Details of DPV and References
DPV NO: 384 December 2001
Family: Bromoviridae
Genus: Oleavirus
Species: Olive latent virus 2 | Acronym: OLV-2
Olive latent virus 2
G. P. Martelli Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
F. Grieco Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
V. Savino Dipartimento di Protezione delle Piante, Università degli Studi and Centro di Studio del CNR sui Virus e le Virosi delle Colture Mediterranee, Bari, Italy.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Savino et al. (1984); Grieco et al. (1992).
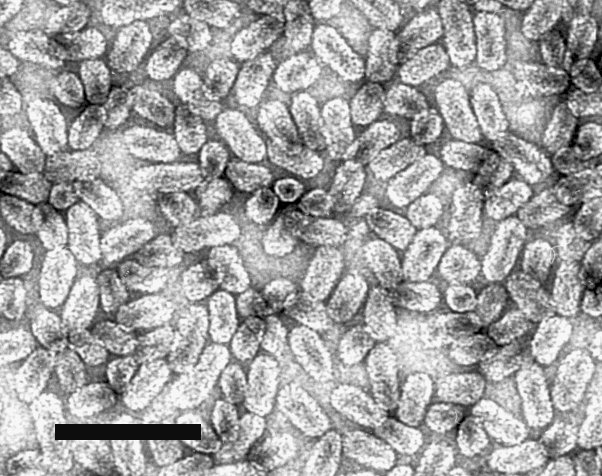
A virus with particles that are quasi-isometric about 26 nm in diameter and bacilliform 37, 43, 48 and 55 nm long and 18 nm wide. The virus has a linear tripartite positive-sense ssRNA genome. Each particle contains about 19% RNA and has a single protein species of Mr c. 20,000. Geographical distribution and natural host range are very restricted.
Main Diseases
The virus infects olive (Olea europaea) symptomlessly (Savino et al., 1984), and induces mottling and vein clearing in the leaves of castor bean (Ricinus communis) (C. Vovlos, personal communication).
Geographical Distribution
Recorded from Apulia (Southern Italy) and Greece.
Host Range and Symptomatology
Olive and castor bean are the only known natural hosts (Savino et al., 1984; C. Vovlos, personal communication). The virus is transmitted by mechanical inoculation of sap from olive leaves or young fruits in 0.1M phosphate buffer pH 7, containing 2.5% nicotine. The reported experimental host range consists of 10 plant species in four different families (Savino et al., 1984; Grieco et al., 1992).
Diagnostic species
N. clevelandii and N. occidentalis. Chlorotic/necrotic lesions in uninoculated leaves.
Chenopodium quinoa. Necrotic local lesions.
Gomphrena globosa. Reddish local lesions with a necrotic centre 5-6 days after inoculation followed by systemic mottling and distortion of leaves.
Propagation species
Assay species
Strains
None reported.
Transmission by Vectors
The virus was not transmitted in a non-persistent manner by the aphid Myzus persicae to N. benthamiana (F. Grieco, unpublished information).
Transmission through Seed
The virus was not seed-transmitted in G. globosa (F. Grieco, unpublished information).
Transmission by Grafting
The virus persists in budwood; transmission by grafting is therefore likely to occur in olive.
Serology
The virus is poorly immunogenic giving antisera with titres up to 1/256. Antisera produce a single precipitin line in immunodiffusion tests and decorate all types of virus particle (Grieco et al., 1992). An antiserum to a recombinant virus movement protein was used for identifying the intracelluar sites of accumulation of this protein by immuno-gold-labelling (Grieco et al., 1999).
Nucleic Acid Hybridization
Random-primed cDNA probes obtained using fractionated nucleic acid extracted from purified virus as template, hybridized with the homologous template but not with RNA preparations from Alfalfa mosaic virus (Grieco et al., 1992). A RNA-2-specific molecular probe successfully detected viral RNA by Northern blot analysis of total nucleic acid extracts from infected N. benthamiana (Grieco et al., 1995).
Relationships
The virus is the type species and the sole member of the genus Oleavirus. This genus is included in the family Bromoviridae because of similarities in genome structure, organization and comparative analysis of structural and nonstructural proteins (Martelli & Grieco, 1997). The virus is not related serologically to any of the members of this family.
Stability in Sap
In sap of G. globosa, the virus lost infectivity after diluting 10-6, heating for 10 min at 60 °C, or storage at 20 °C for more than a week (Savino et al., 1984).
Purification
Harvest N. benthamiana leaves 8-10 days after inoculation and homogenize in 70mM phosphate buffer pH 7 containing 10 mM EDTA and 0.1% 2-mercaptoethanol. Filter the homogenate through cheesecloth, and clarify by adding an equal volume of 1:1 chloroform/butanol. Stir the mixture for 15 min at room temperature and centrifuge at 12,000 g for 10 min. Recover the upper aqueous phase and add PEG mol. wt 6,000 and NaCl to a concentration (w/v) of 10% and 1%, respectively, and allow to stand on ice for 1 h. Collect the precipitate by low speed centrifugation (12,000 g for 10 min) and resuspend overnight in 20 mM phosphate buffer, pH 7. Centrifuge the preparation at 80,000 g for 1h and resuspend the pellet in the same buffer. Centrifuge virus suspension through 10-40% sucrose density gradients in 20 mM phosphate buffer, pH 7 at 24,000 rpm in a Beckman SW27 rotor (Grieco et al., 1992). In sucrose density gradients, virus preparations show a complex sedimentation profile indicating the presence of two major and 3 or 4 minor centrifugal components. Fractions corresponding to the various sedimentation peaks contain particles of different shape and size, but a clear-cut separation of the diverse classes of particles is not achieved. Infectivity is mostly associated with the fast-sedimenting bacilliform components (Grieco et al., 1992). Virus yields are 3-4 mg/100 g of tissue (Savino et al., 1984).
Properties of Particles
Sedimentation coefficient: in the analytical ultracentrifuge, purified virus preparations sediment as two components with a sedimentation coefficient (s20,w) of 77 S and 82 S, respectively, not extrapolated to infinite dilution (Savino et al., 1984).
Buoyant density: virus particles are unstable in CsCl and Cs2SO4, but are stabilized by fixation with 2% formaldehyde. They band as a single component at a density of 1.36 g.cm-3 in CsCl and 1.30 g.cm-3 in Cs2SO4 (Savino et al., 1984; Grieco et al., 1992).
A260/A280: 1.58 (uncorrected for light scattering) (Savino et al., 1984).
Particle Structure
Virus particles are quasi-isometric about 26 nm in diameter, and bacilliform, with four different size classes with lengths of 37, 43 (the most frequent value), 48, and 55 nm, and a diameter of about 18 nm (Fig. 2). Very long particles, up to 85 nm long, are present occasionally. The surface structure of negatively stained particles is poorly resolved (Grieco et al. 1992).
Particle Composition
Nucleic acid: virus particles encapsidate four major and three minor species of linear, positive sense ssRNA, accounting for about 19% of the particle weight. The sizes of the four major RNA species are: RNA-1, 3,126 nt; RNA-2, 2,734 nt; RNA-3, 2,438 nt; RNA-4, 2,078 nt. The three minor RNA species have an apparent size of c. 500, 300 and 200 nt (Grieco et al., 1992, 1995, 1996). The larger RNA species are packaged in the bacilliform particles, whereas the smaller RNA species are contained in the quasi-isometric particles (Grieco et al., 1992).
Protein: the coat protein is a single species with Mr of about 24,000 as estimated by PAGE (Savino et al., 1984; Grieco et al., 1992) or, of 19,841 as deduced from the amino acid sequence of the coat protein cistron (Grieco et al., 1995).
Properties of Infective Nucleic Acid
A mixture of preparations of RNA-1, -2, and -3 obtained by density gradient centrifugation are as infective as unfractionated virus particles. The addition of virus coat protein to RNA preparations does not enhance infectivity (Grieco et al, 1995). In vitro translation of a mixture of the four major RNA species yields three major virus-specific proteins with estimated Mr of 110,000, 90,000, and 35,000. In vitro translation of RNA-4 alone does not yield any product (Grieco et al., 1995).
Genome Properties
The virus has a tripartite genome and all three RNA molecules (RNA-1, RNA-2, and RNA-3) are necessary for infectivity. RNA-1 and RNA-2 are monocistronic, RNA-3 is bicistronic. An additional encapsidated RNA (RNA-4) has no coding capacity (Fig. 3). A fifth RNA is a non-encapsidated subgenomic form of RNA-3. The complete nucleotide sequences of the four major RNA species have been determined, and all have a capped 5' terminus and 3' termini that are not polyadenylated and have a conserved untranslated region of 260-300 nt in size with a predicted similar "stem-loop" secondary structure (Grieco et al., 1996). RNA-1 is 3,128 nt (accession No. X94346, Grieco et al., 1996) and contains a single ORF that extends between nt 104 and 2830 and encodes a polypeptide of 102,689 Da (p1a) with the conserved motifs of helicase and methyltransferase. RNA-2 is 2,735 nt (accession No. X94347, Grieco et al., 1996) and contains a single ORF that extends between nt 114 and 2477 and encodes a polypeptide of 90,631 Da (p2a) with the GDD motif of RNA-dependent RNA-polymerase. RNA-3 is 2,438 nt (accession No. X76993, Grieco et al., 1995) and contains two ORFs. ORF1 (nt 360-1373) encodes a 36,471 Da polypeptide possessing the conserved motifs of the "30K superfamily" of movement proteins (MP), and ORF2 (nt 1445-1993) encodes a 19,841 Da polypeptide identified as the coat protein (CP). RNA-4 (accession No. X77115, Grieco et al., 1995) is a copy of the 3'-most 2,078 nt of RNA-3 with no apparent messenger activity. A non-encapsidated subgenomic RNA, 1,042 nt in size with strong sequence homology with RNA-3 and RNA-4, occurs in RNA extracts from infected plants (Grieco at al., 1995).
Satellite
No accessory RNAs have been found. The three small RNAs of 200 - 500 nt in size are not satellite RNA molecules because their cDNA probes hybridize strongly with sucrose density gradient-fractionated preparations of the larger genomic RNAs (Grieco et al., 1992).
Relations with Cells and Tissues
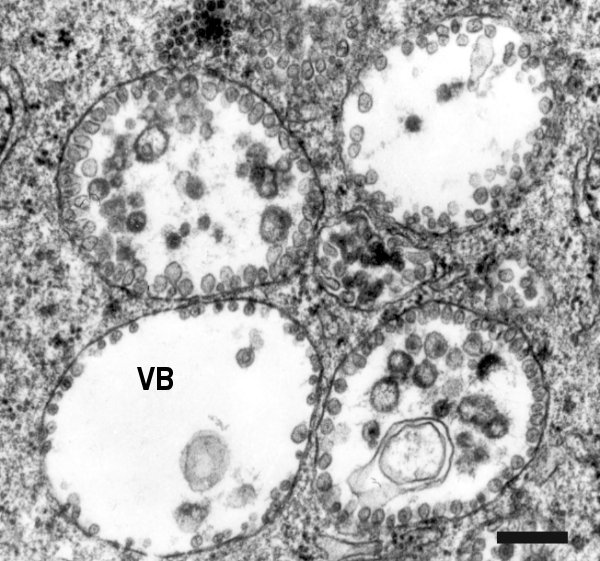
Cells of different plant host species infected locally (G. globosa) or systemically (N. benthamiana and N. megalosiphon) show a much deranged cytology and modifications of major organelles, except for the nuclei. The cytoplasm contains globose aggregates of vesicular elements with fibrillar content, surrounded by a unit membrane (vesiculated bodies), possibly representing sites of virus replication (Fig. 4). Cell walls show localized thickenings, protrusions centred on plasmodesmata, and frequent paramural bodies. Tubular structures with variable length and a diameter of about 40 nm occur in the cytoplasm of many cells, either scattered or connected with plasmodesmata (Fig. 5). Many of these tubules contain rows of electron-dense round to ovoid bodies, interpreted as virus particles (Castellano et al., 1987; Grieco et al., 1999). Virus particles are also present in the cytoplasm in small disorderly aggregates that may gather into larger clumps occupying distinct cytoplasmic areas (Castellano et al., 1987). Immunoblot analysis of infected N. benthamiana showed that the product of the expression of ORF 1 of RNA-3, identified as the putative movement protein (MP), accumulates in the early stages of infection and is associated with a subcelluar fraction enriched for cytoplasmic membranes. Immunogold labeling localized the MP in the plasmodesmata of infected cells and on the virus-containing tubules. When the MP was transiently expressed in transfected tobacco protoplasts it induced the formation of tubular structures protruding from the protoplast surface with which it was associated. (Grieco et al., 1999).
Ecology and Control
No information.
Notes
The virus resembles Alfalfa mosaic virus in particle shape, size of coat protein subunits and number of infective RNA species, but differs from it in, (i) the size and electrophoretic banding pattern of encapsidated RNA species, (ii) serology, (iii) sequence homology, (iv) intracellular behaviour and cytopathology, and (v) natural and experimental host range (Castellano et al.,1987; Grieco et al., 1992). Phylogenetically, the virus is placed in the family Bromoviridae and shows sequence homology of its putative MP with the MP of cucumoviruses and bromoviruses, and of its CP with that of alfamovirus (Grieco et al., 1995). The virus differs from all genera in the family because of the presence of RNA-4, an encapsidated RNA with undetermined significance. This RNA is co-terminal with RNA-3, having virtually the same sequence, but it is not a subgenomic messenger form of RNA-3 for it lacks coding capacity. Unverified hypotheses suggest that RNA-4 may be a degradation, or a premature termination, product of RNA-3, or may represent a defective interfering RNA (Grieco et al., 1995).
Figures
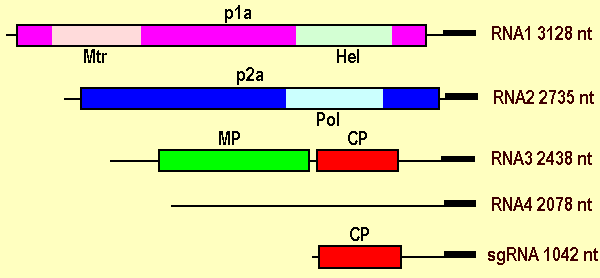
Virus genome organization showing encapsidated (RNA-1, -2, -3, and -4) and a non-encapsidated (sgRNA) RNA species, the relative positions of their ORFs, and their translation products. p1a and p2a, replication-associated proteins; Mtr, methyltransferase; Hel, helicase; MP, movement protein; CP, coat protein.
References list for DPV: Olive latent virus 2 (384)
- Castellano, Di Franco & Martelli, Journal of Submicroscopic Cytology 19: 495, 1987.
- Grieco, Martelli, Savino & Piazzolla, Rivista di Patolologia Vegetale (S.V.) 2: 125, 1992.
- Grieco, Martelli & Savino, Journal of General Virology 76: 929, 1995.
- Grieco, Dell'Orco & Martelli, Journal of General Virology 77: 2637, 1996.
- Grieco, Castellano, Di Sansebastiano, Maggipinto, Neuhaus & Martelli, Journal of General Virology 80: 1103, 1999.
- Martelli & Grieco, Archives of Virology 142: 1933, 1997.
- Savino, Piazzolla, Di Franco & Martelli, Proceedings of the 5th Congress of the Mediterranean Phytopathological Union ,Cairo, 1984: 24, 1984.