Details of DPV and References
DPV NO: 372 April 2000
Family: Luteoviridae
Genus: Enamovirus
Species: Pea enation mosaic virus | Acronym: PEMV
This description includes PEMV-1 (Genus Enamovirus), PEMV-2 (Genus Umbravirus) and the associated small linear satellite RNA
This is a revised version of DPV 257
Pea enation mosaic virus
Jihad S. Skaf Department of Botany and Plant Pathology, Michigan State University, East Lansing, MI 48824, USA.
Gustaaf A. de Zoeten Department of Botany and Plant Pathology, Michigan State University, East Lansing, MI 48824, USA.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
The first description of what we currently refer to as the pea enation mosaic disease was made by Taubenhaus in 1914. Further description and characterization of the disease, including the first use of the term "enation", was provided by Osborn in 1935.
Synonyms:
Enation pea mosaic virus (Stubbs, 1935)
Pea virus 1 (Rev. Appl. Mycol. 15:418)
Pisum virus 1 (Smith, 1937)
Until recently, the causal agent of pea enation mosaic disease [pea enation mosaic virus (PEMV)], was classified in the monotypic genus Enamovirus (Demler et al., 1996b). However, advances in molecular biology and the determination of the complete sequences of viral RNAs has led to the reclassification of PEMV. The current classification indicates that this disease is caused by the obligate symbiosis between two distinct, yet dependent viruses: the Enamovirus pea enation mosaic virus-1 (PEMV-1), and the Umbravirus pea enation mosaic virus-2 (PEMV-2). From hereon, we shall use "PEMV" to refer to the combination of PEMV-1 and PEMV-2. Whenever possible, data pertaining only to one of the two viruses will be discussed. The genome of each virus consists of single-stranded, plus-sense RNA and the genomes are encapsidated separately in two morphologically distinct isometric particles (22-28 nm). The encapsidating coat protein (CP, Mr ~21,000) is encoded by PEMV-1. Some isolates contain a third, non-essential satellite RNA. PEMV has a restricted host range, is aphid transmissible in the persistent (circulative), non-propagative manner, and is readily transmitted by mechanical inoculation. The distinguishing features of PEMV infections include diagnostic symptoms and the ultrastructural alterations of infected cells. Also, virus titer in infected plants is high compared to other members of the Luteoviridae (related to PEMV-1).
Main Diseases
The symptoms produced by PEMV infections vary widely with host species and cultivars, environmental conditions, and viral isolate. Infected garden pea (Pisum sativum L.) plants develop a slight downward rolling of the trifoliate leaves 4-6 days postinoculation (DPI), followed by an often bright, distinct yellow mosaic on the leaves. The yellow mosaic spots become translucent and clearly delineated (Fig. 1). As the disease progresses, plants develop growth malformation, stunting, and sometimes a top and/or tip necrosis resulting in the loss of apical dominance. Later in infection (21 DPI), diagnostic blisters or enations (hyperplastic outgrowths perpendicular to the leaf plane and associated with the veins) may develop on the underside of the leaves (Figs 2 & 3). Pods are often malformed and warty looking, and contain few if any seeds.
Geographical Distribution
PEMV has been documented in legume growing areas worldwide particularly in northern temperate regions (summarized in Hagedorn, 1996).
Host Range and Symptomatology
PEMV has a narrow host range limited mainly to the family Leguminosae. It includes members of the genera Anthyllis, Astragalus, Cicer, Glycine, Lathyrus, Lens, Lotus, Lupinus, Medicago, Melilotus, Phaseolus, Pisum, Trifolium, and Vicia. Nonleguminous hosts include Gomphrena globosa, Nicotiana benthamiana, N. clevelandii, N. tabacum, and members of the genus Chenopodium (C. quinoa, C. amaranticolor, and C. album) which serve as local lesion hosts (Hagedorn et al., 1964; Izadpana & Shepherd, 1966a; Gonsalves & Shepherd, 1972; Hull & Lane, 1973; Mahmood & Peters, 1973). The mosaic, curling, and stunting developed in infected Nicotiana clevelandii and N. benthamiana are less vivid than in pea (Hagedorn et al., 1964; Demler et al., 1994b). Infections in N. tabacum White Burley are symptomless (Motoyoshi & Hull, 1974). Also, enations have not been reported in Nicotiana species. Chenopodium species can be used as local lesion hosts albeit with high variability. Infected Chenopodium plants show pinpoint chlorotic spots 3-10 DPI. In laboratory settings, pea and N. benthamiana can become infected with PEMV-2. Infected plants are either symptomless or display mild mosaic and leaf curling (Demler et al., 1996b). The satellite RNA associated with some isolates of PEMV does not affect symptom timing or severity in pea (Gonsalves & Shepherd, 1972; Hull & Lane, 1973; Demler & de Zoeten, 1994b). However, its presence attenuates symptom severity in N. benthamiana (Demler & de Zoeten, 1994b).
- Diagnostic species
P. sativum (garden pea): symptoms described in the Main Diseases section.
T. incarnatum (crimson clover): small yellowish spots on the upper surface of the leaves and distinct enations on the underside of the leaves (Osborn, 1935).
V. faba (broad or faba bean): mosaic pattern in the form of spotting rather than mottling. The spots are irregular in shape and tend to appear along the veins of infected leaves (Osborn, 1935).
Propagation species
P. sativum (especially Progress No. 9 and cv. 8221) is the most satisfactory host for the propagation of PEMV. The virus reaches its highest titer 10-14 DPI when peas are grown at 18-22 °C (Izadpana & Shepherd, 1966b).
- Assay species
The aforementioned Chenopodium species can serve as suitable local lesion hosts.
Strains
Several PEMV isolates have been distinguished mainly on the basis of differences in aphid transmissibility and electrophoretic mobility (summarized in Demler et al., 1996b). Sequence variability in PEMV-1 and in PEMV-2 resulting in alterations in aphid transmissibility and systemic movement account for the complexity of strain interactions. A study by Demler et al. (1997) clearly indicated that isolates of PEMV exist as quasi-species of aphid-transmissible and non-transmissible genotypes. The prevalence or total dominance of a particular genotype is determined by the specific selection pressure (bottle-neck) inflicted upon the quasi-species. Therefore, it is imperative that the history of isolates and their maintenance is carefully documented for any strain comparisons to be valid. Strains do not seem to vary in their host range, or serologically.
Transmission by Vectors
PEMV can be transmitted in the circulative, non-propagative manner by at least 10 aphid species including Acyrthosiphon pisum, A. solani, Aphis gossypii, Aulacorthum solani, Macrosiphum avenae, Macrosiphum euphorbiae, Myzus ornatus, M. persicae, Rhopalosiphum padi and Schizaphis graminum (Osborn, 1935; Kennedy et al., 1962; Ehrhardt & Schmutterer, 1964; Schmutterer & Ehrhardt, 1964; Nault, 1967; Nault, 1975; Harris, 1979). Of these species, A. pisum and M. persicae are the most important. Nymphs can acquire the virus in 15 min, and adults in 120 min. After a temperature-dependent latency of 4-70 hours , the virus requires a remarkably short inoculation period of 7-120 seconds. The virus is retained through moulting, is not transmitted to progeny, and does not multiply in aphids (Demler et al., 1996b and references therein).
Transmission through Seed
PEMV is seed-transmissible at a low frequency (1.5%) in P. sativum (Kovachevsky, 1978).
Serology
PEMV is moderately immunogenic in rabbits, with titers ranging from 128 to 1024 attainable by standard methods. Antisera against aphid-transmissible strains of PEMV contain two antibody populations. One reacts with the coat protein and the other with the coat protein-aphid transmission protein readthrough product (see Particle Composition). Antisera generated against an aphid-nontransmissible strain contain only a single population of antibodies.
Relationships
No serological cross-reaction between PEMV and any other virus has been reported (Shepherd, 1970; Peters, 1982).
Stability in Sap
The thermal inactivation temperature is about 65 °C for 10 min. Dilution end point in pea sap is 10-4 and longevity at 20 °C is 4 days (Osborn, 1938). The virus is inactivated by freezing.
Purification
The following procedure for large scale purification of PEMV is based on protocols described by German & de Zoeten (1975) and Demler & de Zoeten (1989). Higher yields (3-5 X) of purified virions can be obtained when P. sativum L. cv. 8221 plants are mechanically inoculated with the naturally occurring "deletion" isolate of PEMV (PEMV-D, GenBank accession No. Y09098, Demler et al., 1997). Symptomatic plants are cut near soil level 10-14 dpi and immediately ground in 1.5 volumes (w/v) of 200 mM sodium acetate, pH 6.0. The homogenate is then clarified using a volume of chloroform equal to the initial tissue weight and centrifuged 20 min at 7,500 gmax. The supernatant is dialyzed overnight against several liters of 200 mM sodium acetate. After another low speed spin, the supernatant is centrifuged three hours at 100,000 gmax and the resulting pellet is resuspended in 100 mM sodium acetate buffer containing 5% (w/v) sucrose in the cold room overnight. After a low speed spin, the supernatant is subjected to another round of high speed centrifugation and resuspension. Virus concentration is estimated spectrophotometrically using an extinction coefficient (260 nm) of 7.5. An aliquot containing about 2 mg virus is loaded onto the top of a 10-40% (w/v) sucrose gradient and centrifuged at 100,000 gmax for 3 hr at 4 °C. The gradient is then fractionated and fractions containing virus particles are pooled, concentrated by high speed centrifugation overnight, and resuspended in 100 mM sodium acetate buffer containing 5% sucrose. Under optimal conditions, virus yield may reach 5 mg/kg.
Properties of Particles
In sucrose density gradients, purified virions sediment as two nucleoprotein components, top (T) and bottom (B). There is no evidence of empty (RNA-free) shells and the relative amounts of the two components vary greatly between isolates (Hull & Lane, 1973). Under high salt and high pH conditions, the top component degrades more rapidly than the bottom component (Hull, 1977).
Sedimentation coefficients (s20,w) at infinite dilution are T: 91-106S and B: 107-122S (Hull, 1981).
Isoelectric point is between 5 and 7 (Adam 1976; Shepherd et al., 1968).
Diffusion coefficient (D20,w) by sedimentation (T and B): 1.89 x 10-7 cm2/sec (Bozarth & Chow, 1966), and by electrophoresis: 1.76 x 10-7 cm2/sec (T) and 1.55 x 10-7 cm2/sec (B) (Hull & Lane, 1973).
Extinction coefficient for T and B mixtures at 260 nm (1 mg/ml, 1 cm lightpath): 7.2 (Adam, 1976) and 7.5 (Shepherd et al., 1968).
Buoyant density (g/cm3) in D2O-sucrose: 1.36 (T and B mixture) (Hull & Lane, 1973), in caesium sulphate: 1.38 (T and B mixture) (Hull, 1976), and in caesium chloride: 1.42-1.43 (B only, T is degraded by CsCl) (Hull & Lane, 1973).
Virion mol. wt. ( x 106) from the Svedberg equation: 4.4 (T) and 5.6 (B) (Hull & Lane, 1973); from sequence data: 4.6-5.2 (T) and 5.7 (B) (Demler & de Zoeten, 1991).
Particle Structure
Most particles in purified preparation are isometric with diameters ranging from 22-28 nm (Fig. 4) (Bozarth & Chow, 1966; Gibbs et al., 1966; Izadpanah & Shepherd, 1966b; Musil et al., 1970; Volvas & Rana, 1972; Hull & Lane, 1973). However, some structural differences between T and B components have been documented. T particles are irregular, pentagonal, and ovoid in appearance (Gibbs et al., 1966; Peters, 1982) and have a diameter of about 25 nm (German & de Zoeten, 1975). B particles have a hexagonal appearance (Gibbs et al., 1966; Peters, 1982) and are about 28 nm in diameter. The particles of PEMV have T=3 icosahedral symmetry (Caspar & Klug, 1962) with the calculated number of 180 protein subunits per B particle and 140-150 subunits per T particle (Hull & Lane, 1973).
Particle Composition
Nucleic acid: PEMV virions contain two different genomic RNA species and occasionally a third satellite RNA. The RNAs are termed RNA1 (the genome of PEMV-1), RNA2 (the genome of PEMV-2), and RNA3 (or sat-RNA) with sedimentation coefficients of 34S, 30S, and 12S, respectively (Gonsalves & Shepherd, 1972). The nucleotide sequences of all three RNAs have been determined and assigned the following accession numbers: L04573 (RNA1 of an aphid non-transmissible isolate), Y09099 (part of RNA1 of an aphid transmissible isolate), U03563 (RNA2), and U03564 (RNA3). Based on nucleotide sequence determination, the RNAs are composed of 5706 nucleotides (mol. wt. 1.9 x 106), 4253 nucleotides (mol. wt. 1.4 x 106), and 717 nucleotides (mol. wt. 2.4 x 105) for RNAs 1, 2, and 3, respectively (Demler & de Zoeten, 1991; Demler et al., 1993, 1994b). All three RNAs are single-stranded and constitute about 28% by weight of the particles. The RNAs of PEMV are not polyadenylated and cannot be aminoacylated at their 3' termini (German et al., 1978). No m7G RNA cap structure has been detected (German, 1974).
Protein: Purified particles of all PEMV isolates contain a 21 kDa coat protein (CP). In addition, aphid transmissible isolates contain a minor component of 54 kDa, shown to mediate aphid transmissibility (Demler et al., 1997). Also, a viral protein (VPg) has been shown to be covalently linked to the 5' terminus of viral RNA (Reisman & de Zoeten, 1982; Wobus et al., 1998; Skaf et al., 2000).
Genome Properties
Natural infections with PEMV are caused by the symbiotic interaction of two unrelated viruses belonging to two different genera: Enamovirus (PEMV-1) and Umbravirus (PEMV-2). Occasionally a third, smaller RNA species (RNA3) is associated with PEMV. The following is a synopsis of the characteristics and the interactions of the three RNAs.
PEMV-1:
PEMV-1 is the sole member of the newly established Enamovirus genus (Family: Luteoviridae). Its RNA (RNA1) bears strong organizational and sequence similarity to the RNAs of viruses in the Polerovirus genus encompassing beet western yellows virus (BWYV), potato leafroll virus (PLRV), and the NY-RPV isolate of barley yellow dwarf virus (NY-RPV BYDV) (Veidt et al., 1988; Mayo et al., 1989; Demler & de Zoeten, 1991; Vincent et al., 1991). RNA1 (5706 nucleotides) contains five open reading frames (ORF) (Fig. 5) (Demler & de Zoeten, 1991). ORF0 can potentially encode a protein of 34 kDa of unknown function or relationship to other proteins. ORF1 encodes an 84 kDa protein and overlaps (out of frame) 90% of ORF0. In vitro translation of RNA1 generated products of 36 and 88 kDa, suggesting that ORFs 0 and 1 are expressed by independent translational initiation (Demler & de Zoeten, 1991). A picornavirus 3C-like protease core sequence has been identified within the ORF1 protein, suggesting that proteolytic processing may be involved in the expression of the viral genome. Similar to Poleroviruses, the genome-linked protein (VPg) of PEMV is encoded by ORF1 of RNA1 downstream of the putative protease motif (Fig. 5) (Wobus et al., 1998). It is not known at this time if this VPg is covalently linked to all three viral RNAs. Protease treatment does not abolish infectivity suggesting that the VPg is not required for the initial translation of viral RNA. ORF2 overlaps (in a different frame) the C-terminal 23% of ORF1. ORF2 encodes a 67 kDa protein that is postulated to be the core of the viral RNA-dependent RNA polymerase (Demler and de Zoeten, 1991; Habili & Symons, 1989; Koonin, 1991; Koonin & Dolja, 1993). In vitro translation analysis suggests that this protein is expressed as a fusion of the 84 kDa and 67 kDa protein reading frames by translational frameshift. ORF3 encodes the 21 kDa viral CP, and is immediately followed in the same reading frame by ORF5 (encodes a 33 kDa protein). The 33 kDa protein is expressed as a translational readthrough (RT) fusion with the 21 kDa CP (Demler et al., 1997). The resulting 54 kDa minor protein is dispensable for infectivity but its presence is mandatory for aphid transmission. In response to repeated mechanical inoculation, PEMV loses aphid transmissibility (resulting in aphid nontransmissible isolates) by a number of mechanisms including the generation of genotypes lacking the 33 kDa protein or the downregulation of the expression of the translational readthrough protein (Demler et al., 1997). Deletions in the CP ORF do not inhibit the systemic movement of PEMV or alter the accumulation of truncated viral RNA (compared to wild-type isolates) (Skaf et al., 1997). However, pea plants infected with CP-less mutants exhibit wilting and severe necrosis, which appears earlier and is more severe than the symptoms observed in wild-type infections. Interestingly, the absence of the CP (and hence virions) from cells infected with CP deletion mutants does not affect the formation of replication complexes or the double-membrane bound vesicles characteristic of PEMV infection (see Relations with Cells and Tissues). The association of these vesicles, whose production is controlled by RNA1 (Demler et al., 1994a), with viral RNA-dependent RNA-polymerase (Powell & de Zoeten, 1977), viral ss- and ds-RNA (de Zoeten et al., 1976), as well as with plasmodesmata and phloem tissue (de Zoeten & Gaard, 1983) suggests their involvement in virus movement throughout the plant. This CP-independent movement of PEMV is in sharp contrast to the absolute necessity of the CP for the movement of other viruses of the Luteoviridae. This contrast may be due to functions provided in trans by RNA2 to the PEMV complex. RNA1 lacks ORF4 (encoding a 17-19 kDa protein) found nested within the Polerovirus CP genes (Martin et al., 1990). Neither the CP nor the 33 kDa protein are expressed in in vitro translation of RNA1. There is evidence from infected tissue for the presence of an 1800-nucleotide RNA specific to the 3'-terminal region of RNA1 (Demler & de Zoeten, 1991; Demler et al., 1994a). These data suggest that expression of these two proteins occurs from a single subgenomic messenger. RNA1 is capable of replicating in isolated protoplasts but cannot establish systemic infections in plants on its own regardless of the inoculation method used (Demler et al., 1996b).
PEMV-2:
One of the most interesting features of PEMV is the surprising lack of sequence homology (especially at the 5' and 3' termini) between RNA2 and RNA1 (Demler et al., 1993). The only exception is a short region that immediately precedes ORFs hypothesized to be expressed from subgenomic mRNAs (ORFs 3 and 5 of RNA1 and ORFs 3 and 4 of RNA2) (Demler et al., 1994b). RNA2 contains four complete ORFs and one incomplete one (Fig. 5). ORF1 can potentially encode a 33 kDa protein of unidentified function. ORF1 overlaps (out of frame) the first 37 amino acids of the 65 kDa ORF2. In vitro translation indicates that the expression of ORF1 continues (by frame shifting) into ORF2 producing a 94 kDa fusion protein (Demler et al., 1996b). ORF2 contains sequence motifs typical of RNA polymerases and bears a strong taxonomic relationship to those of carmo-like viruses (Habili & Symons, 1989; Koonin, 1991; Koonin & Dolja, 1993; Miller et al., 1995). The polymerase encoded by RNA2 is unrelated to the polymerase encoded by RNA1. ORF2 is followed by two largely overlapping (in distinct reading frames) ORFs (ORFs 3 and 4) potentially encoding 26 and 27 kDa proteins, respectively. Circumstantial as well as experimental evidence indicate that both cell-to-cell and systemic movement, mechanical transmissibility, and the relaxation of phloem limitation of PEMV are mediated by ORFs 3 and 4 and their proteins. ORFs 3 and 4 are followed by a 293 nucleotide non-coding region leading to a potential fifth ORF (15 kDa) that lacks a conventional stop codon. The loss of this highly variable ORF in some clones confers no distinctive phenotype in pea or in N. benthamiana (Demler et al., 1993). Serological screening with anti-PEMV antibodies has failed to identify a CP encoded by RNA2. In vitro translation analysis of RNA2 has demonstrated only a single product of 45 kDa. RNA2 is mechanically transmissible and can establish a systemic infection in whole plants in the absence of RNA1 (see Host Range and Symptomatology).
RNA3:
Some isolates of PEMV possess a third RNA (RNA3) with B-type satellite RNA characteristics. RNA3 is not infectious on its own and was long considered an artifact of virus purification (German & de Zoeten, 1975; Adam et al., 1979). RNA3 shares no sequence homology with RNA1; however, the first 12 and 7 nucleotides at the 5' and 3' termini, respectively, are identical between RNAs 2 and 3. This 717 nucleotides RNA is encapsidated in the CP encoded by RNA1 and replicated by the polymerase encoded by RNA2 (Demler & de Zoeten, 1989; Demler et al., 1994b). In return, the presence of RNA3 may result in more stable virions (Demler et al., 1996b). This trilateral interaction between RNA3 and the genomic components of PEMV is reminiscent of that described for the satellite RNA of groundnut rosette virus (GRV). Indeed, both satellite RNAs share considerable sequence homology and can be interchangeably replicated by their counterparts' helper umbraviruses (Demler et al., 1996a). In contrast with the GRV satellite, RNA3 is dispensable for the encapsidation and aphid transmission of PEMV.
Relations with Cells and Tissues
In contrast with the other members of the Luteoviridae, which are phloem-limited, particles of PEMV can be found in relatively high concentration in most tissues of infected plants. This high concentration makes it easy to detect PEMV in infected plants by electron microscopy. Virus particles are observed either individually or in clusters in both the nuclei and cytoplasm of infected cells (Shikata & Maramorosch, 1966; Shikata et al., 1966; de Zoeten et al., 1972; Burgess et al., 1974; Motoyoshi & Hull, 1974; Demler et al.,1994a). The site of virus assembly is still unclear.
However, the most striking characteristic of PEMV infection is, perhaps, the proliferation of fibril-containing vesicular structures (Fig. 6) in infected tissue, including all parenchymatic cell types and particularly in phloem tissues. These vesicles originate from the inner nuclear membrane and are deposited into the perinuclear space from which membrane-bound groups of vesicles are extruded into the cytoplasm. Fractions enriched for nuclei and for these vesicular structures contained both the negative- and positive-sense strands of PEMV RNA (de Zoeten et al., 1976), and PEMV induced RNA-dependent RNA polymerase activity was also localized to these fractions (Powell & de Zoeten, 1977). These observations support the idea that the membranous vesicles contain the replicative complex of PEMV. In addition, nuclei from uninfected pea cells have been shown to be capable of replicating PEMV RNA (Powell & de Zoeten, 1977). This suggests that PEMV can replicate in the nucleus; PEMV is to date the only plus-sense RNA plant virus known to do so. The membranous vesicles are also found associated with, or traversing plasmodesmata of mesophyll cells, phloem parenchyma cells, sieve elements, and fully developed sieve tubes (de Zoeten & Gaard, 1983). The systemic mobility of these structures coupled with their demonstrated polymerase activity suggests a possible role for them in the systemic spread of PEMV infection. In addition, transient, electron-dense, dagger-like structures protruding into the cytoplasm can often be found associated with plasmodesmata of both uninfected and necrotic companion cells and sieve elements (de Zoeten & Gaard, 1983). Another cytopathic feature characteristic of PEMV infection in legumes is the presence of unusual feather-like crystalline inclusions in epidermal cells. It is not clear whether these structures are of viral origin or represent a possible defence or stress-induced reaction of the host. Protoplasts infected with RNA1 form the above mentioned double membrane vesicles and contain large crystalline aggregates of virus particles. Neither the vesicles nor the aggregates could be observed in RNA2-infected protoplasts (Demler et al., 1994a).
In viruliferous aphids, PEMV virions were identified in the gut lumen, in fat bodies, in epithelium and muscle cells of the midgut, in hemocytes, and in electron dense viroplasm-like structures believed to be part of the lysosomal apparatus (Shikata et al., 1966; de Zoeten et al., 1972; Richardson & Sylvester, 1965; Harris et al., 1975; Adam et al., 1980). Virions of aphid-transmissible (but not of aphid-nontransmissible) isolates of PEMV were found to be associated with the basal lamina and plasma-membrane complex of the accessory salivary gland, implying a directional membrane-mediated shuttling of virions from the hemocoel to the stylet (Harris et al., 1975; Harris, 1979). It is currently accepted that PEMV does not replicate in its aphid vectors (Demler et al., 1996b).
Ecology and Control
PEMV is an important viral disease of pea and other legumes worldwide (Hagedorn, 1974, 1996). Its importance as a pathogen is related to the fact that, for a variety of reasons, insecticides for aphid control in primary and secondary hosts have not been used routinely in commercial production (Hagedorn, 1996). Another logical approach to controlling PEMV is to reduce the transfer of the virus from overwintering sources by breeding aphid-resistant perennial crop legume species. This approach has not been used actively to reduce disease incidence. Therefore, it is recommended that peas or faba beans should not be planted next to alfalfa or clover fields. A number of tolerant lentil (USDA accessions PI Nos. 472547 and 472609) and pea (developed from USDA PI No. 140295) varieties (containing the single dominant resistance gene En) have been described (de Zoeten et al., 1995; Hagedorn, 1996). However, these traits have not been used in commercial varieties. In addition, tolerance to PEMV used as a control measure would have adverse epidemiological consequences, because it would tend to enlarge the virus reservoir.
Notes
Since the first description of "pea mosaic", our understanding of PEMV has evolved as new molecular, biochemical, and genetic techniques have become available. It is clear now that the interactions between the genetic components of PEMV manifest themselves in the apparently obligate symbiosis between RNAs 1 and 2 required to produce wild-type infections. In this symbiosis, the vital functions lacking from each RNA are provided in trans by the other component resulting in a highly infectious entity. Determining the development (in evolutionary terms) of the components of this complex from or to independence or symbiosis may provide answers related to the evolution of other viruses.
Figures

Electron micrograph of purified PEMV particles negatively stained with uranyl acetate. Scale bar: 100 nm.
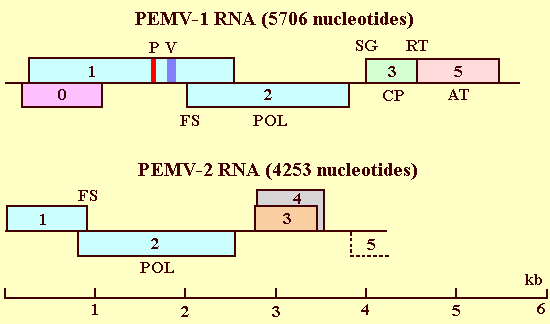
Schematic representation of the genome structure and organization of the RNAs of PEMV-1 and PEMV-2. Numbered hollow boxes indicate open reading frames. AT: aphid transmission, CP: coat protein, FS: frameshift, P: picornavirus 3C-like protease core sequence, POL: RNA polymerase, SG: 5'-end of subgenomic RNA, RT: translational readthrough, V: VPg.
References list for DPV: Pea enation mosaic virus (372)
- Adam, Dissertation am Fachbereich Biologie der Universität Tübingen, FRG, 1976.
- Adam, Sander & Shepherd, Virology 92: 1, 1979.
- Adam, Sander & Lee, Invertebrate Systems in vitro, Elsevier-North Holland, Amsterdam, p.483, 1980.
- Bozarth & Chow, Contributions, Boyce Thompson Institute for Plant Research 23: 301, 1966.
- Burgess, Motoyoshi & Fleming, Planta 119: 247, 1974.
- Caspar & Klug, Cold Spring Harbor Symposia on Quantitative Biology 27: 1, 1962.
- Demler & de Zoeten, Journal of General Virology 70: 1075, 1989.
- Demler & de Zoeten, Journal of General Virology 72: 1819, 1991.
- Demler, Rucker & de Zoeten, Journal of General Virology 74: 1, 1993.
- Demler, Borkhsenious, Rucker & de Zoeten, Journal of General Virology 75: 997, 1994a.
- Demler, Rucker, Nooruddin & de Zoeten, Journal of General Virology 75: 1399, 1994b.
- Demler, Rucker, de Zoeten, Ziegler, Robinson & Murant, Journal of General Virology 77: 2847, 1996a.
- Demler, de Zoeten, Adam & Harris, The Plant Viruses, Volume 5, Polyhedral Virions and Bipartite RNA Genomes, Plenum Press, NY, p. 303, 1996b.
- Demler, Rucker, Skaf & de Zoeten Journal of General Virology 78: 511, 1997.
- de Zoeten & Gaard, Intervirology 19: 85, 1983.
- de Zoeten, Gaard & Diez, Virology 48: 638, 1972.
- de Zoeten, Powell, Gaard & German, Virology 70: 459, 1976.
- de Zoeten, Demler & Zlatkina, Pathogenesis and Host Specificity in Plant Diseases, Histopathological, Biochemical, Genetic, and Molecular Bases, Volume III: Viruses & Viroids, Pergamon, Elsevier Science Inc., NY, 1995.
- Ehrhardt & Schmutterer, Zeitschrift für Pflanzenchutz 71: 381,1964.
- German, Purification and properties of the replicative forms and replicative intermediates of pea enation mosaic virus, Ph. D. Thesis, 1974.
- German & de Zoeten, Virology 66: 172, 1975.
- German, de Zoeten & Hall, Intervirology 9: 226, 1978.
- Gibbs, Harrison & Woods, Virology 29: 348, 1966.
- Gonsalves & Shepherd, Virology 48: 709, 1972.
- Habili & Symons, Nucleic Acids Research 17: 9543, 1989.
- Hagedorn, American Phytopathological Society Monograph No. 9, 1974.
- Hagedorn, The Plant Viruses, Volume 5, Polyhedral Virions and Bipartite RNA Genomes, Plenum Press, NY, p. 345, 1996.
- Hagedorn, Layne & Ruppell, Phytopathology 64: 843, 1964.
- Harris, Leafhoppers Vectors and Plant Disease Agents, Academic Press, NY, p. 217, 1979.
- Harris, Bath, Thottappilly & Hooper, Virology 65: 148, 1975.
- Hull, Virology 75: 18, 1976.
- Hull, Aphids as Virus Vectors, Academic Press, NY, p. 137, 1977.
- Hull, Handbook of Plant Virus Infections, Comparative Diagnosis, Elsevier/North-Holland, Amsterdam. p. 239, 1981.
- Hull & Lane, Virology 55: 1, 1973.
- Izadpana & Shepherd, Phytopathology 56: 458, 1966a.
- Izadpana & Shepherd, Virology 28: 463, 1966b.
- Kennedy, Day & Eastop, A Conspectus of Aphids as Vectors of Plant Viruses, 1962.
- Koonin, Journal of General Virology 72: 2197, 1991.
- Koonin & Dolja, Critical Reviews in Biochemistry and Molecular Biology 28: 375, 1993.
- Kovachevsky, Rasteniev dni Nauki, 15: 108, 1978.
- Mahmood & Peters, Netherlands Journal of Plant Pathology 79: 138, 1973.
- Martin, Keese, Young, Waterhouse & Gerlach, Annual Review of Phytopathology 28: 341, 1990.
- Mayo, Robinson, Jolly & Hyman, Journal of General Virology 70: 1037, 1989.
- Miller, Dinesh-Kumar & Paul, Critical Reviews in Plant Sciences 14: 179, 1995.
- Motoyoshi & Hull, Journal of General Virology 24: 89, 1974.
- Musil, Marcinka & Ciampar, Acta Virologica 14: 285, 1970.
- Nault, Journal of Economic Entomology 69: 1586,1967.
- Nault, Phytopathology 65: 496, 1975.
- Osborn, Phytopathology 25: 160, 1935.
- Osborn, Phytopathology 28: 923, 1938.
- Peters, CMI/AAB Description of Plant Viruses No. 257, 1982.
- Powell & de Zoeten, Proceedings of the National Academy of Sciences USA 74: 2919, 1977.
- Reisman & de Zoeten, Journal of General Virology 62: 187, 1982.
- Richardson & Sylvester, Virology 25: 472, 1965.
- Schmutterer & Ehrhardt, Phytopathologische Zeitschrift 50: 80, 1964.
- Shepherd, CMI/AAB Descriptions of Plant Viruses No. 25, 1970.
- Shepherd, Wakeman & Ghabrial, Virology 35: 255, 1968.
- Shikata & Maramorosch, Virology 30: 439, 1966.
- Shikata, Maramorosch & Granados, Virology 29: 426, 1966.
- Skaf, Rucker, Demler, Wobus & de Zoeten, Molecular Plant-Microbe Interactions, 10: 929, 1997.
- Skaf, Schultz, Hirata & de Zoeten, Journal of General Virology 81: 1103, 2000.
- Smith, Textbook of Plant Virus Diseases, 1937.
- Stubbs, Viroses of the garden pea (Pisum sativum), Ph. D. Thesis, 1935.
- Taubenhaus, Delaware Agriculture Experiment Station Bulletin No. 106, 1914.
- Veidt, Lot, Leiser, Scheidecker, Guilley, Richards & Jonard, Nucleic Acids Research 16: 9917, 1988.
- Vincent, Lister & Larkins, Journal of General Virology 72: 2347, 1991.
- Volvas & Rana, Phytopathologia Mediterranea 11: 97, 1972.
- Wobus, Skaf, Schultz & de Zoeten, Journal of General Virology 79: 2023, 1998.



