Details of DPV and References
DPV NO: 365 September 1998
Family: Pospiviroidae
Genus: Apscaviroid
Species: Pear blister canker viroid | Acronym: PBCVd
Pear blister canker viroid
R. Flores Instituto de Biología Molecular y Celular de Plantas (UPV-CSIC), Universidad Politécnica de Valencia, Camino de Vera 14, 46022 Valencia, Spain
C. Hernández Instituto de Biología Molecular y Celular de Plantas (UPV-CSIC), Universidad Politécnica de Valencia, Camino de Vera 14, 46022 Valencia, Spain
S. Ambrós Instituto de Biología Molecular y Celular de Plantas (UPV-CSIC), Universidad Politécnica de Valencia, Camino de Vera 14, 46022 Valencia, Spain
G. Llácer Departamento de Citricultura y Otros Frutales, Instituto Valenciano de Investigaciones Agrarias, 46113 Moncada, Valencia, Spain
J. C. Desvignes Centre Technique Interprofessionel des Fruits et Légumes, Centre de Lanxade, BP 21, F-24130 Prigonrieux, France.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Diseases consisting of bark disorders on certain sensitive pear cultivars have
been variously described in Europe and the USA as pear blister canker
(Cropley, 1960),
pear rough bark
(Kristensen & Jorgensen, 1957;
Thomsen, 1961),
pear bark split and bark necrosis
(Kegler, 1965,
1967),
and pear bark measles
(Cordy & MacSwan, 1961).
Although the possibility was considered that distinct strains of the
same infectious agent may induce these bark alterations
(Lemoine, 1986),
the term pear blister canker (PBC) is now reserved for the
disease that appears in the pear indicator A 20
(Ambrós et al., 1995a).
The viroid nature of the causal agent of PBC disease was suggested by
Flores et al. (1991)
and
Hernández et al. (1992a,
1992b),
and
demonstrated by
Ambrós et al. (1995b).
A viroid with a circular single-stranded RNA molecule of 315-316 nt. Transmitted by graft propagation and mechanically. Recorded in Europe.
Main Diseases
Symptomlessly infects most commercial pear (Pyrus communis) varieties such as Williams, Comice and Beurré Hardy (Ambrós et al., 1995a; Desvignes, unpublished data). Approximately 10% of the old pear cultivars in France are symptomlessly infected (J. C. Desvignes, unpublished data). Of the recognized virus indicators, only the pear indicator A 20, selected in England as a pear vein yellows indicator, is sensitive enough to develop the bark canker (Desvignes, 1970) characteristic of PBCVd infection. These symptoms, that occur solely in the bark, usually appear during the second year of infection as pustules or superficial cracks on the epidermis that turn progressively into scattered cankers and scaly bark or deep bark splits (Fig. 1); plants die within 5-8 years. Leaves and fruits do not display pathological alterations.
Geographical Distribution
Recorded in Europe (France, Spain and Italy) (Desvignes, 1970; Hernández et al., 1992b; Ambrós et al., 1995a; Loreti et al., 1997).
Host Range and Symptomatology
Pear (Pyrus communis) and quince (Cydonia oblonga) are
the only known natural hosts. Quince layers, often used as pear rootstocks in
Europe, are probably a reservoir of the viroid. PBCVd can be mechanically
transmitted experimentally to cucumber (Cucumis sativus) in which it
induces mild leaf rugosity or no symptoms
(Flores et al., 1991).
It also
infects symptomlessly species of Pyrus, Chaenomeles,
Cydonia and Sorbus. It is not transmitted to species of
Malus, Amelanchier, Aronia, Cotoneaster,
Crataegus and Pyracantha (J. C. Desvignes, unpublished data).
Transmitted by grafting, budding and experimentally by razor-slashing.
Diagnostic and assay species
-
Pyrus communis (pear) clone A 20. Pear seedlings or quince layers are
doubly grafted with the indicator clone A 20 and with the pear material to be
tested in upper and lower positions, respectively. Under field conditions the
characteristic symptoms of the disease appear in the bark of A 20 approximately
2 years after inoculation. More recently, an analysis of the sensitivity to PBCVd
of a large number of pear seedlings (Fieudière) has selected two indicator
clones, Fieud 37 and Fieud 110 that, under greenhouse conditions, develop
cankers in leaves and young shoots in only 3-4 months
(Fig. 2)
and then die
rapidly (J. C. Desvignes, unpublished data).
PBCVd can also be detected by molecular methods including polyacrylamide gel electrophoresis (Flores et al., 1991) and dot-blot hybridization with radioactive and non-radioactive probes (Ambrós et al., 1995a). It should be also possible to detect PBCVd by RT-PCR using primers derived from the reference sequence (Hernández et al., 1992a), although no reports using this approach have been published. Molecular hybridization has detected PBCVd in symptomless plants of the pear cv. Williams (Ambrós et al. 1995a) and cvs Abbé Fetel, Beurré Bosc, Beurré Diel, Beurré Giffard, Beurré Hardy, Comice, Conférence, Delbias, Dr. Jules Guyot, Harrow Sweet, Highland, Louise Bonne, Pierre Corneille, Président Héron and Sérenade (J. C. Desvignes, unpublished data).
Propagation species
- Pyrus communis is the only host from which the viroid has been purified (Flores et al., 1991).
Strains
Symptoms induced on the pear indicators A 20 and Fieud 37 are similar and the severity of the symptoms depends on the isolate used. For three PBCVd isolates that have been cloned, symptoms increased in severity using isolates P1914T, P2098T and P47A respectively. The nucleotide sequence changes found between these isolates (Ambrós et al., 1995b) may be the cause of these differences in symptom severity.
Transmission by Vectors
No information.
Transmission through Seed
Grafts of the pear indicator A 20 onto 200 pear seedlings obtained from a source originally infected with the reference isolate, P2098T, failed to show any symptoms 4 years after inoculation, suggesting that PBCVd is not transmitted through seed (J. C. Desvignes, unpublished data).
Relationships
PBCVd contains the central region which is conserved in members of the genus Apscaviroid, as well as a terminal conserved region located in the left- terminal domain of apscaviroids and other viroid genera (Fig. 3) (Hernández, et al., 1992a; Flores et al., 1997).
Stability in Sap
No information.
Purification
PBCVd can be purified from infected pear leaves by extracting tissue in buffer- saturated phenol and partially purifying the nucleic acids using chromatography on non-ionic cellulose (CF-11) (Flores et al., 1991). Polysaccharides are removed by treatment with 2-methoxyethanol and the nucleic acids recovered subsequently by precipitation with cetyltrimethylammonium bromide. Viroid circular molecules are obtained by two consecutive steps of polyacrylamide gel electrophoresis, the first under non-denaturing conditions and the second under denaturing conditions, and the viroid eluted from the gel.
Properties of Infective Nucleic Acid
A covalently closed single-stranded RNA. The reference sequence from isolate P2098T has 315 residues with a base composition (G:C:A:U) of (31.4:29.2:17.1:22.2). It adopts a quasi-rod-like conformation (Fig. 3) in the proposed secondary structure of minimum free energy (Hernández, et al., 1992a). Variants from isolates P1914T, P47A and an Italian isolate, have 315-316 residues and minor sequence changes (Ambrós et al., 1995b; Loreti et al., 1997).
Relations with Cells and Tissues
PBCVd can be isolated from infected pear leaves and stem bark with yields of 0.5 to 1 µg of purified RNA per 100 g of fresh tissue (Flores et al., 1991).
Ecology and Control
Control is possible only through the use of viroid-free propagating material. Attempts to eliminate the agent by thermotherapy have not been successful (J. C. Desvignes, unpublished data).
Figures

Bark symptoms induced by PBCVd (isolate P2098T) approximately 3 years after inoculation to the pear indicator A 20 grafted on quince and grown under field conditions.

Symptoms induced by PBCVd (isolate P2098T) 3-4 months after inoculation on the pear indicator Fieud 37, grown under greenhouse conditions. Similar symptoms are observed in the pear indicator Fieud 110.
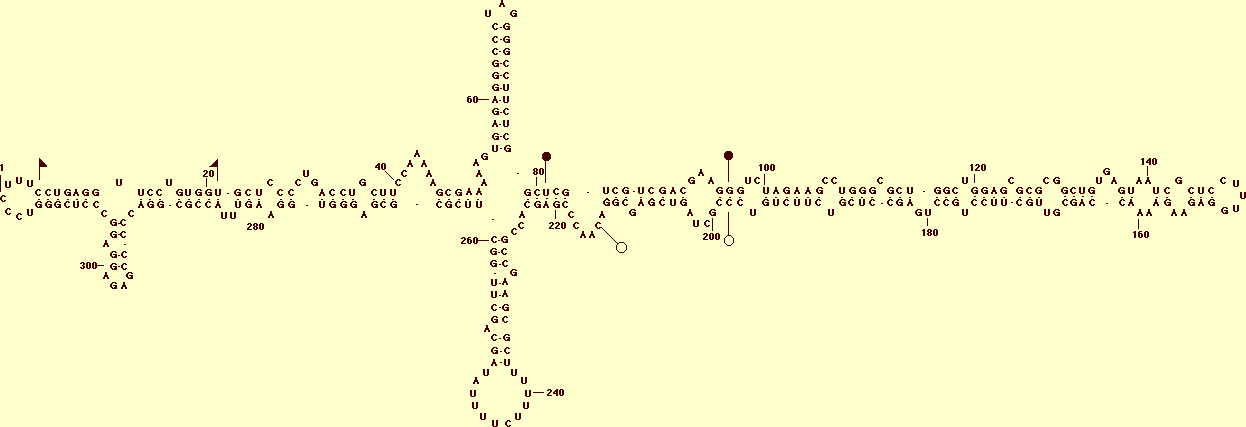
Primary and proposed secondary structure of the lowest free energy for the reference sequence from PBCVd isolate P2098T (D12823). The upper and lower strands of the central conserved region (CCR) typical of apscaviroids are delimited by filled and open circles respectively, and the terminal conserved region (TCR) by flags (from Hernández et al., 1992a).
References list for DPV: Pear blister canker viroid (365)
- Ambrós, Desvignes, Llácer & Flores, Acta Hort. 386: 515, 1995a.
- Ambrós, Desvignes, Llácer & Flores, J. gen. Virol. 76: 2625, 1995b.
- Cordy & MacSwan, Pl. Dis. Reptr 45: 891, 1961.
- Cropley, Rep. E. Malling Res. Stn for 1959: 104, 1960.
- Desvignes, CTIFL Docum. 26: 12, 1970.
- Flores, Hernández, Llácer, & Desvignes, J. gen. Virol. 72: 1199,1991.
- Flores, Di Serio, & Hernández, Semin. Virol. 8: 65, 1997.
- Hernández, Elena, Moya & Flores, J. gen. Virol. 73: 2503, 1992a.
- Hernández, Llácer, Desvignes, & Flores, Acta Hort. 309: 319, 1992b.
- Kegler, Zast. Bilja 16: 311, 1965.
- Kegler, Obstbau 7: 21, 1967.
- Kristensen & Jorgensen, Tidsskr. Planteavl 61: 617, 1957.
- Lemoine, Acta Hort. 193: 251, 1986.
- Loreti, Faggioli & Barba, J. Phytopath. 145: 541, 1997.
- Thomsen, Tidsskr. Planteavl 65: 69, 1961.