Details of DPV and References
DPV NO: 359 September 1998
Family: Potyviridae
Genus: Potyvirus
Species: Sorghum mosaic virus | Acronym: SrMV
Sorghum mosaic virus
D. D. Shukla CSIRO Division of Molecular Science, Parkville, Victoria 3052, Australia
R. W. Toler Department of Plant Pathology, Texas A&M University, College Station, Texas 77843, USA
S. G. Jensen USDA, ARS, Department of Plant Pathology, University of Nebraska, Lincoln, Nebraska 68583, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Originally described by
Abbott (1961)
as a strain of
sugarcane mosaic virus
(Pirone, 1972),
but later shown
(Shukla et al., 1989b)
to be an
independent member of the genus Potyvirus
and named sorghum mosaic
virus.
Synonyms
- Sugarcane mosaic virus strains H
(Abbott & Tippett, 1966), I
(Tippett & Abbott, 1968)
and M
(Koike & Gillaspie, 1976)
-
A virus with flexuous filamentous particles c. 750 nm long containing ssRNA. It induces the formation of cytoplasmic, cylindrical (pinwheel and scrolls) and amorphous inclusions in host cells. It is transmissible mechanically and by aphids in a non-persistent manner.
Main Diseases
Sorghum (Sorghum bicolor) and sugarcane (Saccharum officinarum) are the only natural hosts. Sorghum cvs develop red-leaf (Fig. 1) and/or mosaic symptoms (Koike & Gillaspie, 1976; Giorda et al., 1986) whereas sugarcane cvs produce mild mottle (Fig. 2) (Abbott & Tippett, 1966; Tippett & Abbott, 1968; Koike & Gillaspie, 1976). Genetic resistance is available in some inbred sorghum lines (QL3-Texas, QL3- India, QL11) (Giorda et al. 1986) and in sugarcane cvs (C.P. 36-13 and C.P. 47-192) (Abbott & Tippett, 1966).
Geographical Distribution
Occurs mainly in USA in sorghum (Giorda et al., 1986) and sugarcane (Abbott 1961; Abbott & Tippett, 1966; Tippett & Abbott, 1968; Koike & Gillaspie 1976), and has been reported in sugarcane from India (Kondaiah & Nayudu, 1984), Japan (Gillaspie & Mock, 1979) and the Philippines (Dosayla & Benigno, 1980).
Host Range and Symptomatology
The natural host range is restricted to sorghum and sugarcane
(Abbott & Tippett, 1966;
Giorda et al., 1986).
Sorghum cvs develop short,
yellow-green streaks turning into reddish-brown necrotic streaks followed by
extensive reddish-brown necrosis of leaves (red-leaf symptoms)
(Fig. 1).
Some
sorghum cultivars develop only mosaic
(Koike & Gillaspie, 1976;
Giorda et al., 1986;
Tosic et al., 1990).
In sugarcane the virus causes
mild mottling
(Fig. 2),
marked stunting and subsequent excessive tillering. Mild
mottling generally disappears as the leaves age
(Abbott & Tippett, 1966;
Tippett & Abbott, 1968;
Koike & Gillaspie, 1976).
Sweet sorghum
(Sorghum vulgare), Sudangrass (S. sudanense) and
different maize cvs are susceptible. Does not infect Johnsongrass or oat
(Tippett & Abbott, 1968;
Koike & Gillaspie, 1976;
Tosic et al., 1990).
Transmission to other plant species in the family Gramineae has not
been attempted.
-
Diagnostic species
- Sorghum bicolor (sorghum). Cvs Atlas, NM 30, R 430, SA 8735,
Trudan-7 and Trudex develop typical red-leaf symptoms whereas cvs Aunis,
BTX398 and OKY8 develop systemic mosaic symptoms.
- Zea mays (maize). Different maize cultivars are readily infected and show systemic mosaic symptoms.
- Saccharum officinarum (sugarcane). Some differential cvs (C.P. 31- 294, C.P. 31-588, C.P. 55-30, C.P. 62-264) produce diagnostic symptoms such as mild mottle, chlorosis, necrosis and severe stunting (Abbott & Tippett, 1966; Tippett & Abbott, 1968; Koike & Gillaspie, 1976).
- Zea mays (maize). Different maize cultivars are readily infected and show systemic mosaic symptoms.
-
Propagation species
- Sorghum bicolor cvs which react with mosaic symptoms (e.g. Aunis,
BTX 398, OKY8, and Trudan-5) are suitable for maintenance and virus
purification.
- Zea mays is a good propagation host for virus purification.
- Saccharum officinarum cvs showing mild mottle symptoms (e.g. C.P. 31-588) are good propagation hosts.
- Zea mays is a good propagation host for virus purification.
Strains
Three strains of sorghum mosaic virus (previously known as strains of sugarcane mosaic virus; Pirone, 1972), namely H (Abbott, 1961), I (Tippett & Abbott, 1968) and M (Koike & Gillaspie, 1976), have been identified (Shukla et al., 1989b). The strains are differentiated on the basis of their reactions on differential sugarcane and sorghum cvs. Strain I induces severe chlorosis and necrosis in sugarcane cv. C.P.31-294 whereas H produces only mild mottle on this cultivar. Sorghum cv. Rio shows pronounced chlorosis and necrosis after infection with strain I whereas strain H induces only mild mosaic symptoms on this cultivar (Tippett & Abbott, 1968). Strain M differs from strains H and I in inducing more severe red-leaf symptoms on sorghum cv. Rio. No serological differences have been observed between strains H, I and M (Jarjees & Uyemoto, 1984; Giorda et al., 1986; Shukla et al., 1989b).
Transmission by Vectors
The aphid Dactynotus ambrosiae, common in sugarcane fields in USA, transmitted strains I and M from and to sorghum cv. Rio with frequencies of 48 and 28%, respectively (Koike & Gillaspie, 1976). In these tests the aphids were given an acquisition feeding time of 16 h and an inoculation feeding time of 24 h. However, like other members of the genus Potyvirus, sorghum mosaic virus should have a non-persistent type of virus-vector relationship.
Transmission through Seed
Not detected in 495 sorghum seedlings grown from seed collected from plants of cv. Rio infected with strain M (Koike & Gillaspie, 1976).
Serology
Antisera with homologous titres of 1/32-1/256 have been produced by injecting rabbits with purified preparations of particles (Snazelle et al., 1971; Derrick, 1975; Koike & Gillaspie, 1976; Jarjees & Uyemoto, 1984; Giorda et al., 1986). Antisera can be used in microprecipitation tests (Snazelle et al., 1971; Koike & Gillaspie, 1976), sodium dodecyl sulphate-agar gel immunodiffusion tests (Giorda et al., 1986), ELISA (Jarjees & Uyemoto, 1984), immunosorbent electron microscopy (Derrick, 1975; Giorda et al., 1986) and Western blotting (Shukla et al., 1989b).
Relationships
Sorghum mosaic virus is placed in the genus Potyvirus of the family Potyviridae because of its filamentous particles c. 750 nm long, its induction of pinwheel inclusions in cells and its mechanical and aphid transmissibility. On the basis of similar biological properties and an apparently distant serological relationship, strains H, I and M were considered to be strains of sugarcane mosaic virus (Abbott & Tippett, 1966; Tippett & Abbott, 1968; Snazelle et al., 1971; Pirone, 1972; Derrick, 1975; Koike & Gillaspie, 1976; Jarjees & Uyemoto, 1984). However, later tests by Western blotting with polyclonal antibodies directed to the virus-specific N-termini of the coat proteins have shown that the former sugarcane mosaic virus strains H, I and M are unrelated to US strains of sugarcane mosaic virus A, B, D and E, maize dwarf mosaic virus strains A, B and O, and Australian sugarcane mosaic virus strains JG, SC, BC and Sabi (Shukla et al., 1989b). The distant relationships observed and reported previously are now known to be due to antibodies directed to the conserved core regions (devoid of N and C termini) of the coat proteins which show high sequence homology throughout the Potyviridae (Shukla et al., 1988b, 1989a, 1989b, 1989c).
Stability in Sap
The H strain has a thermal inactivation point between 49°C and 51°C (Abbott & Tippett, 1966).
Purification
1. (Snazelle et al., 1971).
Homogenize infected tissue with 0.017
M ascorbic acid containing 0.14 M 2-mercaptoethanol and 0.01 M sodium
diethyldithiocarbamate (1.5-2 ml/g tissue). Clarify by emulsifying with cold
chloroform (1 ml/2 g tissue) followed by low-speed centrifugation. Pellet virus by
ultracentrifuging 2 h at 28,000 rev/min. Resuspend pellet in 0.1 M borate buffer,
pH 8.2. Clarify by low-speed centrifugation and further purify by rate-zonal,
sucrose density-gradient centrifugation 2.5 h at 23,000 rev/min.
2. (Jarjees & Uyemoto, 1984).
Homogenize infected tissue with 0.1
M sodium citrate buffer, pH 7.5 containing 0.5% 2-mercapoethanol. Filter
through cheesecloth. Emulsify with 0.5 vol. of chloroform followed by low-speed
centrifugation. Centrifuge the buffer phase 60 min at 30,000 rev/min.
Resuspend the pellet in 0.005 M borate buffer pH 8.6, and give a second low-
and high-speed centrifugation. Resuspend the pellet in 0.0125 M borate buffer,
pH 8.6, containing 0.3 M NaCl and 40% sucrose and purify further by sucrose
gradient zone electrophoresis.
3. (Giorda et al., 1986, modified from Gough & Shukla, 1981). Homogenize infected tissue in 0.5 M sodium borate buffer, pH 8.0, containing 0.15% thioglycollic acid and 0.01 M ethylenediamine-tetraacetic acid (1.5 ml/1 g tissue). Emulsify with 1:4 vol. each of chloroform and carbon tetrachloride followed by low-speed centrifugation. Filter the supernatant fluid through glass wool. Add Triton X-100 5% and 0.5 M deionized urea and centrifuge at low speed. Pellet virus by centrifuging 2 h at 28,000 rev/min. Resuspend the pellet in 0.05 M borate buffer, pH 8.0, containing 0.01 M ethylenediaminete-tetraacetic acid and 0.5 M deionized urea followed by low-speed centrifugation. Purify further by sucrose density gradient (10% to 40% in the resuspension buffer) centrifugation 2 h at 25,000 rev/min.
Particle Structure
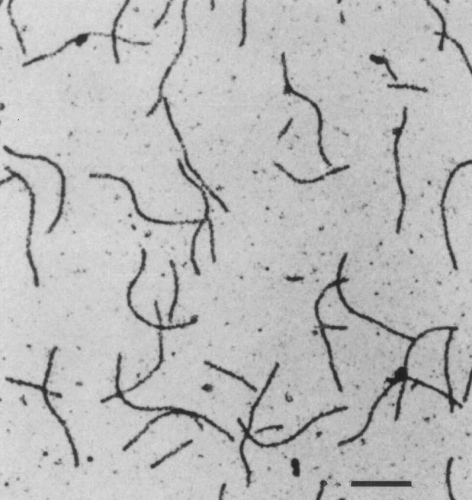
Particles are flexuous filaments c. 750 nm long (Fig. 3) (Koike & Gillaspie, 1976).
Particle Composition
Nucleic acid: A single ssRNA
(Jilka, 1990).
Protein: A single polypeptide species of 36,850 daltons consisting of 329 amino acid residues (Jilka, 1990). Amino acid sequence is given by Jilka (1990) and Yang & Mirkov (1997).
Genome Properties
(Jilka, 1990). The segment of the genome encoding the viral coat protein is adjacent to the 3' non-coding region. The nuclear inclusion protein gene is located upstream from the coat protein gene. The 3' non-coding region is 238 nucleotides long and is polyadenylated.
Relations with Cells and Tissues
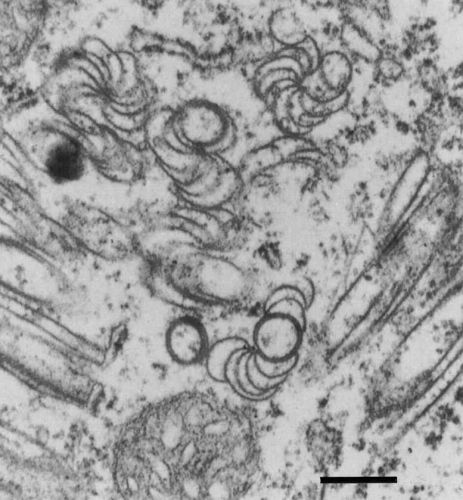
Cytoplasmic inclusions of ‘pinwheel’ and ‘scroll’ types, characteristic of Edwardson’s (1974) subdivision I of the potyvirus group, have been found in cells of Sorghum bicolor (Fig. 4) (Hearon et al., 1981, Giorda et al., 1986) and Zea mays (Lesemann et al., 1992) after infection with H and I strains. As with many other potyviruses, these strains of SrMV induce the formation of large clusters of small vesicles which accumulate together with cytoplasmic inclusions to form large complexes. The virus also induces the formation of amorphous inclusions and cytoplasmic crystals in Zea mays (Lesemann et al., 1992).
Notes
Recent investigation on the taxonomy of aphid-borne potyviruses (genus Potyvirus) infecting species of Gramineae (Shukla et al., 1989b; 1992) has shown that virus isolates previously included as strains of sugarcane mosaic virus in fact comprise four distinct potyviruses, namely sugarcane mosaic virus (USA strains A, B, D and E, Australian strains SC, BC and Sabi, and USA maize dwarf mosaic virus strain B; Teakle et al., 1989), Johnsongrass mosaic virus (Australian sugarcane mosaic virus strain JG, USA maize dwarf mosaic virus strains O and Kansas 1; Shukla & Teakle, 1989; McKern et al., 1990), maize dwarf mosaic virus (USA strains A, D, E and F; Ford et al., 1989), and sorghum mosaic virus (USA sugarcane mosaic virus strains H, I and M; Shukla et al., 1989b). The four viruses induce similar symptoms in some hosts, have host ranges usually restricted to Gramineae and have some aphid vectors in common. However, they are serologically unrelated or only distantly related (Snazelle et al., 1971; Jarjees & Uyemoto, 1984; Shukla & Gough, 1984; McDaniel & Gordon 1985; Giorda et al., 1986; Hewish et al., 1986; Shukla et al., 1983; 1988b; 1989a, 1989b, 1989c). Previous reports of serological relationships among these viruses and their strains are caused by the presence in polyclonal antisera of antibodies to epitopes in conserved regions of potyvirus coat proteins; these antibodies recognise most potyviruses (Shukla et al., 1988b; 1989a, 1989c). The four viruses also differ considerably in coat protein amino acid composition (Hill et al., 1973; Gough & Shukla, 1981; Von Baumgarten & Ford, 1981) and sequence (Shukla et al., 1987; Jilka, 1990; Frenkel et al., 1991; Yang & Mirkov, 1997) and can be distinguished readily by high performance liquid chromatography of coat protein digests (Shukla et al., 1988a, McKern et al., 1990; 1991) and by morphology and serology of their cytoplasmic inclusions (Hearon et al., 1981; Jensen & Staudinger, 1989; Lesemann et al., 1992). The four viruses can also be distinguished by their reactions on certain inbred sorghum lines (Teakle & Grylls, 1973; Persley et al., 1985; Giorda et al., 1986; Tosic et al., 1990). Strains of sugarcane mosaic virus induce necrosis of new leaves of cv. Atlas sorghum whereas sorghum mosaic virus induces typical red-leaf symptoms, and the other two viruses induce only mosaic symptoms. Maize dwarf mosaic virus induces mosaic and necrotic spots in systemically infected leaves of sorghum cv. Rio whereas the other three viruses induce either mosaic or symptomless infection. Johnsongrass mosaic virus induces necrotic red stripes in sorghum lines OKY8 and SA8735 whereas the other three viruses induce only mosaic symptoms.
Figures

Sorghum bicolor cv. Atlas infected by the H strain of sorghum mosaic virus showing typical red-leaf symptoms.

A leaf of sugarcane cv. CP 31-588 infected by the I strain of sorghum mosaic virus showing mild mottling. (Courtesy Dr A. G. Gillaspie.)
References list for DPV: Sorghum mosaic virus (359)
- Abbott, Phytopathology 51: 642, 1961.
- Abbott & Tippett, Tech. Bull. US Dep. Agric. No.1340, 25 pp.,1966.
- Derrick, Proc. Am. phytopath. Soc. 2: 42, 1975.
- Dosayla & Benigno, Proc. Int. Soc. Sugarcane Technol. 17: 1510, 1980.
- Edwardson, Monogr. Ser. Fla Agric. Exp. Stn No. 4, 398 pp, 1974.
- Ford, Tosic & Shukla, AAB Descr. Pl. Viruses 341, 5 pp., 1989.
- Frenkel, Jilka, McKern, Strike, Clarke, Shukla & Ward. J. gen. Virol. 72: 237, 1991.
- Gillaspie & Mock, Sugarcane Pathol. Newsl. 22: 21, 1979.
- Giorda, Toler & Miller, Pl. Dis. 70: 624, 1986.
- Gough & Shukla, Virology 111: 455, 1981.
- Hearon, Gillaspie & Mock, Phytopathology 71: 879, 1981.
- Hewish, Shukla & Gough, J. virol. Meth. 13: 79, 1986.
- Hill, Ford & Benner, J. gen. Virol. 20: 327, 1973.
- Jarjees & Uyemoto, Ann. appl. Biol. 104: 497, 1984.
- Jensen & Staudinger, Phytopathology 79: 1215, 1989.
- Jilka, Cloning and characterization of the 3' terminal regions of RNA from select strains of maize dwarf mosaic virus and sugarcane mosaic virus. Doctoral Thesis, Univ. Illinois, Urbana, 160 pp., 1990.
- Koike & Gillaspie, Pl. Dis. Reptr 60: 50, 1976.
- Kondaiah & Nayudu, Curr. Sci. 53: 273, 1984.
- Lesemann, Shukla, Tosic & Huth, in Potyvirus Taxonomy, ed. O.W. Barnett, Arch. Virol., Suppl 5: 353, 1992.
- McDaniel & Gordon, Pl. Dis. 69: 602, 1985.
- McKern, Whittaker, Strike, Ford, Jensen & Shukla, Phytopathology 80: 907, 1990.
- McKern, Shukla, Toler, Jensen, Tosic, Ford, Leon & Ward, Phytopathology 81: 1025, 1991.
- Persley, Henzell, Greber & Teakle, Pl. Dis. 69: 1046, 1985.
- Pirone, CMI/AAB Descr. Pl. Viruses 88, 4 pp., 1972.
- Shukla & Gough, Pl. Dis. 68: 204, 1984.
- Shukla & Teakle, AAB Descr. Pl. Viruses 340, 5 pp., 1989.
- Shukla, O'Donnell & Gough, Acta phytopath. Acad. Sci. Hung. 18: 79, 1983.
- Shukla, Gough & Ward, Arch. Virol. 96: 59, 1987.
- Shukla, McKern, Gough, Tracy & Letho, J. gen. Virol. 69: 493, 1988a.
- Shukla, Strike, Tracy, Gough & Ward, J. gen. Virol. 69: 1497, 1988b.
- Shukla, Jilka, Tosic & Ford, J. gen. Virol. 70: 13, 1989a.
- Shukla, Tosic, Jilka, Ford, Toler & Langham, Phytopathology 79: 223, 1989b.
- Shukla, Tribbick, Mason, Hewish, Geysen & Ward, Proc. natl Acad. Sci. USA 86: 8192, 1989c.
- Shukla, Frenkel, McKern, Ward, Jilka, Tosic & Ward, in Potyvirus Taxonomy, ed. O. W. Barnett, Arch. Virol. Suppl 5: 363, 1992.
- Snazelle, Bancroft & Ullstrup, Phytopathology 61: 1059, 1971.
- Teakle & Grylls, Aust. J. agric. Res. 24: 465, 1973.
- Teakle, Shukla & Ford, AAB Descr. Pl. Viruses 342, 5 pp., 1989.
- Tippet & Abbott, Pl. Dis. Reptr 52: 449, 1968.
- Tosic, Ford, Shukla & Jilka, Pl. Dis. 74: 549, 1990.
- Von Baumgarten & Ford, Phytopathology 71: 36, 1981.
- Yang & Mirkov, Phytopathology 87: 932, 1997.