Details of DPV and References
DPV NO: 368 August 1999
Family: Geminiviridae
Genus: Begomovirus
Species: Tomato yellow leaf curl virus | Acronym: TYLCV
Tomato yellow leaf curl virus-Israel
Henryk Czosnek Department of Field Crops and Genetics, and the Otto Warburg Center for Biotechnology in Agriculture, Faculty of Agricultural, Food and Environmental Quality Sciences, The Hebrew University of Jerusalem, Rehovot 76100, P.O.Box 12, Israel
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease first reported in Israel in 1939-40 associated with outbreaks of Bemisia tabaci. The causal agent was described in 1964 and named tomato yellow leaf curl virus (TYLCV) (Cohen and Harpaz, 1964). The virus was isolated in 1988 (Czosnek et al., 1988) and its genome sequenced in 1991 (Navot et al., 1991). The virus isolate from Israel is denoted as TYLCV-Is.
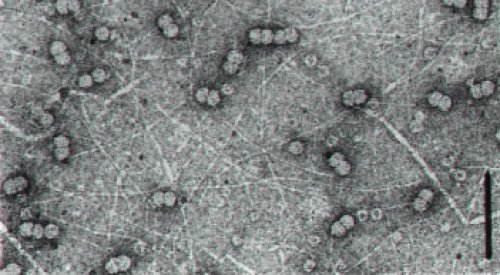
A virus exclusively transmitted by the whitefly Bemisia tabaci (synonym B. argentifolii) in natural conditions (Cohen and Nitzany, 1966) (Fig. 1). Geminate particle approximately 20x30 nm in size (Fig. 2) encapsidating a single molecule of covalently closed circular ssDNA of 2787 nucleotides (Navot et al., 1991). The virus is not sap transmissible and is not transmissible through seeds. It infects a wide range of wild (weeds, wild tomato species) and domesticated plants (bean, lisianthus, petunia, tomato). TYLCV-Is is present in the Middle East, Spain, the Caribbean Islands and Florida. Other TYLCV strains are found in Southwest Europe, Northern and tropical Africa and Southeast Asia. If cultures are not protected in the affected regions by using insecticides or nets, losses may reach 100% of the yield.
Main Diseases
The virus induces the yellow leaf curl disease in tomato. Infected plants are severely stunted. The leaves are small, curled and chlorotic. Those leaflets that appear soon after infection are cupped down and inwards. Subsequently developing leaves are chlorotic and show an upward curling of the leaflet margins (Fig. 3). Seedlings infected during the first month after planting are arrested in their growth and do not set fruits (Cohen & Harpaz, 1964).
Infection of common bean (Phaseolus vulgaris) was first observed in Israel in 1989 associated with massive whitefly populations. Symptoms consist of severe distortion and crumpling of the leaves (Fig. 4), arrest of growth and decrease in yield. The disease could be transmitted to tomato by whiteflies. Molecular analyses by hybridization and PCR indicated that TYLCV was the causal agent (Navot et al., 1992). The bean disease caused by TYLCV was also identified in Southern Spain (Navas-Castillo et al., 1999).
TYLCV also infects flowers in commercial nurseries in Israel. Infected petunia (Czosnek et al., unpublished) and lisianthus (Cohen et al., 1995) present distorted yellowing leaflets, distortion of the growing tips, and swelling of veins on the lower surface of the leaves. Plants infected at an early stage do not yield flowers. In both cases, the virus was identified using DNA probes and antibodies, and by whitefly-mediated transmission.
Geographical Distribution
The name tomato yellow leaf curl virus (TYLCV) was coined in the early 1960's to describe a virus transmitted by the whitefly Bemisia tabaci that affected tomato cultures in Israel (Cohen & Harpaz, 1964). TYLCV outbreaks that were sporadic in the 1960's have become a serious economic problem, starting in the early 1970's, when losses often reached 100 % of the yield. By the end of the 1970's, all regions of tomato culture in the Middle East were affected by the virus (Czosnek & Laterrot, 1997). The Israeli strain of TYLCV (TYLCV-Is), as characterized by its DNA sequence (Navot et al., 1991), is also present in Egypt (Nakhla et al., 1993). It is probably also present in Lebanon (Makkouk et al., 1979), Jordan (Makkouk, 1978) and Cyprus (Ioannou, 1985), although this has not been confirmed by sequencing. In Western Europe, TYLCV-Is has been reported in Spain (Navas-Castillo et al., 1999), coexisting with TYLCV-Sardinia. TYLCV-Is was identified in the Caribbean Islands (Cuba, Martinez Zubiaur et al., 1996; Dominican Republic, Nakhla et al., 1994; Jamaica, Wernecke et al., 1995) and in Florida (Polston, 1998). The TYLCV isolates in the New World, were more than 95% homologous to TYLCV-Is, indicating a recent importation.
Host Range and Symptomatology
The virus cannot be mechanically transmitted from tomato to tomato (Cohen & Harpaz, 1964). Besides the domesticated tomato Lycopersicon esculentum, several cultivated plants are hosts of TYLCV-Is and present severe symptoms upon whitefly-mediated inoculation: bean (Phaseolus vulgaris), petunia (Petunia hybrida), and lisianthus (Eustoma grandiflorum). Most of the wild tomato species, such as L. chilense, L. hirsutum, L. peruvianum and L. pimpinellifolium, are symptomless carriers (Zakay et al., 1991). Weeds, such as Datura stramonium and Cynanchum acutum, present distinct symptoms while others, such as Malva parviflora, are symptomless carriers. Plants used to rear whiteflies, such as cotton (Gossypium hirsutum) and eggplant (Solanum melongena), are immune to the virus (Czosnek et al., 1993). Some plants, such as Nicotiana benthamiana and N. tabacum, which cannot be inoculated by whiteflies, may be infected by agroinoculation or by bombardment with an infectious TYLCV DNA clone (Czosnek et al., unpublished).
In tomato, first symptoms appear about 2 weeks after inoculation of young susceptible plants. Leaflets are small, cupped inward and with yellow margins (Fig. 3). There is severe stunting of growth, the shoots are erect and the plant becomes bushy (Cohen & Harpaz, 1964). Seedlings infected immediately after planting will stop growing and will not bear fruits. Losses in tomato crops routinely reach 100% if the plant is not protected from the insect vector by using insecticides and insect-proof nets. Susceptible tomato (L. esculentum) and datura (D. stramonium) plants have been used as diagnostic species for whitefly transmission of TYLCV from symptomless carrier plants (Cohen & Nitzany, 1966).
Strains
An infectious TYLCV DNA (TYLCV-Is) was cloned from field-infected tomato plants collected in 1987 (Navot et al., 1991). Another TYLCV isolate (TYLCV-Is/Mild) maintained in D. stramonium plants from the early 1960's has also been cloned. Agroinoculation of this cloned DNA produced mild symptoms in tomato (Antignus & Cohen, 1994).
Transmission by Vectors
The whitefly Bemisia tabaci is the only known natural vector. Adults and crawlers (1st instar) are the only stages where the insect is able to acquire and transmit TYLCV (Cohen and Nitzany, 1966; Mehta et al., 1994). Parameters of acquisition and transmission of virus by adults have been studied in depth (Cohen and Nitzany, 1966; Zeidan and Czosnek, 1991; Mansour & Al-Musa, 1992; Mehta et al., 1994, Atzmon et al., 1998). Single insects are able to acquire TYLCV and transmit it to tomato plants. Minimum effective acquisition access and inoculation access periods are approximately 10-20 min. The rate of transmission increases with longer acquisition and inoculation access periods. A minimum of 8 h (latent period) from the time acquisition started is required for B. tabaci to be able to infect tomato test plants. In a one insect/one plant inoculation test, female B. tabaci are more efficient (~95%) than male insects (~25%). Viral DNA can be detected in single insects by PCR after 5 min of access feeding, and in tomato plants as early as 5 min after inoculation feeding (Atzmon et al., 1998). A GroEL homologue produced by the insect coccoid endosymbionts is involved in circulative transmission of the virus (Morin et al., 1999).
TYLCV is associated with the insect vector for its entire adult life. Insects that emerged during a 24-h period and were reared on a non-host plant after a 24 h-acquisition period retained TYLCV for their entire 35-40 day life (Rubinstein & Czosnek, 1997). During this period, transmission rates decreased from 100% to 15%. The viral DNA was detected during the entire life of the insect whereas the capsid protein was undetectable after 12 days. The long-term association of TYLCV with the insect led to a reduction of ~20% in their life expectancy and of ~50% in the number of eggs laid (Rubinstein & Czosnek, 1997). TYLCV can be transmitted through the egg for at least two generations (Ghanim et al., 1998).
Transmission through Seed
The virus is not transmitted through seed (Nitzany, 1975) or through soil (Makkouk, 1978).
Serology
Western blots were used to detect TYLCV-Is in leaf extracts of infected tomato plants (Michelson et al., 1994), using polyclonal antibodies raised against TYLCV-Sar virions (from B. Gronenborn). Similarly the monoclonal antibody SCR18 (Macintosh et al., 1992) was used to detect the virus in plant extracts (Morin et al., 1999).
Relationships
TYLCV-Is is part of a complex of distinct whitefly-transmitted geminiviruses infecting tomato, which includes isolates from Southwest Europe (Italy: Sardinia and Sicily; Spain), the Middle East (Iran, Saudi Arabia, Yemen) and Southeast Asia (Thailand). All TYLCV isolates contain a single DNA-A-like genomic component, except for the isolate from Thailand which has a genome comprising two molecules, DNA-A and DNA-B. Comparison of the genome nucleotide sequences (Fig. 5) and of the coat protein amino acid sequences (Fig. 6) pointed to a geographically associated variation (Kheyr-Pour et al., 1991; Rybicki, 1994; Padidam et al., 1995; Czosnek & Laterrot, 1997). Moreover, viruses in different geographical regions have different epitope profiles, whereas those from the same region have similar profiles (Macintosh et al., 1992). Differences in the sequence of the capsid protein lead to differences in virus transmissibility by B. tabaci biotypes, a fact that may point to adaptation of the geminivirus to its vector (McGrath & Harrison, 1995).
Stability in Sap
The virus is not sap transmitted. Preparations of virions can be kept for months at -80 °C without losing their capacity to infect tomato tests plants when fed to whiteflies through membranes.
Purification
Leaves of tomato plants infected at the 4-leaf stage are frozen in liquid nitrogen, crushed to a fine powder, and homogenized in a blender after adding ice-cold buffer containing 100 mM trisodium citrate, 18.5 mM ascorbic acid, 60 mM sodium sulphite, 5 mM EDTA, pH 8.0, and 1 % (v/v) 2-mercaptoethanol (2 ml/g of tissue). The homogenate is made 2.5 % (v/v) in Triton X-100, stirred for 18 h, squeezed through four layers of cheesecloth, and clarified by a 10-min centrifugation at 8,000 g. The supernatant is centrifuged for 3 h at 90,000 g in a SW28 Beckman rotor. Pellets are suspended in a buffer containing 10 mM trisodium citrate, 1 mM EDTA, pH 8.0, and 0.1% (v/v) 2-mercaptoethanol (buffer CEM) and subjected to a similar low-speed centrifugation followed by a high-speed centrifugation. Pellets are suspended in 10% (w/v) sucrose in CEM (1 ml of buffer per equivalent of 100 g of leaves), homogenized with a teflon-glass homogenizer, and clarified by a 10-min centrifugation at 4,000 g. The supernatant is applied to a 32-ml linear 10-50% (w/v) sucrose gradient in CEM (extracts obtained from 100 g of tissue are applied on each gradient). The gradients are fractionated (2 ml per fraction) after being subjected to a 14-h centrifugation at 90,000 g in the SW28 rotor.
The presence of the virus in the sucrose gradient fractions is confirmed by electron microscope observations, by the appearance of a specific nucleic acid band after agarose gel electrophoresis, and by infectivity tests using whiteflies fed through membranes on the fractions. Virus-containing fractions are diluted threefold, and virus is concentrated by a 3-hr centrifugation at 90,000 g. Pellets are suspended in 10% sucrose-CEM and stored at -80 °C (Czosnek et al., 1988).
Properties of Particles
Properties of TYLCV-Is particles have not been determined but are probably similar to those of other geminiviruses (Francki et al., 1985). Purified particles are infectious when fed through membranes to whiteflies (Czosnek et al., 1988).
Particle Structure
Geminate particles approximately 20x30 nm in size (Czosnek et al., 1988). Like those of other geminiviruses, the virion is constituted of two incomplete isocahedra with a T=1 surface lattice containing a total of 22 capsomeres each containing five units of the 30.3 kDa coat protein (Francki et al. 1985), and contains a single molecule of covalently closed circular ssDNA of 2787 nucleotides.
Particle Composition
The virion comprises 110 copies of the 30.3 kDa (260 amino acids) capsid protein (total m.w. 3,330,000) and a single genomic DNA molecule of 2787 nucleotides (total m.w. 980,000).
Genome Properties
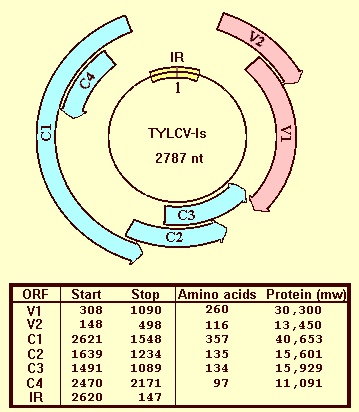
TYLCV-Is is a begomovirus with a single covalently closed ssDNA genomic component of 2787 nucleotides (Navot et al, 1991). The genomes of both TYLCV-Is (accession number X15656) and TYLCV-Is/mild (accession number X76319) encode 6 open reading frames, two on the virus genomic strand (V1 and V2), and four on the complementary genomic strand (C1 to C4) (Fig. 7).
By analogy with other TYLCV and ToLCV isolates, V1 encodes the coat protein, V1 and V2 are involved in virus movement in the infected plant, C1 or replication-associated protein (Rep) is essential for replication and C3 enhances replication. C2 is required for infectivity (Wartig et al., 1997). C4 seems to be involved in the induction of symptoms (Jupin et al., 1994). TYLCV-Is DNA includes a 313 nucleotide-long intergenic region containing a 29 nucleotide-long stem-loop structure with the conserved nonanucleotide TAATATTAC.
Although replication of TYLCV-Is has not been studied, the genomes of other begomoviruses replicate using the rolling circle strategy. In a first step, the viral DNA strand is used as template for the synthesis of the complementary strand to generate a dsDNA replicative form (RF). In a second step the RF serves as template for viral strand synthesis, which is initiated through a DNA cleavage event at the TAATATT¦AC site (Heyraud et al., 1993; Stanley, 1995) mediated by the Rep protein (Laufs et al., 1995). The origin of replication and the Rep binding site have been mapped to the 5' end of the intergenic region. The Rep protein mediates termination of virus strand replication and resolution of concatameric DNAs into circular monomers (Heyraud-Nitschke et al., 1995; Laufs et al., 1995).
TYLCV is transcribed in a bi-directional manner, resulting in mRNAs that correspond to both the virion and complementary-sense open reading frames (Bendahmane, 1991). The mRNAs are polyadenylated and initiate downstream of TATA box motives.
Relations with Cells and Tissues
Following whitefly-mediated inoculation of susceptible tomato plants, TYLCV-DNA concentration reaches a maximum about 4 days before symptoms appear. The highest concentration of TYLCV-DNA is found in rapidly growing tissues (shoot apex, young leaves, roots) and in the stem; the lowest concentrations were found in the older leaves and cotyledons. Young leaves and shoot apices are the best targets for virus inoculation. In these tissues, the viral DNA accumulates at the site of inoculation and is transported first to the roots, then to the shoot apex and to the neighboring leaves and to the flowers. Inoculation through the oldest leaves is inefficient (Ber et al., 1990).
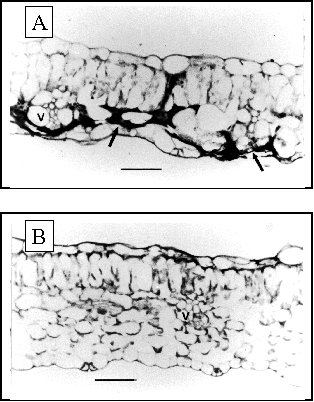
Two weeks after whitefly-mediated inoculation, most of the spongy mesophyll of the inoculated tomato leaf has collapsed (Fig. 8). As a result, the veins, which are usually situated in the middle of the cross section, are now adjacent to the abaxial epidermis. Collapse of the spongy mesophyll causes the leaf to curl upwards because of the turgor pressure now present in the apparently intact palisade mesophyll cells; this is the cause of the typical curling symptoms. Ultrastructural alterations include occluded proteinaceous bodies and swollen thylakoids visible in chloroplasts. Starch granules, which are conspicuous in the plastids of non-inoculated control plants, are absent in the inoculated leaf. The mitochondria become amoeboid and cristae are vesiculated. No apparent cytopathy is visible in the vascular tissues or epidermal cells. The palisade cells remain apparently intact (Michelson et al., 1997). The appearance of systemic disease symptoms is accompanied by major ultrastructural changes affecting primarily the phloem parenchyma cells. The cytopathology includes a degeneracy of nuclei characterized by a reduction of chromatin, hypertrophy of nucleoli and massive accumulation of virus-like particles, together with a severe disorganization of chloroplast structure (Russo et al., 1980).
Localization of TYLCV DNA by in situ hybridization shows strong signals mainly on the perimeter of collapsed spongy mesophyll cells; lighter signal are observed near phloem strands and palisade cells. Well defined virus-specific signals in vascular and mesophyll tissue are best seen four weeks post inoculation in both the vascular bundles and the mesophyll (Michelson et al., 1997).
The virus coat and V2 proteins are apparently involved in cell-to-cell virus movement. TYLCV-Is coat protein possesses a typical nuclear localization signal at its N-terminus (Kunik et al., 1998) that might be instrumental in transporting a coat protein-viral DNA complex (Palanichelvam et al., 1998) through the nucleus of the infected cells, via a karyopherin a -dependent pathway (Kunik et al., 1999).
Ecology and Control
TYLCV infects tomatoes in open fields and greenhouses, causing up to 100% losses in crop production in many countries in the Middle East, Southwest Europe, tropical Africa, Southeast Asia and the Caribbean Islands (Czosnek & Laterrot, 1997). Disease management strategies include the elimination of initial inoculum (eradication of residual infected plants and weeds), planting when whitefly population pressure is low (usually in early spring) and using insecticides (Picó et al., 1996; Nakhla & Maxwell, 1998). Most of the tomato production in Israel is under cover of insect-proof nets (Fig. 9). Tunnels made of UV absorbing plastic sheets that repel whiteflies are starting to be used experimentally (Antignus et al., 1996).
Breeding programs aimed at producing tomato cultivars resistant to TYLCV started in the late 1960's and have expanded since. These programs are based on the introgression of resistance or tolerance found in some accessions of wild tomato species into the domesticated tomato (Fig. 10). Certain accessions of the wild tomato species L. chesmanii, L. chilense, L. hirsutum, L. peruvianum, and L. pimpinellifolium are resistant/tolerant (with mild or no symptoms, but containing virus) to whitefly-mediated inoculation (Zakay et al., 1991). Certain accessions of L. chilense (LA1969) and L. hirsutum (LA1777 and LA386) contain immune individuals (symptomless, no detectable virus) (Zakay et al., 1991; Vidavsky and Czosnek, 1998). Depending on the plant source, resistance was reported to be controlled by one to five genes, either recessive or dominant (Picó et al., 1996). The first commercial tolerant cultivar, TY20, carrying tolerance from L. peruvianum (Pilowsky & Cohen, 1990) and the later advanced lines, show delayed symptoms and lower accumulation of viral DNA (Friedman et al., 1998). An established breeding line, with resistance derived from L. hirsutum, shows total immunity to whitefly-mediated inoculation (Vidavsky & Czosnek, 1998) (Fig. 11). An interspecific hybrid L. esculentum x L. pennellii expressing the TYLCV-Is coat protein showed delay and remission of symptoms upon inoculation (Kunik et al., 1994).
A TYLCV-tolerance gene originating from L. chilense LA1969, Ty-1, has been mapped to tomato chromosome 6 using RFLP markers and has been introgressed into L. esculentum (Zamir et al., 1994). Upon whitefly-mediated inoculation, the resulting line is symptomless and virus replication and movement is impaired (Michelson et al., 1994). Another tolerance gene originating from L. pimpinellifolium has been mapped using RAPD markers, also to chromosome 6 but to a locus different from Ty-1 (Chague et al., 1997).
Notes
The relationship between TYLCV-Is and its whitefly vector presents many aspects of a pathogen-host partnership. The virus is present in the insect for its entire adult life. This long-term association is accompanied by a reduction in the life expectancy and of the fertility of the whitefly (Rubinstein & Czosnek, 1997). The virus can pass transovarially to progeny (Ghanim et al., 1998) and is transmitted from insect to insect by sexual contact (Ghanim & Czosnek, 2000). In addition, the plants that are commercially important hosts such as tomato are the result of man-made breeding. Their wild relatives are either immune or symptomless carriers (Zakay et al., 1991). Therefore we raise the question of whether TYLCV is primarily an insect virus.
Acknowledgements
Supported by grants from the Ministry of Agriculture (Israel), the Ministry of Science (Israel), the United States-Israel Binational Science Foundation (BSF) and the United States-Israel Binational Agriculture Research and Development Fund (BARD).
Figures
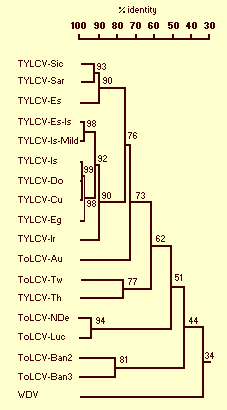
Dendogram showing similarities among the complete nucleotide sequences of the DNA-A-like genomes of TYLCV/ToLCV isolates; percent identity is indicated. Virus isolates were from Italy (Sicily -Sic and Sardinia -Sar), Spain (Es and Es-Is), Israel (Is and Is-Mild), Dominican Republic (Do), Cuba (Cu), Egypt (Eg), Iran (Ir), Australia (Au), Taiwan (Tw), Thailand (Th), and India (New Delhi NDe, Lucknow Luc, and Bangalore Ban2 and 3).

Rooted phylogenetic tree based on coat protein amino acid sequences showing relationships among TYLCV/ToLCV isolates; the numbers above the branches indicate percent bootstrap scores (5,000 replicates). Virus isolates were from Italy (Sicily -Sic and Sardinia -Sar), Spain (Es and Es-Is), Israel (Is and Is-Mild), Dominican Republic (Do), Cuba (Cu), Egypt (Eg), Iran (Ir), Australia (Au), Taiwan (Tw), Thailand (Th), and India (New Delhi NDe, Lucknow Luc, and Bangalore Ban2 and 3). Wheat dwarf virus (WDV) was included as an outgroup.
References list for DPV: Tomato yellow leaf curl virus-Israel (368)
- Antignus & Cohen, Phytopathology 84: 707, 1994.
- Antignus, Mor, Ben Joseph, Lapidot & Cohen, Environmental Entomology 25: 919, 1996.
- Atzmon, van Hoss & Czosnek, European Journal of Plant Pathology 104: 189, 1998.
- Bendahmane, Ph.D. Thesis, Paris, France, 1991.
- Ber, Navot, Zamir, Antignus, Cohen & Czosnek, Archives of Virology 112: 169, 1990.
- Chague, Mercier, Guenard, de Courcel & Vedel, Theoretical and Applied Genetics 95: 671, 1997.
- Cohen & Harpaz, Entomologia Experimentalis et Applicata 7: 155, 1964.
- Cohen & Nitzany, Phytopathology 56: 1127, 1966.
- Cohen, Gera, Ecker, Joseph, Perlsman, Gokkes, Lachman & Antignus, Plant Disease 79: 416, 1995.
- Czosnek & Laterrot, Archives of Virology 142: 1391, 1997.
- Czosnek, Ber, Antignus, Cohen, Navot & Zamir, Phytopathology 78: 508, 1988.
- Czosnek, Kheyr-Pour, Gronenborn, Remetz, Zeidan, Altman, Rabinowitch, Vidavsky, Kedar, Gafni & Czosnek, Plant Molecular Biology 22: 995, 1993.
- Ghanim, Morin, Zeidan & Czosnek, Virology 240: 295, 1998.
- Francki, Milne & Hatta, Atlas of Plant Viruses, CRC Press, Boca Raton, FL, 1: 33, 1985.
- Friedman, Lapidot, Cohen & Pilowsky, Journal of the American Society for Horticultural Science 123: 1004, 1998.
- Heyraud, Matzeit, Schaeffer, Schell & Gronenborn, Biochimie 75: 605, 1993.
- Heyraud-Nitschke, Schumacher, Laufs, Schaefer, Schell & Gronenborn, Nucleic Acids Research 23: 910, 1995.
- Ioannou, Plant Pathology 345: 428, 1985.
- Jupin, De Kouchkovsky, Jouanneau & Gronenborn, Virology 204: 82, 1994.
- Kheyr-Pour, Bendahmane, Matzeit, Accotto, Crespi & Gronenborn, Nucleic Acids Research 19: 6763, 1991.
- Kunik, Salomon, Zamir, Navot, Zeidan, Michelson, Gafni & Czosnek, Bio/Technology 12: 500, 1994.
- Kunik, Palanichelvam, Czosnek, Citovsky & Gafni, The Plant Journal 13: 121, 1998.
- Kunik, Mizrachy, Citovsky & Gafni, Journal of Experimental Botany 50: 731, 1999.
- Laufs, Traut, Heyraud, Matzeit, Rogers, Schell & Gronenborn, Proceedings of the National Academy of Science USA 92: 3879, 1995.
- Macintosh, Robinson & Harrison, Annals of Applied Biology 121: 297, 1992.
- Makkouk, Plant Disease Reporter 62: 259, 1978.
- Makkouk, Shehab & Madjalani, Phytopathologische Zeitschrift 96: 263, 1979.
- Mansour & Al-Musa, Plant Pathology 41: 122, 1992.
- Martinez Zubiaur, Zabalgogeazcoa, De Blas, Sanchez, Peralta, Romero & Ponz, Journal of Phytopathology 144: 277, 1996.
- McGrath & Harrison, Annals of Applied Biology 126: 307, 1995.
- Mehta, Wyman, Nakhla & Maxwell, Journal of Economic Entomology 87: 1291, 1994.
- Michelson, Zamir & Czosnek, Phytopathology 84: 928, 1994.
- Michelson, Zeidan, Zamski, Zamir & Czosnek, Acta Horticulturae 447: 407, 1997.
- Morin, Ghanim, Zeidan, Czosnek, Verbeek, & van den Heuvel, Virology 30: 75, 1999.
- Nakhla & Maxwell, in Plant Virus Disease Control, p. 565, eds. A. Hadidi, R.K. Khetarpal & H. Koganezawa, St Paul: APS Press, 1998.
- Nakhla, Mazyad & Maxwell, Phytopathologia Mediterranea 32: 163, 1993.
- Nakhla, Maxwell, Martinez, Carvalho & Gilbertson, Plant Disease 78: 926, 1994.
- Navas-Castillo, Sánchez-Campos, Díaz, Sáez-Alonso & Moriones, Plant Disease 83: 29, 1999.
- Navot, Pichersky, Zeidan, Zamir & Czosnek, Virology 185: 151, 1991.
- Navot, Zeidan, Pichersky, Zamir & Czosnek, Phytopathology 82: 1199, 1992.
- Nitzany, Phytopathologia Mediterranea 14: 127, 1975.
- Padidam, Beachy & Fauquet, Journal of General Virology 76: 249, 1995.
- Palanichelvam, Kunik, Citovsky & Gafni, Journal of General Virology 79: 2829, 1998.
- Picó, Diez & Nuez, Scientia Horticulturae 67: 151, 1996.
- Pilowski & Cohen, Plant Disease 74: 248, 1990.
- Polston, 2nd Intl. Workshop on Bemisia and Geminiviral Diseases, San Juan, Puerto Rico, L-40, 1998.
- Rubinstein & Czosnek, Journal of General Virology 78: 2683, 1997.
- Russo, Cohen & Martelli, Journal of General Virology 49: 209, 1980.
- Rybicki, Archives of Virology 139: 49, 1994.
- Stanley, Virology 206: 707, 1995.
- Vidavsky & Czosnek, Phytopathology 88: 910, 1998.
- Wartig, Kheyr-Pour, Noris, de Kouchkovsky, Jouanneau, Gronenborn & Jupin, Virology 228: 132, 1997.
- Wernecke, Roye & McLaughlin, Proceedings "Biology and Molecular Epidemiology of geminiviruses", Tucson, AZ. p. 17, 1995.
- Zakay, Navot, Zeidan, Kedar, Rabinowitch, Czosnek & Zamir, Plant Disease 75: 279, 1991.
- Zamir, Michelson, Zakay, Navot, Zeidan, Sarfatti, Eshed, Harel, Pleban, van-Oss, Kedar, Rabinowitch, & Czosnek, Theoretical and Applied Genetics 88: 141, 1994.
- Zeidan & Czosnek, Journal of General Virology 72: 2607, 1991.
- Ghanim & Czosnek, Journal of Virology 74:4738, 2000.