Details of DPV and References
DPV NO: 376 July 2000
Family: Betaflexiviridae
Genus: Capillovirus
Species: Apple stem grooving virus | Acronym: ASGV
This is a revised version of DPV 31
Apple stem grooving virus
N. Yoshikawa Faculty of Agriculture, Iwate University, Morioka 020-8550, Japan.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Lister et al. (1965) from apple, Wallace & Drake (1962) from citrus, and Inouye et al. (1979) from lily.
- Selected Synonyms
- Apple latent virus Type 2 (C-431 isolate)(
Lister et al., 1965)
- Dark green epinasty virus (in Chenopodium quinoa) ( Waterworth & Gilmer, 1969)
- Citrus tatter leaf virus ( Wallace & Drake, 1962)
- Citrange stunt virus ( Wallace & Drake, 1968; Roistacher, 1988)
- Dark green epinasty virus (in Chenopodium quinoa) ( Waterworth & Gilmer, 1969)
A virus with flexuous filamentous particles 620-650 long and 12nm in diameter containing linear positive-sense, ssRNA. Probably occurs world-wide in Rosaceae fruit trees and citrus in which it is usually symptomless. Its natural mode of spread is unknown. It is symptomlessly in most Rosaceae fruit trees and citrus plants. Transmissible by mechanical inoculation to herbaceous plants.
Main Diseases
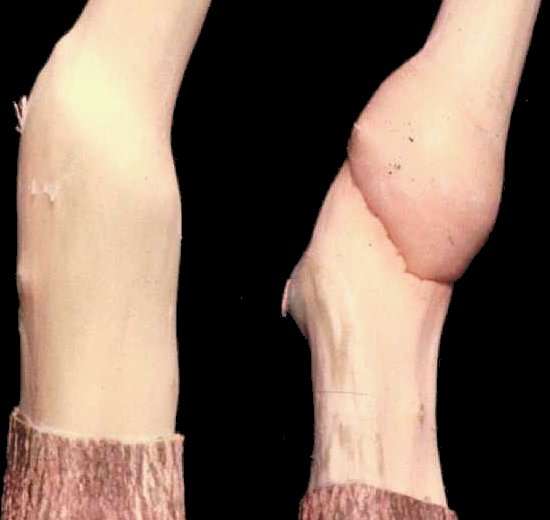
Causes stem grooving, brown line and graft union abnormalities in Virginia Crab (Lister, 1970), topworking disease of apple trees grown on Mitsuba kaido (Malus sieboldii) in Japan ( Yanase, 1974; 1982), and bud union abnormalities of citrus trees on trifoliate orange (Fig. 1; Fig. 2; Fig. 3) (Calavan et al., 1963; Miyakawa & Matsui, 1976; Miyakawa, 1980).
Geographical Distribution
Probably distributed wherever apples are cultivated. Natural spread in citrus plants is reported in China and Japan (Ke & Wu, 1991; Miyakawa, 1980; Miyakawa & Matsui, 1976; Su & Cheon, 1984; Zhang et al., 1988).
Host Range and Symptomatology
Infects the Rosaceae fruit trees, apple, European pear, Japanese pear, and Japanese apricot and citrus plants. Infects experimentally more than 40 species in 17 plant families (Aizoaceae, Amaranthaceae, Apocynaceae, Caryophyllaceae, Chenopodiaceae, Compositae, Convolvulaceae, Cucurbitaceae, Graminae, Labiatae, Leguminosae, Liliaceae, Pedaliaceae, Rosaceae, Rutaceae, Scrophulariaceae, Solanaceae) (Inouye et al., 1979; Lister et al., 1965; Semancik & Weathers, 1965; Waterworth & Gilmer, 1969), many symptomlessly. Transmissible from apple to C. quinoa by mechanical inoculation of extracts from buds, young leaves or petals ground in 0.05M phosphate buffer (pH7-8) containing 2% (v/v) nicotine base or 2% (w/v) polyvinyl pyrrolidone (Lister, 1970).
- Diagnostic species
Malus pumila cv. Virginia Crab (apple). Stem grooving in the wood. Necrosis and pitting at the union of Viriginia Crab and rootstock (Fig.1).
M. sieboldii "MO65". Necrosis and pitting at the scion-rootstock union (Fig.2).
Citrus excelsa. Tatter leaf (Fig.4).
Citrange (Poncirus trifoliata x C. sinensis) cvs. Rusk and Troyer. Leaf malformation and chlorotic mottling (Fig.5).
P. trifoliata and citrange. Bud-union crease between infected scion and rootstock (Fig.6).
Chenopodium quinoa. Systemic leaf epinasty, distortion, and mottle (Fig.7) and stunting. Some isolates induce necrotic lesions 0.5-2 mm in diameter in inoculated leaves.
Nicotiana glutinosa. Systemic conspicuous yellow to mild mosaic symptoms depending on the isolate.
Phaseolus vulgaris cv. Pinto. Purple-brown spots or rings 0.5-3 mm in diameter or chlorotic spots on the inoculated leaves. Some isolates induced veinal necrosis in inoculated leaves. Symptoms are generally were more severe in winter.
- Propagation species
C. quinoa is the most useful plant for propagation. Zucchini squash is also reported as a propagation host for some isolates (Uyemoto & Gilmer, 1971). To transmit the virus from fruit trees, N. occidentalis, a symptomless host is useful., N. glutinosa can be used to eliminate contaminating viruses from apple such as Apple chlorotic leaf spot virus and Tobacco mosaic virus.
- Assay species
C. quinoa and P. vulgaris cv. Pinto are useful local lesion hosts.
Strains
Many isolates are reported from apple, Japanese pear, European pear, Japanese apricot, lily and citrus plants, but most are not well characterized biologically and serologically. Some isolates have been differentiated only on symptomatology (Lister, 1970).
This virus and Citrus tatter leaf virus (CTLV) have been regarded as distinct viruses. However, CTLV from citrus and lily is indistinguishable from ASGV from Rosaceae fruit trees biologically, serologically, in genome organization and in nucleotide sequence (Kawai et al., 1991; Magome et al., 1997a; Ohira et al., 1995; Yoshikawa et al., 1993; 1996). Isolate Li-23 from lily can infect citrus plants (N. Inouye, personal communication) and isolate V-3 from Japanese pear induced symptoms similar to those by CTLV from citrus on Rusk citrange (Iwanami et al. 1991). From these results, CTLV is regarded as an isolate of ASGV.
Virus isolates from apple, Japanese pear and European pear trees comprise at least two to four variants that differ considerably from each other in nucleotide sequence (Magome et al., 1997a; Yoshikawa et al., 1996). The composition of sequence variants within a tree differed among leaves from different branches, showing that each sequence variant is distributed unevenly within an individual tree (Magome et al., 1999).
Transmission by Vectors
No vectors are reported.
Transmission through Seed
Transmitted through seed to progeny seedlings of lily (1.8%) and C. quinoa (2.5 - 60.0%) (Inouye et al., 1979).
Serology
Moderately antigenic, giving antisera from rabbits with titers of 1/128 to 1/4,096. Polyclonal antisera have been prepared in rabbits against purified virus or coat protein expressed in Escherichia coli (de Sequeira & Lister, 1969a; Fuchs, 1980; Inouye et al., 1979; Nishio et al., 1989; Yanase et al., 1986). ELISA has been used to detect the virus in fruit trees (Fuchs, 1980; Namba et al., 1992; Yanase et al., 1986). Monoclonal antibodies were produced in mice against an isolate from citrus; three selected monoclonal antibodies react with all isolates tested, including 9 isolates from citrus in Japan, 4 isolates from citrus in the USA, 6 isolates from Chinese citrus, and isolates from lily, apple and Japanese apricot (Kawai et al., 1991).
Relationships
Serologically unrelated to all known virus species in the genus Capillovirus. Similarities exist between the virus and virus species in the genera Trichovirus and Vitivirus in the amino acid sequences of conserved polymerase motif, putative movement protein and coat protein (Jelkmann, 1995; Yoshikawa et al., 1992; 1997).
Stability in Sap
In bentonite-clarified C. quinoa sap, the thermal inactivation point (10 min) is between 60 and 63°C, the half-life at 45°C is 105±8 min, the dilution end-point is about 10-4, and infectivity is retained for more than 2 days at 20°C and more than 27 days at 4°C (Lister, 1970). In crude sap of infected C. quinoa, an isolate from lily has a thermal inactivation point between 65 and 70°C, a dilution end-point between 10-4 and 10-5 and longevity in vitro between 4 and 8 days at 20°C (Inouye et al., 1979).
Purification
Purified from infected C. quinoa (de Sequeira & Lister ,1969b) by homogenizing 100g leaves in 0.01M or 0.05M phosphate buffer (pH7-8) containing 0.15% sodium thioglycollate or 1% 2-mercaptoethanol. Clarify the extract by low speed centrifugation and squeeze through cheesecloth. Add a bentonite suspension (30-40 mg/ml in 0.01M phosphate buffer, pH7-8) slowly and clarify by centrifuging at low speed. Repeat this clarification step until the supernatant fluid is clear. Precipitate the virus from the supernatant fluid by adding PEG (mol. wt 6,000) to 4-8 %(w/v) and NaCl to 0.02M, incubate for 1 hr and centrifuge at low speed. Resuspend the pellet in 0.01M phosphate buffer (pH7.0) and clarify by centrifuging at low speed. Concentrate the virus by ultracentrifugation and purify further by sucrose density gradient centrifugation. Yields are 50 - 250 µg/100g leaf tissue.
Properties of Particles
Sedimentation coefficient, S20,w (svedbergs) c. 112 at 0.3-0.6 mg/ml in 0.01M neutral phosphate buffer.
Isoelectric point; c. pH 4.3 at ionic strength of 0.1.
Electrophoretic mobility; -10.3 and -6.5 X 10-5 cm2. sec-1. V-1 respectively at pH 7.0 and pH 6.0 (ionic strength = 0.1).
A260/A280 : 1.18 not corrected for light-scattering (Lister, 1970).
Particle Structure
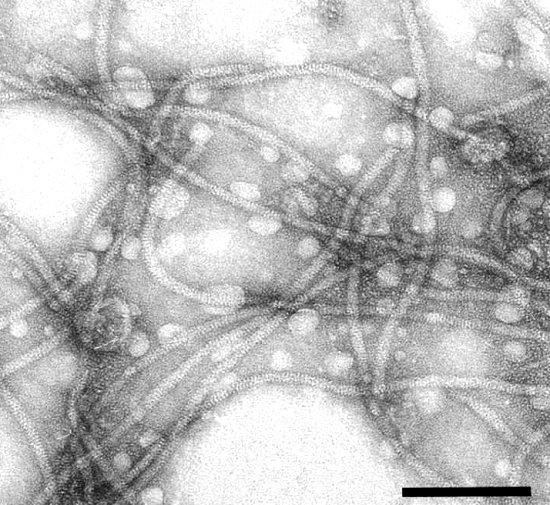
Particles are very flexuous filaments, 619±14 nm long and c.12 nm in width, with obvious cross-banding and helical symmetry (Fig. 8) and a pitch of c. 3.8 nm. (Lister, 1970).
Particle Composition
Nucleic acid: Linear positive-sense ssRNA of Mr c. 2.30 x 106 or 6,496 nucleotides excluding a polyA-tail (Yoshikawa & Takahashi, 1988; Yoshikawa et al., 1992); about 5% of particle weight. Nucleotide base ratios for isolate P-209 from apple are: G 23.0%; A 30.6%; C 18.4%; U 28.0% (Yoshikawa et al., 1992).
Protein: Particles contain a single polypeptide species of Mr 27,000 (Nishio et al., 1989; Yoshikawa & Takahashi, 1988).
Genome Properties
The complete nucleotide sequences (6496 bases) of the single RNA genome of three isolates have been determined. They are isolates P-209 (accession no. D14995) from apple, L (D16681) and Li-23 (AB004063) from lilies (Yoshikawa et al., 1992; Ohira et al., 1995; Terauchi et al., 1997). Identities of the nucleotide sequences were 82.9% (P-209/L), 83.0% (P-209/Li-23) and 98.4% (L/Li-23) (Terauchi et al., 1997). The genomic RNA has two overlapping ORFs in the positive strand (Fig. 9). ORF1 (bases 37-6341) encodes a 241- to 242 kDa polyprotein (2105 amino acids) containing the consensus motifs of methyltransferase, papain-like protease, nucleotide triphosphate- binding helicase, RNA polymerase, and coat protein (CP) in the C-terminal region. ORF2 (bases 4788-5747) encodes a 36 kDa putative movement protein (320 aa). A region (designated V-region) (aa position 1585-1868) of the ORF1-encoded protein between the polymerase and the CP, that encodes ORF2 in another frame does not have any functional motifs found in other known plant virus genomes. This region shows high variability among isolates and sequence variants (Fig. 9) (Magome et al., 1997a).
Although the CP is located in the C-terminal region of the ORF1-encoded polyprotein and genomic RNA directed the synthesis of a polypeptide of c. 200 kDa as a major product in in vitro translation, and was immunoprecipitated by antiserum to virus particle preparations (Yoshikawa & Takahashi, 1992), the following evidence suggests that the CP is expressed from a subgenomic RNA (Magome et al., 1997b). Analysis of dsRNA from infected tissues indicate that all isolates tested contain five virus-specific dsRNAs (6.5, 5.5, 4.5, 2.0 and 1.0 kbp). The 6.5 kbp species represents the double-stranded form of the full-length genome, whereas the 2.0 and the 1.0 kbp species may be the double-stranded forms of subgenomic RNAs coding for the putative movement protein and the CP, respectively. The size of the E. coli -expressed protein corresponding to the C-terminal region of the ORF1-encoded protein, which starts with the Met at aa position 1869, agreed with that of the CP.
Relations with Cells and Tissues
In infected C. quinoa leaves, the particles occur singly or as aggregates in the cytoplasm of mesophyll and phloem parenchyma cells. No virus-specific inclusion bodies, such as pinwheels, viroplasmas or vesicles were observed (Ohki et al., 1989).
Ecology and Control
Because the virus is thought to be transmitted in the field only by grafting, planting virus-free plants is the best means of controlling decline problems in apple and citrus plants due to the virus.
Figures

Necrosis at the union of Virginia Crab and an infected rootstock (right ) compared with a healthy union (left) (Courtesy I. Machida).

Basal scion swelling "Dai-Make" , at a bud-union of infected Satsuma Mandarin on P. trifoliata rootstock (Courtesy T. Miyakawa).
References list for DPV: Apple stem grooving virus (376)
- Calavan, Christiansen & Roistacher, Plant Disease Reporter 47: 971, 1963.
- de Sequeira & Lister, Phytopathology 59: 572, 1969a.
- de Sequeira & Lister, Phytopathology 59: 1740, 1969b.
- Fuchs, Acta Phytopathologica Academiae Scientiarum Hungaricae 15: 69, 1980.
- Inouye, Maeda & Mitsuhata, Annals of the Phytopathological Society of Japan 45: 712, 1979.
- Iwanami, Kano & Koizumi, Annals of the Phytopathological Society of Japan 57: 74, 1991.
- Jelkmann, Journal of General Virology 76: 2015, 1995.
- Kawai, Kobayashi, Tsukamoto, Dai, Kimishima, Kimura & Goto, Research Bulletin of the Plant Protection Service Japan 27: 55, 1991.
- Ke & Wu, Proceedings of the 11th Conference of the International Organization of Citrus Virologists : 358, 1991.
- Lister, CMI/AAB Descriptions of Plant Viruses 31: 1970.
- Lister, Bancroft & Nadakavukaren, Phytopathology 55: 859, 1965.
- Magome, Yoshikawa, Takahashi, Ito & Miyakawa, Phytopathology 87: 389, 1997a.
- Magome, Terauchi, Yoshikawa & Takahashi, Annals of the Phytopathological Society of Japan 63: 450, 1997b.
- Magome, Yoshikawa & Takahashi, Phytopathology 89: 136, 1999.
- Miyakawa, Proceedings of the 8th Conference of the International Organization of Citrus Virologists : 220, 1980.
- Miyakawa & Matsui, Proceedings of the 7th Conference of the International Organization of Citrus Virologists : 125, 1976.
- Namba, Maekawa, Tanaka & Yamashita, Research Bulletin of the Plant Protection Service Japan 28: 13, 1992.
- Nishio, Kawai, Namba & Yamashita, Annals of the Phytopathological Society of Japan 55: 245,1989.
- Ohira, Namba, Rozanov, Kusumi & Tsuchizaki, Journal of General Virology 76: 2305, 1995.
- Ohki, Yoshikawa, Inouye & Inouye, Annals of the Phytopathological Society of Japan 55: 245, 1989.
- Roistacher, Proceedings of the 10th Conference of the International Organization of Citrus Virologists : 353, 1988.
- Semancik & Weathers, Phytopathology 55: 1354, 1965.
- Su & Cheon, Phytopathologist and Entomologist, NTU 11: 42, 1984.
- Terauchi, Magome, Yoshikawa, Takahashi & Inouye, Annals of the Phytopathological Society of Japan 63: 432, 1997.
- Uyemoto & Gilmer, Annals of Applied Biology 69: 17, 1971.
- Wallace & Drake, Plant Disease Reporter 46: 211, 1962.
- Wallace & Drake, Proceedings of the 4th Conference of the International Organization of Citrus Virologists : 171, 1968.
- Waterworth & Gilmer, Phytopathology 59: 334, 1969.
- Yanase, Bulletin of the Fruit Tree Research Station C 1: 47, 1974.
- Yanase, Acta Horticulturae 130 : 117, 1982.
- Yanase, Nakatani, Munakata & Machida, Bulletin of the Fruit Tree Research Station A 13: 69, 1986.
- Yoshikawa & Takahashi, Journal of General Virology 69: 241, 1988.
- Yoshikawa & Takahashi, Journal of General Virology 73: 1313, 1992.
- Yoshikawa, Sasaki, Kato & Takahashi, Virology 191: 98, 1992.
- Yoshikawa, Imaizumi, Takahashi & Inouye, Journal of General Virology 74: 2743, 1993.
- Yoshikawa, Sasamoto, Sakurada, Takahashi & Yanase, Annals of the Phytopathological Society of Japan 62: 119, 1996.
- Yoshikawa, Iida, Goto, Magome, Takahashi & Terai, Archives of Virology 142: 1351, 1997.
- Zhang, Liang & Roistacher, Plant Disease 72: 543, 1988.