(Ordon et al., 1992).
-
Propagation species
- Hordeum vulgare
(barley). Infected leaves bearing clear mosaic symptoms are good sources of virus for purification.
Assay species
- No local lesion host is known. Barley cultivars that have high susceptibility to the virus and produce clear mosaic symptoms (e. g. Maris Otter, Igri, and New Golden) can be used for systemic assays.
Strains
Two strains from Japan (Ka1 and Na1) and one from Germany can be differentiated by their pathogenicity and symptomatology on barley cultivars; serologically, strain Ka1 and the German strain are indistinguishable, but strain Na1 is different from both (Fig. 3) (Nomura et al., 1996). Reassortment experiments with strains Ka1 and Na1 indicate that the pathogenicity and symptomatology are determined solely by RNA-1 (Kashiwazaki & Hibino, 1996).
Transmission by Vectors
Transmitted by the plasmodiophorid fungus Polymyxa graminis. Zoospores, released either from resting spores (cystosori) or from zoosporangia in barley roots, transmit the virus. The virus was detected by ELISA in extracts of dried roots heavily infected with cystosori and in zoospores liberated from the cystosori (Adams et al., 1988). Immunogold-labelled bundles of virus-like particles were observed in the cytoplasm of about 1% of zoospores liberated from plant roots and in zoospores inside zoosporangia (Chen et al., 1991). Resting spores remain viruliferous in dried roots and dried soil for several years (Huth, 1991; Chen et al., 1991).
Transmission through Seed
None reported.
Serology
Moderately immunogenic. A rabbit polyclonal antiserum with a titre of 1/1280 in ring precipitin tests has been obtained. In double diffusion tests in 1% agar containing 0.5% lithium 3,5-diiodosalicylate, the virus particles form a single precipitin band (Fig. 3) (Kashiwazaki et al., 1989). Immunosorbent electron microscopy (ISEM), ELISA and tissue print immunoassay are useful for virus detection ( Huth et al., 1984; Adams et al, 1987a; Laing & Coutts, 1988; Huth, 1997).
Relationships
Barley mild mosaic,
barley yellow mosaic,
wheat yellow mosaic,
wheat spindle streak mosaic,
rice necrosis mosaic and
oat mosaic viruses are
placed together in the genus
Bymovirus within the family
Potyviridae
on the basis of their particle morphology, transmission by Polymyxa graminis
and formation of pinwheel inclusions in infected plant cells
(Usugi et al., 1989;
Barnett, 1991;
Murphy et al., 1995). Although, among these viruses, only
barley mild mosaic and barley yellow mosaic viruses occur in barley, they differ
in biological and physical properties
(Huth, 1988a). Immunodiffusion,
complement-fixation, ELISA or immunoelectron microscopy (ISEM and decoration)
tests have failed to detect relationships between barley mild mosaic virus and the
other species in the genus Bymovirus, whereas relationships have been
detected among the other species
(Usugi et al., 1984;
Huth et al., 1984;
Adams et al., 1987b;
Huth, 1988a;
Kashiwazaki et al., 1989;
Chen & Adams, 1991).
Hybridization and restriction enzyme analyses of cDNAs and in vitro translation experiments indicate that the genomes of barley mild mosaic and barley yellow mosaic viruses have no significant sequence homology (Batista et al., 1989; Pröls et al., 1990). Sequence analyses indicate that the coat protein of barley mild mosaic virus has only 35-37% amino acid sequence identity with those of barley yellow mosaic and wheat spindle streak mosaic viruses, and 20-26% identity with those of aphid-borne potyviruses (Kashiwazaki et al., 1992; Kashiwazaki, 1996).
Stability in Sap
Sap from infected barley leaves remains infective after heating for 10 min at 45-50°C but not at 55°C, after 5 days but not 7 days at 4°C, after 2 days but not 3 days at 20-22°C, and after dilution to 1/100 but not 1/500 (S. Kashiwazaki, unpublished data).
Purification
The method for purification of
barley yellow mosaic virus
(Usugi & Saito, 1976) can also be used for barley mild mosaic virus
(Kashiwazaki et al., 1989).
Huth et al. (1984)
tested a number of modifications
of the method and adopted the following procedure.
Homogenize 100 g of fresh
or frozen leaf material (avoid necrotic tissues) in a blender with 300 ml citrate
buffer (0.1 M potassium citrate, adjusted with 0.1 M citric acid to pH 7.0), and
squeeze through cheesecloth. Add 60 ml carbon tetrachloride and stir for 15 min.
Break the emulsion by low-speed centrifugation (8000 g) for 15 min.
Collect the aqueous phase and centrifuge on to a cushion of 30% (w/v) sucrose for
2 h at 140,000 g (35,000 rev./min) in a Beckman 45Ti rotor.
Resuspend the pellets in 0.1 M citrate buffer pH 7.0 and centrifuge at low speed.
Mix the supernatant fluid with CsCl and citrate buffer to make a final CsCl concentration
of 28% (w/w), and centrifuge overnight (10°C) at 130,000 g
(35,000 rev./min) in a Beckman SW 55Ti rotor. Collect the light-scattering
virus-containing zone, dilute with citrate buffer and concentrate by centrifugation
for 2 h at 160,000 g (40,000 rev./min) in a Beckman 40 rotor.
All steps should be done as quickly as possible and the samples kept below
10°C to avoid aggregation and degradation of virus particles.
Adams et al. (1987a) modified this procedure: 2.5% Triton X-100 was added to the homogenate, and clarification with carbon tetrachloride was omitted. Partially purified virus preparation was loaded on the top of a gradient prepared with 25, 30 and 35% (w/w) CsCl solution, and centrifuged for 2 h.
Properties of Particles
A260/A280: 1.16 (not corrected for light-scattering)
(S. Kashiwazaki, unpublished data).
Buoyant density in CsCl: 1.314 g/cm3 (Huth, 1988a).
Particle Structure
Slightly flexuous filaments with a diameter of c. 12 nm and two modal lengths of 270 and 568 nm in 2% sodium phosphotungstate, pH 7 (Fig. 4). A faint central canal can be seen in phosphotungstate or ammonium molybdate (Huth et al., 1984).
Particle Composition
Nucleic acid: Two species of ssRNA, c. 5% of particle weight (estimated
from A260/A280 measurements) (S. Kashiwazaki,
unpublished data). Polyacrylamide-agarose composite gel electrophoresis under
non-denaturing conditions indicates mol. wt of 2.5-2.7 x 106 (RNA-1)
and 1.4-1.5 x 106 (RNA-2)
(Huth et al., 1984;
Kashiwazaki et al., 1993).
Protein: A single coat protein species consisting of 251 amino acids. The mol. wt of 30,900-35,000 estimated by SDS-polyacrylamide gel electrophoresis is larger than that (28,400-28,500) calculated from the sequence; the factor affecting the electrophoretic mobility is unknown (Ehlers & Paul, 1986; Kashiwazaki et al., 1992; Foulds et al., 1993; Schlichter et al., 1993). The coat protein undergoes partial proteolysis during purification of virus particles (Ehlers & Paul, 1986; Kashiwazaki et al., 1989).
Genome Properties
Bipartite, positive-sense ssRNA with poly(A) at the 3'
terminus and probably with genome-linked protein (VPg) at the 5' terminus.
There is no extensive nucleotide sequence homology between RNA-1 and RNA-2
except for the 5' non-coding regions
(Kashiwazaki, 1996). Both RNA species are
needed for infection
(Koenig & Huth, 1988). Possible double-stranded forms
of both RNA species are found in infected barley plants
(Kashiwazaki et al., 1987;
Andersen et al., 1990).
Complete nucleotide sequences have been determined for the genomes of
strain Na1 (accession numbers D83408 and D83409;
Kashiwazaki et al., 1992;
Kashiwazaki, 1996), a mechanically transmitted isolate from France
(L49381 and X82625;
Dessens et al., 1995;
Meyer & Dessens, 1996), and
fungus-transmitted
(Y10973 and X90904)
and mechanically transmitted
(Y10974
and X84802) isolates from UK
(Foulds et al., 1993;
Jacobi et al., 1995;
Peerenboom et al., 1996,
1997). Nucleotide sequences have also
been reported for the 3'-terminal regions (including the coat protein genes) of
RNA-1 of strain Ka1 (D10949;
Kashiwazaki et al., 1992), a German isolate
(X69204;
Schlichter et al., 1993), a fungus-transmitted isolate from France
(Dessens & Meyer, 1995) and a Korean isolate
(D83410;
Lee et al., 1996), and for the entire RNA-2 of a German isolate (X75933;
Timpe & Kühne, 1994). The bipartite genome of barley mild mosaic virus has the
same gene organization as that of
barley yellow mosaic virus, and the organization
of RNA-1 of these bymoviruses resembles that of the 3'-terminal three-quarters of
the monopartite genome of potyviruses
(Kashiwazaki, 1996:
Meyer & Dessens, 1996).
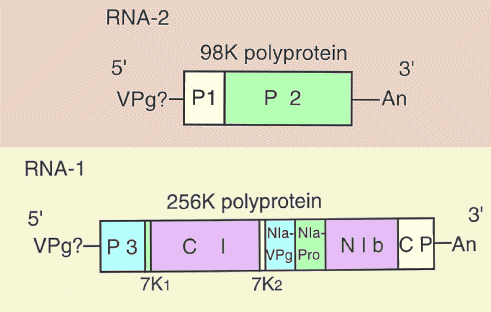
RNA-1
(Fig. 8) consists of 7261-7263 nucleotides, excluding the 3' poly(A),
and encodes a single large polyprotein of 2258 amino acids (256K). The coat
protein (CP) is mapped by amino acid sequencing at the C terminus of this
polyprotein. Amino acid sequence alignments and motif comparisons indicate
that this polyprotein corresponds to the C-terminal three-quarters of the potyvirus
polyprotein, and contains seven putative non-structural proteins, P3,
7K1, CI (cytoplasmic inclusion), 7K2, NIa
(nuclear inclusion a)-VPg, NIa-Pro (proteinase) and NIb proteins in the direction
of the N to C termini, followed by the C-terminal CP. The NIa-Pro may act as a
proteinase to catalyse cleavage of the 256K polyprotein at QA, QS or EG dipeptides
to give rise to the functional proteins
(Kashiwazaki et al., 1992;
Kashiwazaki, 1996;
Meyer & Dessens, 1996).
RNA-2 (Fig. 8) consists of 3524 nucleotides, excluding the 3' poly(A), and encodes a single polyprotein of 894 amino acids (98K) which contains two putative functional proteins of 23K (P1) and 75K (P2). P1 contains a protease domain that corresponds to the helper component proteinase of potyviruses, and probably cleaves the 98K polyprotein at a GA dipeptide. P2 shares homologies with the coat read-through protein of furoviruses, and is possibly involved in fungus transmission. After serial mechanical passages, the virus loses its fungus transmissibility, and has a shortened RNA-2 with a large deletion in the region encoding the C-terminal part of P2 (Timpe & Kühne, 1994, 1995; Dessens et al., 1995; Peerenboom et al., 1996). The P1 and P2 proteins are found in infected barley plants (Dessens & Meyer , 1995).
Relations with Cells and Tissues
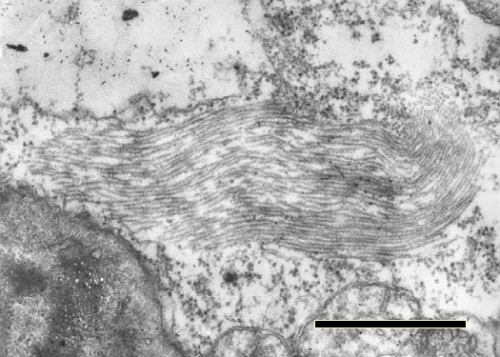
Bundles of virus-like particles are occasionally found in the cytoplasm of infected leaves (Fig. 5). Pinwheel inclusions and membrane arrays are often observed in the cytoplasm of mesophyll, vascular parenchyma and epidermal cells. In cross-section, the pinwheel inclusions show laminated aggregates but no scroll structures (Fig. 6). Vesicle accumulations develop into very dense aggregations of membranes which sometimes appear as three- dimensional, crystalline arrays of membrane materials (Fig. 7). The elements of the membrane arrays are continuous at their periphery with normal rough endoplasmic reticulum (Huth et al., 1984). Bundles of virus-like particles and pinwheel inclusions are observed also in the cytoplasm of infected barley roots (Chen et al., 1991).
Ecology and Control
Barley mild mosaic and
barley yellow mosaic viruses
have a common natural host and vector, and often co-exist in naturally infected
plants. They may be similar in their epidemiology. In nature they affect only
autumn-sown barley and mainly occur in areas where winter barley is grown
intensively. However, the possibility that spring-sown barley might also act as a
natural host of the virus in fields cannot be excluded
(Adams et al. 1986).
Within fields, viruliferous resting spores of the vector are spread when agricultural
machines move soil, an effect which is often indicated by elongated yellow patches
of affected barley
(Fig. 1). Such patches enlarge in successive years until the
whole field becomes infested. Agricultural machines almost certainly spread the
viruses and vector to adjoining fields, but other factors in long-distance spread
may include dispersal of soil particles by wind, or distribution of contaminated
waste soil from sugar factories
(Huth, 1991).
The only practical way to control the disease is to grow resistant (immune) barley cultivars. Cultivars that possess the recessive gene ym4 do not support virus replication, and vector zoospores released from their roots are virus-free. Thus, exclusive planting of these resistant cultivars may be beneficial, not only by avoiding yield losses but also by decreasing inoculum levels (Adams et al., 1987a). However, large-scale use of resistant cultivars may increase the selection pressure for resistance-breaking virus strains, as experienced with barley yellow mosaic virus in Europe (Huth, 1991).
Notes
Barley mild mosaic virus was first reported as a strain of barley yellow mosaic virus (called the M or Streatley isolate), but later was recognized on the basis of its biological, physical and serological properties to be a distinct virus (Huth et al., 1984; Huth & Adams, 1990). However, in barley cultivars susceptible to both viruses, the field symptoms caused by either one, or both together, cannot be distinguished. Cultivars that differ in susceptibility to the two viruses (e. g. Ishukushirazu, Haganemugi and Ea52; Kashiwazaki et al., 1989; Götz & Friedt, 1993; Nomura et al., 1996) can serve as differential hosts in field trials or by mechanical inoculation. Barley yellow mosaic virus may also be distinguished by its poor transmissibility by mechanical inoculation (Huth & Adams, 1990). The two viruses can easily be differentiated serologically, as antisera to these viruses show no cross-reaction in ELISA or ISEM. However, it is noteworthy that, when field-grown barley plants are doubly infected, barley yellow mosaic virus becomes more difficult to detect by ELISA later in the spring, whereas barley mild mosaic virus remains at high levels (Huth, 1988a). Both viruses can easily be distinguished from other viruses causing streak or mosaic symptoms in barley, e.g. barley stripe mosaic, soil-borne wheat mosaic, wheat streak mosaic, hordeum mosaic and brome mosaic viruses, by particle morphology, cytopathology and serology.
Figures

Yellow patches on barley fields in Germany in April. Patches elongated following the direction of machine working.

Immunodiffusion test with purified preparations of barley mild mosaic virus strains Ka1 (1 and 4), Na1 (2) and the German strain (3), barley yellow mosaic virus strains II-1 (5) and III (6), and antiserum to Ka1 (A).
References list for DPV: Barley mild mosaic virus (356)
- Adams, Pl. Path. 40: 53, 1991.
- Adams, Swaby & Macfarlane, Ann. appl. Biol. 109: 561, 1986.
- Adams, Jones & Swaby, Ann. appl. Biol. 110: 321, 1987a.
- Adams, Swaby & Jones, Pl. Path. 36: 610, 1987b.
- Adams, Swaby & Jones, Ann. appl. Biol. 112: 133, 1988.
- Andersen, Coutts & Davies, in Proc. 1st Symposium of the International Working Group on Plant Viruses with Fungal Vectors, p. 127, Stuttgart: Ulmer, 186 pp., 1990.
- Barnett, Arch. Virol. 118: 139, 1991.
- Batista, Antoniw, Swaby, Jones & Adams, Pl. Path. 38: 226, 1989.
- Chen & Adams, Pl. Path. 40: 226, 1991.
- Chen, Swaby, Adams & Ruan, Ann. appl. Biol. 118: 615, 1991.
- Chen, Adams, Zhu, Shi & Chen, Ann. appl. Biol. 121: 631, 1992.
- Dessens & Meyer, Virology 212: 383, 1995.
- Dessens, Nguyen & Meyer, Arch. Virol. 140: 325, 1995.
- Ehlers & Paul, J. Phytopath. 115: 294, 1986.
- Fantakhun, Pavlenko & Bobyi, Mikrobiologicheskiï Zh. 49: 76, 1987.
- Foulds, Lea, Sidebottom, James, Boulton, Brears, Slabas, Jack & Stratford, Virus Res. 27: 79, 1993.
- Götz & Friedt, Plant Breed. 111: 125, 1993.
- Hariri, Fouchard & Lappiere, in Proc. 1st Symposium of the International Working Group on Plant Viruses with Fungal Vectors, p. 109, Stuttgart: Ulmer, 186 pp., 1990.
- Hill & Evans, Pl. Path. 29: 197, 1980.
- Huth, in Viruses with Fungal Vectors, p. 61, Cooper & Asher, eds, Wellesbourne: Association of Applied Biologists, 335 pp., 1988a.
- Huth, NachrBl. dt. PflSchutzdienst., Stuttg. 40: 49, 1988b.
- Huth, in Proc. 1st Symposium of the International Working Group on Plant Viruses with Fungal Vectors, p. 113, Stuttgart: Ulmer, 186 pp., 1990.
- Huth, NachrBl. dt. PflSchutzdienst., Stuttg. 43: 233, 1991.
- Huth, NachrBl. dt. PflSchutzdienst., Stuttg. 49: 185, 1997.
- Huth & Adams, Intervirology 31: 38, 1990.
- Huth, Lesemann & Paul, Phytopath. Z. 111: 37, 1984.
- Jacobi, Peerenboom, Schenk, Antoniw, Steinbiss & Adams, Virus Res. 37: 99, 1995.
- Kashiwazaki, Arch. Virol. 141: 2077, 1996.
- Kashiwazaki & Hibino, J. gen. Virol. 77: 581, 1996.
- Kashiwazaki, Usugi, Minobe & Tsuchizaki, Ann. phytopath. Soc. Japan 53: 431, 1987.
- Kashiwazaki, Ogawa, Usugi, Omura & Tsuchizaki, Ann. phytopath. Soc. Japan 55: 16, 1989.
- Kashiwazaki, Nomura, Watanabe, Toshima, Iida, Usugi, Ogawa, Hibino & Tsuchizaki, in Proc. 1st Symposium of the International Working Group on Plant Viruses with Fungal Vectors, p. 105, Stuttgart: Ulmer, 186 pp., 1990.
- Kashiwazaki, Nomura, Kuroda, Ito & Hibino, J. gen. Virol. 73: 2173, 1992.
- Kashiwazaki, Nomura, Okuyama & Hibino, in Proc. 2nd Symposium of the International Working Group on Plant Viruses with Fungal Vectors, p.39, American Society of Sugar Beet Technologists, Denver, 156 pp., 1993.
- Katis, Tzavella-Klonari & Adams, Eur. J. Plant Path. 103: 281, 1997.
- Kegler, Haase, Proeseler, Kleinhempel, Richter, Schlenker & Schutzler, Arch. Phytopath. PflSchutz, Berl. 21: 247, 1985.
- Koenig & Huth, J. Phytopath. 121: 370, 1988.
- Laing & Coutts, Neth. J. Pl. Path. 94: 221, 1988.
- Lee, Kashiwazaki, Hibi & So, Ann. phytopath. Soc. Japan 62: 397, 1996.
- Meyer & Dessens, Virology 219: 268, 1996.
- Murphy, Fauquet, Bishop, Ghabrial, Jarvis, Martelli, Mayo, Summers, Virus Taxonomy. Classification and Nomenclature of Viruses. Sixth Report of the International Committee on Taxonomy of Viruses. New York: Springer, 1995.
- Nomura, Kashiwazaki, Hibino, Inoue, Nakata, Tsuzaki & Okuyama, J. Phytopath. 144: 103, 1996.
- Ordon, Huth & Friedt, J. Phytopath. 135: 84, 1992.
- Peerenboom, Jacobi, Antoniw, Schlichter, Cartwright, Steinbiss & Adams, Virus Res. 40: 149, 1996.
- Peerenboom, Cartwright, Foulds, Adams, Stratford, Rosner, Steinbiss & Antoniw, Virus Res. 50: 175, 1997.
- Proeseler, Arch. Phytopath. PflSchutz, Berl. 24: 267, 1988.
- Proeseler, J. Phytopath. 138: 262, 1993.
- Proeseler, Stanarius, Szigat & Herold, NachrBl. dt. PflSchutzdienst., Stuttg. 43: 65, 1991.
- Pröls, Davidson, Schell & Steinbiss, J. Phytopath. 130: 249, 1990.
- Schlichter, Sohn, Peerenboom, Schell & Steinbiss, Plant Cell Rep. 12: 237, 1993.
- Timpe & Kühne, Eur. J. Plant Path. 100: 233, 1994.
- Timpe & Kühne, J. gen. Virol. 76: 2619, 1995.
- Usugi & Saito, Ann. phytopath. Soc. Japan 42: 12, 1976.
- Usugi, Kuwabara & Tsuchizaki, Ann. phytopath. Soc. Japan 50: 63, 1984.
- Usugi, Kashiwazaki, Omura & Tsuchizaki, Ann. phytopath. Soc. Japan 55: 26, 1989.