Details of DPV and References
DPV NO: 382 December 2001
Family: Tombusviridae
Genus: Tombusvirus
Species: Tomato bushy stunt virus | Acronym: TBSV
This is a revised version of DPV 69
Tomato bushy stunt virus
G. P. Martelli Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
M. Russo Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
L. Rubino Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Smith (1935), Ainsworth (1936) and Bawden & Pirie (1938).
- Synonyms
- Lycopersicum virus 4 (Smith, 1937)
- Marmor dodecahedron (Breed et al., 1948)
- Lycopersicumvirus dodecahedrum (Roland, 1959)
- Virus del rachitismo cespuglioso del pomodoro (Gigante, 1954)
- Tomatenzwergbusch-Virus (Schmelzer, 1958)
- Marmor dodecahedron (Breed et al., 1948)
A soil-borne virus with isometric particles about 30 nm in diameter and rounded outline, occurring in economically important crops. Virus particles contain one major linear positive sense, ssRNA species of c. 4.7 kb and a single coat protein of Mr 41,000. The virus is readily transmitted by mechanical inoculation to a wide range of experimental hosts. Natural transmission is through seed and soil, apparently without a vector. The biological, physicochemical, ultrastructural, and molecular properties of the virus have been reviewed frequently (Martelli et al., 1977; Martelli, 1981; Martelli et al., 1988; Martelli et al., 1989; Russo et al., 1994).
Main Diseases
The virus causes stunting and bushy growth, chlorotic spots, crinkling, deformation and necrosis of leaves of tomato (Pontis et al., 1968; Fischer & Lockhart, 1977; Borges et al., 1979; Cherif & Spire, 1983; Gerik et al., 1990; Luis-Arteaga et al., 1996) (Fig. 1) and pepper (Cherif & Spire, 1983). Fruit setting of both crops may be drastically reduced. Fruits are smaller than normal and show chlorotic blotching, rings, line patterns, and necrosis (Fig. 2, Fig. 3) that lower the economic value of the crop, or make it unmarketable. Yield losses in tomato can be as high as 80% (Gerik et al., 1990). In addition, the virus induces stunting, leaf yellowing and mottling, poor fruit setting, and deformation and necrosis of the fruits in eggplant (Cherif & Spire, 1983; Koenig & Avgelis, 1983; Luis-Arteaga et al., 1996) (Fig. 4, Fig. 5); mosaic, leaf malformation, necrosis and sometimes death of statice (Goniolimon tataricum) (Krczal & Beutel, 1994); extensive necrosis of the leaves and petals of tulip (Mowat, 1972); stunting and mild mottling of the leaves in piggyback (Tolmiea menziesii) (Henriques & Schelegel, 1978); and fruit pitting, veinal necrosis and stunting in cherry (Allen & Davidson, 1967). The virus is also reported from apple (Allen, 1969). Symptom expression, especially in protected vegetable crops, is strongly influenced by photoperiod and temperature (Martinez et al., 1974; Borges et al., 1979; Hillman et al., 1985; Gerik et al., 1990).
Geographical Distribution
Reported from several European countries, North and South America, and North Africa (summarized by Martelli et al., 1988).
Host Range and Symptomatology
The virus has a relatively restricted natural host range, comprising primarily vegetables and ornamentals. Infection of woody plants is much less common. However, the soil-borne nature of the virus and its tendency to remain localized in tissues, make it likely that a wider number of cultivated and wild plants are infected locally in the roots (Kegler & Kegler, 1980; Cherif, 1981). The experimental host range is wide. The type strain of the virus was reported to infect 52 of 157 species (Schmelzer, 1958) and 45 of 62 species (Hollings & Stone, 1965) and the BS3 strain, 33 of 48 species (Cherif & Spire, 1983). In most plants, infection is localized. Virus isolates are readily transmitted between hosts by mechanical inoculation of infective sap in 0.1M neutral phosphate buffer.
Diagnostic species
Gomphrena globosa. Necrotic local lesions in two days, which rapidly enlarge to give red-rimmed whitish rings or spots.
Ocimum basilicum. Dark brown necrotic local lesions with lighter centres (Fig. 6)
Nicotiana benthamiana and N. clevelandii. Chlorotic local lesions in inoculated leaves in 2-3 days, followed in 3-4 days by systemic mottling and severe distortion of the leaves, and, after an additional 4-5 days, apical necrosis and death. If defective-interfering (DI) RNA is present in the inoculum or is generated de novo during infection, plants fail to develop apical necrosis and death, and remain chronically infected.
Propagation species
Assay species
Strains
Three major strains, all pathogenic to commercially cultivated plants, are known: (i) the type strain obtained originally from infected tomato plants in England (Smith, 1935); (ii) the BS3 strain, which was derived from the type strain by serial passages through differential hosts (Steere, 1953), and (iii) the cherry strain (TBSV-Ch) (Allen & Davidson, 1967; Allen 1968). The type and the BS3 strains usually infect herbaceous hosts, whereas TBSV-Ch has the tendency to infect woody plants. Strain differentiation based on the responses of herbaceous hosts is difficult (Martelli et al., 1988). Although the strains are closely related serologically (serological differentiation index 1 to 3), they are distinguishable (Koenig & Gibbs, 1986). The cherry strain is closely related serologically to Petunia asteroid mosaic virus (PAMV) and Artichoke mottled crinkle virus (AMCV). Because of this and of the high sequence homology in their genomes, the suggestion was made that PAMV and AMCV are also strains of TBSV (Luis-Arteaga et al. 1996). In recent years, no natural outbreaks of the type strain of TBSV have been reported, all new records being of TBSV-BS3 (Koenig & Avgelis, 1983; Gerik et al., 1990; Louis-Arteaga et al., 1996) or, in a single instance, to TBSV-Ch (Louis-Arteaga et al., 1996).
Transmission by Vectors
No vector, either aerial or soil-inhabiting, is known. The virus is not transmitted by aphids, either non-persistently or semi-persistently (Smith, 1935; Koenig & Avgelis, 1983), mites (Orlob, 1968), or the chytrid fungus, Olpidium brassicae (Campbell, 1968).
Transmission through Seed
The virus can be transmitted with variable efficiency (4-65%) through seed of pepper, tomato and apple (Allen, 1969; Cherif 1981; Tomlinson & Faithfull, 1984). It was also found in seed and pollen of sweet cherry (Allen & Davidson, 1967; Kegler & Kegler, 1980) but no transmission was obtained to cherry trees hand-pollinated with infected pollen (Allen & Davidson, 1967).
Transmission by Grafting
The virus is transmitted by grafting in vegetatively propagated crops (e.g. cherry) (Kegler & Schimanski, 1982).
Transmission by Dodder
Cuscuta campestris can transmit the type strain (Johnson, 1941).
Serology
The virus is a good immunogen. Antisera with titres of 1:512 to 1:2056 or higher are obtained readily in rabbits with immunization schedules based on two intramuscular injections one week apart, the first emulsified in Freund's complete adjuvant, and the second in Freund's incomplete adjuvant (Koenig & Gibbs, 1986; Hollings & Stone, 1975). Virus particles do not require stabilization by fixatives prior to injection into animals. Antisera give a single precipitin line in agar gel double diffusion tests and decorate virus particles uniformly in immuno-electron microscopy tests (Appiano & D'Agostino, 1985). ELISA can be used for diagnostic purposes (Fuchs et al., 1979; Koenig & Kunze, 1982; Cherif & Spire, 1983; Gerik et al., 1990) with a sensitivity 5000x higher than in gel double diffusion tests (Cherif, 1981).
Relationships
The virus is the type species of the genus Tombusvirus in the family Tombusviridae (Russo et al., 1994). In pairwise comparisons in agar gel double diffusion tests the precipitin lines of all three different virus strains formed a spur at the junction with one another. The virus is distantly related serologically with several other virus species in the genus (Jaegle & Van Regenmortel, 1985; Koenig & Gibbs, 1986). ELISA was also used for measuring the serological cross-reactivity between this virus species and other species of the genus (Jaegle & Van Regenmortel, 1985). Nucleic acid hybridization with random primed cDNA probes was used for assessing the relationship between the virus and other members in the genus (Gallitelli et al., 1985; Koenig & Burgermeister, 1988) and for detecting genomic RNA, DI-RNA, and satellite RNAs in total RNA samples from leaves of naturally infected plants (Celix et al., 1997).
The genome organization is identical to that of members in the genus Aureusvirus, but differences exist in the overall genome size (4.7 versus 4.4 kb) and in the size and sequence homology of the polymerase cistron (Rubino & Russo, 1997). The strategy of expression, is similar to that of members of other genera in the family (Russo et al., 1994). The polymerase shows significant protein sequence similarity with comparable proteins of members of the other genera in the family, as well as with that of the genera Enamovirus and Luteovirus (Russo et al., 1994).
Stability in Sap
Depending on the host from which the sap is expressed and the virus strain, the virus loses infectivity when heated for 10 min between 78 and 90 °C, diluted at 10-4-10-7, and stored at room temperature between 3 and 8 weeks (Smith, 1935; Hollings & Stone, 1965; Cherif, 1981; Koenig & Avgelis, 1983; Tomlinson & Faithful, 1984). Infectivity is retained over the pH range 2 and 10 (Allen & Davidson, 1967). There is little or no loss of infectivity when sap from infected plants is frozen or is freeze-dried by vacuum sublimation, and the sealed ampoules are stored in the dark at room temperature for up to six years (Hollings & Stone, 1965).
Purification
Homogenize systemically infected leaves of N. benthamiana or N. clevelandii collected 7-10 days after inoculation in 2-3 vol of 0.1 M sodium acetate buffer, pH 5.5, containing 0.25% b-mercaptoethanol. Strain through cheesecloth, keep the extract on ice for 30-60 min, and clarify by low speed centrifugation. Add 8% PEG (mol. wt 6000) and 0.2 M NaCl to the supernatant fluid and submit to one or two cycles of differential centrifugation. Resuspend the final high speed pellet in 0.02 M sodium acetate, pH 5.5 (Burgyan and Russo, 1998). Virus yields are very high, regardless of the purification method used, e.g. up to 0.1 mg/g of Nicotiana leaf tissue (Burgyan & Russo, 1998) or 0.5 g/l of tomato sap (Kassanis & Lebeurier, 1969).
RNA is extracted from virus particles by adding one volume of buffer (100 mM glycine-NaOH, pH 9.0, containing 100 mM NaCl, 10 mM EDTA, 2% sodium dodecyl sulfate, and 1% sodium lauryl sarcosine) and two volumes of phenol. The aqueous phase is extracted further with an equal volume of phenol and chloroform, washed with chloroform, and precipitated with 2.5 vol. of ethanol. RNA is resuspended in sterile water (Burgyan and Russo, 1998).
Properties of Particles
Molecular weight: 9.3 x 106 (Weber et al., 1970).
Sedimentation coefficient (s20,w): 132S at infinite dilution (Hollings & Stone, 1965).
Buoyant density in CsCl: c. 1.35 g.cm-3 (Mayo & Jones, 1973).
Diffusion coefficient (D20x10-7 cm2.sec-1): 1.26 (Schachman & Williams, 1959).
Isoelectric point: pH 4.1 (McFarlane & Kekwick, 1938).
Partial specific volume: 0.71 cm3.g-1 (Schachman & Williams, 1959).
Electrophoretic mobility: -4.65 x 105 cm2. sec-1.V-1 at pH 8.2 in veronal-HCl buffer ionic strength 0.02 (Ambrosino et al., 1967).
Zone electrophoretic RØ value: 0.34 (Quacquarelli et al., 1966).
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 4.5 (Ambrosino et al., 1967).
A260/A280: 1.66 (Ambrosino et al., 1967).
P content: 1.54% (Ambrosino et al., 1967).
Particle Structure
The virus has isometric particles about 30 nm in diameter. In phosphotungstic acid, the particles show an angular outline and unresolved surface structure (Quacquarelli et al., 1966; Lovisolo et al., 1967) but when mounted in uranyl acetate, they exhibit a rounded outline and somewhat knobby surface and edges (Francki et al., 1985) (Fig. 7). The protein shell is constructed of 180 subunits, each with Mr 41,000 arranged in a T=3 icosahedral surface lattice. The subunits show a dimeric clustering at the surface of the particles giving rise to 90 morphological units located on the two-fold axes of the lattice (Finch et al., 1970; Crowther & Amos, 1971; Harrison et al., 1978) (Fig. 8). Each coat protein subunit contains 387 amino acid residues (Hopper et al., 1984; Hillman et al., 1989) and has four regions: an internal positive charged domain (R) comprising 66 residues which, together with an "arm" 36 residues long, constitutes the N-terminal region of the polypeptide. This region is flexibly linked to two distinct globular parts, of which one (domain S, 168 residues) forms the shell of the virus particle and the other (domain P, 127 residues) protrudes on the surface of the particle. The P domains are clustered in pairs giving rise to the 90 projections that constitutes the units contrasted by negative staining in the electron microscope. Because of their flexibility, the coat polypeptides can assume three distinct packing orientations, designated A, B and C (Harrison et al., 1978; Olson et al., 1983; Harrison, 1984; Hopper et al., 1984). The connecting arms of the 60 subunits in orientation C are folded in an orderly manner at the bottom of the respective S domains, interlocking to form an internal framework which determines particle size (Olson et al., 1983). The N-termini of the remaining 120 subunits in orientations A and B, and the positively charged residues of the inward-facing S domain surfaces, are potential sites for RNA binding (Harrison et al., 1978; Chauvin et al., 1978; Hopper et al., 1984) (Fig. 8).
Particle stability depends on the P domain dimer contacts, which are very stable, and on trimer interactions between the S domains of A, B and C subunits. The strength of these latter contacts is enhanced by Ca++ ions (two for each pair of interacting subunits), but not Mg++ ions (Robinson & Harrison, 1982; Kruse et al., 1982; Hogle et al., 1983). Removal of Ca++ at about pH 7 induces reversible swelling of the particles. These are not sensitive to RNase, but are partially sensitive to protease (Robinson & Harrison, 1982; Kruse et al., 1982). The genomic RNA is tightly packed within the protein shell, most of it probably being located in the space between the S domains and the internal concentric shell made up of the N-terminal regions (Chauvin et al., 1978).
Particle Composition
Nucleic acid. Virus particles contain a single species of linear, positive sense, single-stranded RNA, accounting for about 17% of the particle weight. The RNA has a mol. wt of c. 1.67 x 106. Molar percentages of nucleotides are reported as: G28.6, A26.3, C21.2, U26.3 (De Fremery & Knight, 1955) or G27.7, A27.0, C20.7, U24.5 (Ambrosino et al., 1967).
Protein. The viral capsid accounts for about 83% of the particle weight and is composed of a single polypeptide of Mr 42,000 calculated by polyacrylamide gel electrophoresis (Gallitelli & Russo, 1987), or Mr 41,000, as deduced from the amino acid sequence of the coat protein gene (Hearne et al., 1990).
Genome Properties
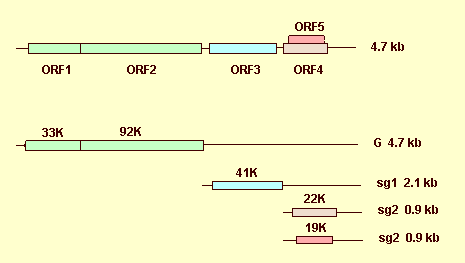
The viral genome is monopartite and that of TBSV-Ch has been completely sequenced and shown to contain 4,776 nucleotides (accession No. M21958) (Hearne et al., 1990). The genome contains five functional ORFs. ORF 5 is completely nested in ORF 4, in a different reading frame (Fig. 9). The 5' terminus does not have a VPg (Mayo et al., 1982) nor, by analogy with the related Carnation Italian ringspot virus, a cap (Russo et al., 1994), and the 3' terminus is not polyadenylated. The 5' region initiates with an untranslated sequence (UTR) of 165 nt, and the 3' region terminates with an UTR of 351 nt. The 5'-proximal ORF1 (nt 166-1056) codes for a 33 kDa protein. Readthrough of the amber stop codon of ORF1 results in the translation on a 92 kDa fusion protein (ORF 2, nt 166-2622) (Fig. 9). The 33 kDa protein does not contain motifs equivalent to any known viral function, but the readthrough domain in the 92 kDa protein contains the conserved motifs of the RNA-dependent RNA polymerase of positive strand RNA viruses. ORF 3 (nt 2652-3818) encodes the coat protein (41 kDa). The protein encoded by ORF 4 (nt 3856-4425) (22 kDa) is required for cell-to-cell spread of the virus (Scholthof et al., 1993) (Fig. 9). The 19 kDa protein encoded by ORF5 (nt 3888-4406) may be involved in the induction of necrotic symptoms (Scholthof et al., 1995a) and in the long-distance spread of the virus, depending on the host (Scholthof et al., 1995b). The strategy of expression is based on direct translation from genomic RNA of the 5' proximal genes (33 and 92 kDa proteins) and translation of the downstream genes through two 3' co-terminal subgenomic RNAs of 2.1 kb (coat protein) and 0.9 kb in size (22 and 19 kDa proteins) (Hearne et al., 1990). dsRNAs isolated from infected N. clevelandii were: (i) two species of 4.7 and 2.2 kbp, identified as the replicative forms of the viral genome and of a subgenomic RNA (Henriques & Morris, 1979); (ii) several species, one of 4.8 kbp (RF of genomic RNA) and three of 1.8 to 1.1 kbp, interpreted as possible RF forms of subgenomic RNAs (Hayes et al., 1984).
Satellite
The virus supports the replication of a linear satellite RNA (satRNA) lacking coding capacity, and with a size of about 0.6 kb (Gallitelli & Hull, 1985). Virus particles of Spanish isolates from tomato and eggplant were found to contain two types of satRNA of 0.8 and 0.6 kb (Celix et al., 1997), whereas a single species of satRNA of 0.6 kb was encapsidated by particles of the statice isolate from Germany (D. Galetzka & M. Russo, unpublished data). Comparison of the sequences of the three satRNAs and of the satRNA associated with Cymbidium ringspot virus infections (Rubino et al., 1990) showed the lack of substantial homology with the viral genome, except for the 5'- and 3'- termini and for a central block of about 50 nt. This latter sequence is conserved in the genome of all members of the genus Tombusvirus and in the defective interfering RNAs (DI-RNA) (Rubino et al., 1995) and is thought to be a signal necessary for RNA replication (Russo et al., 1994). SatRNAs are not thought to interfere greatly with virus symptom expression.
DI-RNAs are smaller than genomic molecules and are generated de novo in the course of infection of experimentally inoculated hosts through recombination events whereby fragments of the viral genome are deleted stepwise (White & Morris, 1994). Their presence interferes with virus replication, thus modulating symptoms. Four regions of the viral genome are conserved in DI-RNAs: the 5' leader sequence of 168 nt; 200-250 nt from the polymerase gene; about 70 nt from the 22 kDa and 19 kDa cistrons; and about 130 nt from the 3' terminal noncoding region (Knorr et al., 1991). DI-RNAs of about 0.4 kb and 0.7 kb were found associated with TBSV-Ch in infected N. clevelandii (Hillman et al., 1987; Celix et al., 1997). A DI-RNA about 0.4 kb in size was identified also in naturally infected statice plants (Galetzka et al., 2000), which may account for the variability of symptoms observed in the field (Krczal & Beutel, 1994).
Relations with Cells and Tissues
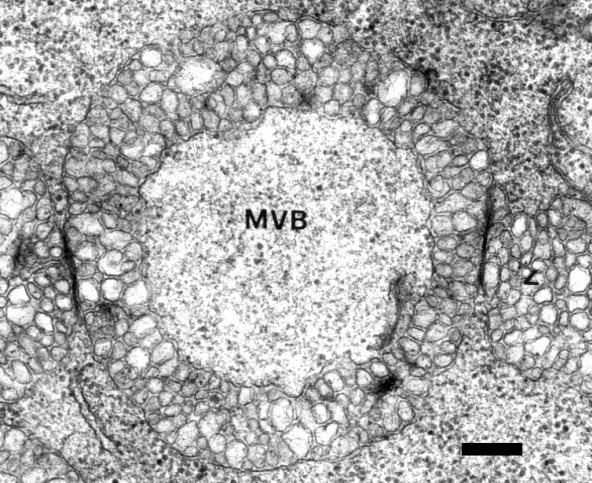
With the noteworthy exception of N. benthamiana and N. clevelandii, the virus tends to remain localized in experimentally inoculated hosts. Lesions induced in inoculated leaves of G. globosa consist of a necrotic centre, surrounded by darkened necrotic tissues and a translucent yellowish halo, which becomes reddish 4-5 days after inoculation (Appiano et al., 1977, 1981; Pennazio et al., 1978, 1979). The centre of a local lesion is made up of collapsed necrotic cells, filled with starch grains and containing many virus particles. The cytology of the cells of the halo differs according to whether its inner, central, or outer part is examined. In the inner part, necrotic cells are interspersed with apparently active ones, which, nevertheless, have a much deranged cytology with cell walls thickened by callose and suberin depositions. The cells of the central part of the halo contain virus particles and cytopathological structures denoted as multivesicular bodies (MVBs), whereas the cells of the outer part of the halo are little affected. The tissues surrounding the lesions have a normal cytology, but are physiologically modified, possessing a high level of localized acquired resistance, which decreases progressively with increasing distance from the lesion (Pennazio and Redolfi, 1977). The virus invades all tissues of systemically infected plants. The cytopathology of systemically infected tissues was studied in Datura stramonium (Russo & Martelli, 1972), G. globosa (Appiano et al., 1981; Martelli et al., 1984) and N. benthamiana (Galetzka et al., 2000). The intracellular hallmark of viral infection is the occurrence of MVBs that consist of a main body surrounded by many spherical to ovoid vesicles 80-150 nm in diameter originating as invaginations of the boundary membrane of peroxisomes (Fig. 10). The vesicles contain dsRNA (Appiano et al., 1983), possibly representing replicative forms of the viral genome. This observation, and the finding that the proteins of the replicase complex are associated with membranous cell components (Scholthof et al., 1995b) suggest that MVBs are the site of virus replication.
Virus particles are occasionally found in the nucleus and are abundant in the cytoplasm, where they may form crystalline inclusions (Russo & Martelli, 1972; Appiano et al., 1978). The cytoplasm of systemically infected cells contains small patches of electron-dense amorphous material, which was identified, by immuno-gold labelling, as excess virus coat protein (Appiano et al., 1985).
Ecology and Control
The virus is firmly established in certain soils, especially clays, where it persists in an infective form for up to five months (Kegler & Kegler, 1980; Cherif, 1981) resisting high temperatures (e.g. autoclaving at 121 °C for 2h) (Kegler & Kegler, 1980), and from which it is readily acquired by bait plants apparently without the mediation of a vector (summarized by Martelli et al, 1988). Transplanting healthy seedlings in soils that previously contained infected crops, or were made infective by the addition of infected plant sap or by watering with virus suspensions, resulted in infection with incidences from 10 to 100% (Roberts, 1950; Kegler et al., 1980; Kegler & Kegler, 1981; Cherif, 1981; Tomlinson & Faithful, 1984; Gerik et al., 1990). Routes for virus entry into the environment are: (i) crop debris (e.g. roots and leaves) containing high concentrations of virus particles; (ii) living roots of infected hosts releasing virus, as suggested by the presence of virus particles in soil leachates (Smith et al., 1969; Kegler & Kegler, 1981; Cherif, 1981; Tomlinson & Faithful, 1984); (iii) irrigation water from lakes and rivers containing infective virus (Tomlinson & Faithful, 1984; Koenig & Lesemann, 1985). The virus may enter rivers from drainage water of soils where infected crops are grown or sewage waters, as the virus passes unharmed through the alimentary tract of humans following consumption of infected raw vegetables (Tomlinson et al., 1982).
The virus is very labile in vivo, notwithstanding its high in vitro thermal inactivation point (> 90 °C). Whole plants of tomato or N. clevelandii were freed from the virus by exposure to 36-38 °C for 3-4 weeks (Kassanis, 1954; Hollings & Stone, 1965; Kassanis & Lebeurier, 1969; McCarthy, 1983). Virus inactivation was due to the cleavage of viral RNA within particles, caused possibly by the increase in concentration of free radicals at high temperatures (McCarthy, 1983).
Notes
Symptoms induced by the virus in some experimental hosts resemble those elicited by members of the genus Necrovirus, which makes diagnosis on this basis difficult. However, none of the TBSV strains is related serologically to any of the necroviruses. A major problem exists in the unambiguous identification of individual species in the cluster comprising TBSV-type, TBSV-BS3, TBSV-Ch, PAMV and AMCV. TBSV-Ch and PAMV were identified as isolates of the same species (PAMV) because of the similarity of their serological properties (Koenig & Kunze, 1982) and biological behaviour (Martelli et al., 1988). This synonymy was not unanimously accepted, so that the available molecular information on "TBSV", comes from studies in which an isolate of TBSV-Ch was used (Hearne et al., 1990, Scholthof et al., 1993, 1995a, 1995b). Recent comparative studies have disclosed molecular similarities among viruses of the TBSV/PAMV/AMCV cluster, indicating that TBSV-Ch is a bona fide strain of TBSV (Louis-Arteaga et al., 1996). This was taken into consideration in the present description of this virus. The taxonomic problem regarding these viruses, however, warrants more extensive comparative investigations.
Acknowledgements
We thank F. Garcia-Arenal and M. Luis-Arteaga for the pictures of virus-infected plants.
Figures

Chlorotic blotching and deformation of fruits from a naturally infected tomato plant (courtesy of M. Luis-Arteaga).

Extensive necrosis in a fruit from a naturally infected tomato plant (courtesy of F. Garcia-Arenal).

Severe deformation and extensive necrosis shown by fruits from naturally infected eggplant plants. A healthy fruit on the left (courtesy of F. Garcia-Arenal).

Necrotic local lesions typically induced by TBSV and other tombusviruses in basil (Ocymum basilicum) (courtesy of M. Luis-Arteaga).

Diagrammatic representation of a TBSV particle. A, B and C refer to coat protein subunits arranged in different orientations. See text for details (from Hopper et al., 1984).
References list for DPV: Tomato bushy stunt virus (382)
- Ainsworth, Journal of the Ministry of Agriculture and Fisheries 43: 266, 1936.
- Allen, Canadian Journal of Botany 46: 229, 1968.
- Allen, Canadian Journal of Plant Science. 49: 797, 1969.
- Allen & Davidson, Canadian Journal of Botany 45: 2375, 1967.
- Ambrosino, Appiano, Rialdi, Papa, Redolfi & Carrara, Atti dell' Accademia delle Scienze, Torino 101: 301, 1967.
- Appiano & D'Agostino, Journal of Ultrastructure Research 85: 239, 1985.
- Appiano, Pennazio, D'Agostino & Redolfi, Physiological Plant Pathology 11: 327, 1977.
- Appiano, Pennazio & Redolfi, Journal of General Virology 40: 277, 1978.
- Appiano, D'Agostino, Redolfi & Pennazio, Journal of Ultrastructure Research 76: 173, 1981.
- Appiano, Bassi & D'Agostino, Ultramicroscopy 12: 162, 1983.
- Appiano, Barbieri, Bassi, D'Agostino, dell'Orto & Viola, Atti 15° Congresso Nazionale di Microscopia Elettronica A3, 1985.
- Bawden & Pirie, British Journal of Experimental Pathology 19: 251, 1938.
- Borges, Sequeira & Louro, Phytopathologia Mediterranea 18: 118, 1979.
- Breed, Murray & Hitchens, Bergey's Manual of Determinative Bacteriology, 6th edition, Baillere, Tindall & Cox, London, 1529 pp., 1948.
- Burgyan & Russo, Methods in Molecular Biology 81: 225, 1998.
- Campbell, Plant Disease Reporter 52: 379, 1968.
- Celix, Rodriguez-Cerezo & Garcìa-Arenal, Virology 239: 277, 1997.
- Chauvin, Witz & Jacrot, Journal of Molecular Biology 124: 641, 1978.
- Cherif, Thèse Diplome Docteur Troisieme Cycle, Université P. & M. Curie, Paris, 84 pp., 1981.
- Cherif & Spire, Agronomie 3: 701, 1983.
- Crowther & Amos, Cold Spring Harbor Symposium on Quantitative Biology No. 36: 489, 1971.
- De Fremery & Knight, Journal of Biological Chemistry 214: 559, 1955.
- Finch, Klug & Leberman, Journal of Molecular Biology 50: 215, 1970.
- Fischer & Lockhart, Phytopathology 67: 1352, 1977.
- Francki, Milne & Hatta, Atlas of Plant Viruses 1: 222, Boca Raton, CRC Press, 1985.
- Fuchs, Marker & Kegler, Archiv für Phytopathologie und Pflanzenschutz 15: 421, 1979.
- Galetzka, Russo, Rubino & Krczal, Journal of Plant Pathology 82: 151, 2000.
- Gallitelli & Hull, Journal of General Virology 66: 1533, 1985.
- Gallitelli & Russo, Journal of Phytopathology 119: 106, 1987.
- Gallitelli, Hull & Koenig, Journal of General Virology 66: 1523, 1985.
- Gerik, Duffus, Perry, Stenger & Van Maren, Phytopathology 80: 1352, 1990.
- Gigante, Bolettino della Stazione di Patologia Vegetale, Roma 12 (S. III): 43, 1954.
- Harrison, Trends in Biochemical Science 9: 345, 1984.
- Harrison, Olson, Schutt, Winkler & Bricogne, Nature, London 276: 368, 1978.
- Hayes, Buck & Brunt, Journal of General Virology 65: 1239, 1984.
- Hearne, Knorr, Hillman & Morris, Virology 177: 141, 1990.
- Henriques & Morris, Virology 99: 66, 1979.
- Henriques & Schlegel, 3rd International Congress of Plant Pathology, Munchen 1978: 43, 1978.
- Hillman, Morris & Schlegel, Phytopathology 75: 361, 1985.
- Hillman, Carrington & Morris, Cell 51: 427, 1987.
- Hillman, Hearne, Rochon & Morris, Virology 169: 42, 1989.
- Hogle, Kirchhausen & Harrison, Journal of Molecular Biology 171: 95, 1983.
- Hollings & Stone, Annals of Applied Biology 56: 87, 1965.
- Hollings & Stone, Annals of Applied Biology 80: 37, 1975.
- Hopper, Harrison & Sauer, Journal of Molecular Biology 177: 701, 1984.
- Jaegle & Van Regenmortel, Journal of Virological Methods. 11: 189, 1985.
- Johnson, Phytopathology 31: 649, 1941.
- Kassanis, Annals of Applied Biology 41: 470, 1954.
- Kassanis & Lebeurier, Journal of General Virology 4: 385, 1969.
- Kegler & Kegler, Tagungs Bericht Akademie f ür Landwirtschaft -Wissenschaft DDR 184: 297, 1980.
- Kegler & Kegler, Archiv für Phytopathologie und Pflanzenschutz 17: 307, 1981.
- Kegler & Schimanski, Archiv für Phytopathologie und Pflanzenschutz 16: 73, 1982.
- Kegler, Kleinhempel & Kegler, Archiv für Phytopathologie und Pflanzenschutz 14: 73, 1980.
- Knorr, Mullin, Hearne & Morris, Virology 181: 193, 1991.
- Koenig & Avgelis, Phytopathologische Zeitschrift 106: 349, 1983.
- Koenig & Burgermeister, Journal of Virological Methods 19: 57, 1988.
- Koenig & Gibbs, Journal of General Virology 67: 75, 1986.
- Koenig & Kunze, Phytopathologische Zeitschrift 103: 361, 1982.
- Koenig & Lesemann, Phytopathologische Zeitschrift 112: 105, 1985.
- Krczal & Beutel, Acta Horticulturae 377: 115, 1994.
- Kruse, Kruse, Witz, Chauvin & Jacrot, Journal of Molecular Biology 162: 393, 1982.
- Lovisolo, Ambrosino, Luisoni & Bellaudo, Atti Accademia delle Scienze. Torino 101: 299, 1967.
- Luis-Arteaga, Rodriguez-Cerezo, Fraile, Saez & Garcia-Arenal, Phytopathology 86: 535, 1996.
- Martelli, in Handbook of Plant Virus Infections and Comparative Diagnosis, p. 61, ed E. Kurstak, Amsterdam: Elsevier/North Holland, 1981.
- Martelli, Russo & Quacquarelli, in The Atlas of Insect and Plant Viruses, p.257, ed. K. Maramorosch, New York: Academic Press, 1977.
- Martelli, Di Franco & Russo, Journal of Ultrastructure Research 88: 275, 1984.
- Martelli, Gallitelli & Russo, in The Plant Viruses vol. 3. Polyhedral virions with Monopartite RNA Genomes, p. 13, ed. R. Koenig. New York: Plenum Press, 1988.
- Martelli, Russo, & Gallitelli, A.A.B. Descriptions of Plant Viruses 353, 1989.
- Martinez, Galindo & Rodriguez, Agrociencia 18: 71, 1974.
- Mayo & Jones, Journal of General Virology 19: 245, 1973.
- Mayo, Barker & Harrison, Journal of General Virology 59: 149, 1982.
- McCarthy, Physiological Plant Pathology 22: 181, 1983.
- McFarlane & Kekwick, Biochemical Journal 32: 1607, 1938.
- Mowat, Plant Pathology 21: 71, 1972.
- Olson, Harrison & Bricogne, Journal of Molecular Biology 171: 61, 1983.
- Orlob, Virology 35: 121, 1968.
- Pennazio & Redolfi, Rivista di Patologia Vegetale. (S.IV) 13: 103, 1977.
- Pennazio, D'Agostino, Appiano & Redolfi, Physiological Plant Pathology 13: 165, 1978.
- Pennazio, Appiano & Redolfi, Physiological Plant Pathology 15: 177, 1979.
- Pontis, Garcia & Feldman, Plant Disease Reporter 52: 676, 1968.
- Quacquarelli, Martelli & Russo, Atti del Primo Congresso dell'Unione Fitopatologica Mediterranea Bari 1966, 1: 163, 1966.
- Roberts, Annals of Applied Biology 37: 385, 1950.
- Robinson & Harrison, Nature, London 297: 563, 1982.
- Roland, Taxon 8: 126, 1959.
- Rubino & Russo, Journal of General Virology 78: 1219, 1997.
- Rubino, Burgyan, Grieco & Russo, Journal of General Virology 71: 1655, 1990.
- Rubino, Burgyan & Russo, Archives of Virology 140: 2027, 1995.
- Russo & Martelli, Virology 49: 122, 1972.
- Russo, Burgyan & Martelli, Advances in Virus Research 44: 381, 1994.
- Schachman & Williams, in The Viruses, ed. F. M. Burnett & W. M. Stanley, Academic Press, New York, 1959.
- Scholthof, Morris & Jackson, Molecular Plant-Microbe Interactions 6: 309, 1993.
- Scholthof, Scholthof & Jackson, Plant Cell 7: 1157, 1995a.
- Scholthof, Scholthof, Kikkert & Jackson, Virology 213: 425, 1995b.
- Schmelzer, Zeitschrift für PflanzenKrankeit und PflanzenSchutz 65: 80, 1958.
- Smith, Annals of Applied Biology 22: 731, 1935.
- Smith, A Textbook of Plant Virus Diseases, J. & A. Churchill, London, 615 pp., 1937.
- Smith, Campbell & Fry, Phytopathology 59: 1678, 1969.
- Steere, Phytopathology 43: 485, 1953.
- Tomlinson & Faithfull, Annals of Applied Biology 104: 485, 1984.
- Tomlinson, Faithful, Flewett & Beards, Nature, London 300: 637, 1982.
- Weber, Rosenbusch & Harrison, Virology 41: 763, 1970.
- White & Morris, Journal of Virology 68: 6092, 1994.