Details of DPV and References
DPV NO: 387 December 2001
Family: Unallocated ssRNA+ viruses
Genus: Sobemovirus
Species: Subterranean clover mottle virus | Acronym: SCMoV
This is a revised version of DPV 329
Subterranean clover mottle virus
Roger A. C. Jones Plant Pathology Section, Department of Agriculture, Baron-Hay Court, South Perth, WA 615, Australia.
John Fosu-Nyarko State Agricultural Biotechnology Centre, Murdoch University, Murdoch, WA 615, Australia.
Michael G.K. Jones State Agricultural Biotechnology Centre, Murdoch University, Murdoch, WA 615, Australia.
Geoffrey I. Dwyer State Agricultural Biotechnology Centre, Murdoch University, Murdoch, WA 615, Australia.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
First described by Francki et al. (1983).
SCMoV has isometric particles, about 25 nm in diameter, which contain one genomic ssRNA molecule (4,258 nucleotides). Most naturally occurring isolates also contain either one or both of two ssRNA satellite molecules. The virus is readily mechanically transmissible. It is transmitted naturally from plant to plant by contact, mainly through trampling by stock and on the wheels of vehicles. SCMoV has a narrow host range among dicotyledonous plants. Its known geographical incidence is limited to the southern parts of Australia where infection is widespread in annual legume-based pastures. It causes significant economic losses in such pastures, especially in those dominated by Trifolium subterraneum.
Main Diseases
SCMoV was originally discovered in 1979 in plots of T. subterraneum cv. Dinninup at Karridale in south-west Australia (McLean, 1983; McLean & Price, 1984). It has also been found naturally infecting some other cultivated annual clovers, such as T. vesiculosum, and some wild annual clovers, such as T. glomeratum (Francki et al., 1983, 1988; Ferris and Jones, 1994). Diseased clover plants show obvious symptoms consisting of leaf mottling, leaf distortion, decreased leaf size and plant stunting (Fig. 1, Fig. 2 and Fig. 3) (Wroth & Jones, 1992a). The incidence of infection often reaches high levels in T. subterraneum plants within old annual pastures (Johnstone & McLean, 1987; Wroth & Jones, 1992b; Helms et al., 1993). Infection decreases herbage and seed production of grazed T. subterraneum swards, diminishing feed for stock and the ability of annual pastures to regenerate from seed (Wroth & Jones, 1992b; Ferris & Jones, 1994, 1995; Barbetti & Jones, 1999). Seeds from infected plants tend to be smaller and produce less vigorous seedlings (Fig. 4) (Wroth & Jones, 1992b). The virus is a major contributor to the decline of the T. subterraneum component within mixed species, self-regenerating annual pastures. Infection of pastures with SCMoV is estimated to cause economic losses of A$31 million per year to the Australian dairy industry. The virus also causes substantial annual economic losses to the Australian wool and meat industries (Ferris & Jones, 1994, 1995; Jones, 1996).
There is natural genetic resistance among commercial cultivars of T. subterraneum, some cultivars having partial resistance and others strong resistance (Fig. 5). This resistance is not expressed at the single cell level but is based on restricted cell-to-cell movement of the virus (Wroth & Jones, 1992a; Njeru et al., 1995; Ferris et al., 1996; McKirdy et al., 1998). Similar resistances are found within some other annual Trifolium species that are sown in pastures (Wroth & Jones, 1992a; Fosu-Nyarko et al., 2002).
Geographical Distribution
The known geographical incidence of SCMoV is limited to the southern parts of Australia, including the States of Western Australia, South Australia, Tasmania, Victoria and New South Wales (Wroth & Jones, 1992b; Helms et al., 1993). No surveys for SCMoV have been done in other regions of the world where annual Trifolium spp. are grown or originate (e.g. the Mediterranean basin). However, some germplasm accessions of T. subterraneum from the Mediterranean region (e.g. from Sardinia) are highly resistant to SCMoV suggesting that natural selection for resistance to the virus may have occurred in the centre of origin of this species.
Host Range and Symptomatology
SCMoV has a narrow natural host range limited to certain wild and cultivated species of the genus Trifolium. The virus is readily transmitted to healthy T. subterraneum plants by brushing infected plants against them (Wroth & Jones, 1992a). It is also readily transmitted using mechanical inoculation by grinding infected leaves in 100 mM potassium phosphate buffer (pH 7.4) mixed with a small amount of diatomaceous earth to act as an abrasive before gently rubbing the mixture onto the upper surface of leaves. Grafting infected T. subterraneum scions onto healthy T. subterraneum rootstocks transmits the virus easily (Wroth & Jones, 1992a; Njeru et al., 1995).
By mechanical inoculation, SCMoV successfully infected 45 species from 10 genera in the Leguminosae, 33 systemically, and 3 species from one genus in the Chenopodiaceae, 1 systemically (Francki et al., 1983, Wroth & Jones, 1992a, Fosu-Nyarko et al., 2002). Systemic infection typically results in leaf mottling, which is often associated with decrease in leaf size, leaf deformation and different degrees of plant dwarfing. Transient vein clearing of young leaves and leaf pallor are other common symptom types. Within some genotypes of clover host species, infection remains restricted to the inoculated leaves of all or a proportion of mechanically inoculated plants (Wroth & Jones, 1992a; Njeru et al., 1995; Fosu-Nyarko et al., 2002). Occasionally, new shoots produced by systemically infected plants of T. subterraneum are healthy and these often rapidly outgrow the infected ones (Wroth & Jones, 1992b).
Diagnostic species
Trifolium clypeatum. Persistent severe vein clearing, decreased leaf size, chlorosis, mottle, necrotic streaking alongside veins and severe plant stunting. Systemic symptoms appear 10-14 days after inoculation. (Fig. 6 and Fig. 7).
Trigonella balansae (SA5054). Obvious necrotic spot lesions in inoculated leaves after 6 days followed by initial vein clearing in young leaves, decreased leaf size, severe mottle, leaf deformation and stunting. Systemic symptoms appear 10-14 days after inoculation.
Chenopodium quinoa. Chlorotic and necrotic spot lesions in inoculated leaves after 7-14 days (Fig. 8). Systemic mottling develops in young leaves 3 weeks after inoculation.
Pisum sativum cv. Greenfeast. Necrotic spot lesions appear in inoculated leaves after 7-14 days (Fig. 9).
Additional hosts that develop distinct systemic symptoms of diagnostic value include Cicer arietinum, Lathyrus ochrus, Lens culinaris, T. purpureum, T. incarnatum and Vicia narbonensis.
Useful non-hosts include Cucumis sativus, Gomphrena globosa, Nicotiana benthamiana and N. glutinosa.
Propagation species
Assay species
C. quinoa and P. sativum cv. Greenfeast can also be used for local lesion assays.
Strains
SCMoV isolates differ markedly in symptom severity when infecting T. subterraneum plants (Wroth & Jones, 1992b) but no serologically distinct strains are reported.
Transmission by Vectors
Readily transmitted by trampling of stock and on the wheels of vehicles. Transmission by grazing and, in particular, mowing is less efficient (Wroth & Jones 1992a; McKirdy et al., 1998). Within pastures, high incidences of infection are frequent in heavily trampled areas, e.g. around gates, and in animal (Fig. 3) and wheel tracks, reflecting the method of spread. No specialised vectors are known.
Transmission through Seed
Seed transmission has been detected so far in T. subterraneum and three wild annual clover species (T. campestre, T. cernuum, and T. tomentosum) that occur within pastures. It probably also occurs in other Trifolium spp. SCMoV is only seed-borne at low levels with rates of transmission to seedlings in the range 0.01 to 0.5% regardless of clover species, cultivar or virus isolate tested (Wroth & Jones, 1992a, 1992b; Njeru et al., 1997). Testing of ungerminated seeds revealed greater SCMoV infection because of seed coat contamination without embryo infection (Francki et al., 1988; Njeru et al., 1997). Pre-treatment of seeds with trisodium phosphate helped eliminate seed coat contamination with SCMoV (Njeru et al., 1997). There is no information on pollen transmission.
Serology
SCMoV particles are strongly immunogenic. Polyclonal antibodies are readily produced in rabbits using purified particles as the immunogen. These antibodies are useful in ELISA for detecting SCMoV in different plant species, and in other serological assays (e.g. gel diffusion tests, immunofluorescence assays, dot blot immunoassays) (Francki et al., 1983; Graddon & Randles, 1986; Wroth & Jones, 1992b; Njeru et al., 1995). No monoclonal antibodies have been prepared.
Relationships
Particle morphology, physical and biochemical properties all place SCMoV in the genus Sobemovirus. Its particles have a remote antigenic relationship with those of lucerne transient streak virus but not with those of cocksfoot mottle, solanum nodiflorum mottle, southern bean mosaic, sowbane mosaic or velvet tobacco mottle viruses (Francki et al., 1983, 1985, 1988; Campbell & Guy, 2001). SCMoV resembles velvet tobacco mottle, solanum nodiflorum mottle, lucerne transient streak and rice yellow mottle viruses in having associated satellite RNA molecules (Francki et al., 1983, 1988; Dall et al., 1990; Davies et al., 1990; Collins et al., 1998). SCMoV satellite RNA was capable of replicating in the presence of lucerne transient streak virus acting as a helper virus under glasshouse conditions in C. quinoa (Dall et al., 1990). Comparisons of the complete nucleotide sequences of SCMoV and 7 other sobemoviruses showed that SCMoV shares most identity with lucerne transient streak virus (Dwyer et al., 2002). Also, although its genome organisation differs from those of most other sobemoviruses, it is similar to that of cocksfoot mottle virus (Makenin et al., 1995; Ryabov et al., 1996) (Fig. 10).
Stability in Sap
In infective sap of T. subterraneum cv. Woogenellup, infectivity was detected after one but not two months when stored at 25 °C, after 10 minutes at 70 °C but not at 75 °C, and at dilutions of 10-7 but not 10-8 (Wroth & Jones, 1992a). Mixtures of infective sap of T. subterraneum with bovine saliva or water both remained infective after storage at 20 °C for 4 weeks. Resuspended sap that had been air dried on a metal surface at 20 °C was still infective after four days of drying but not after 14 days (McKirdy et al., 1998). Readily preserved by freeze-drying.
Purification
SCMoV particles are readily purified from leaves of infected T. subterraneum plants using the methods of Gould et al. (1981) or Njeru (1996). Extract particles from infected leaves in 0.1 M potassium phosphate buffer, pH 7.4 containing 0.1% thioglycollic acid (2 ml/g leaf material). Strain the extract through cheesecloth and clarify the filtrate by centrifugation at 10,000 g for 10 min. Emulsify supernatant with an equal volume of a 1:1 mixture of chloroform and butanol, and centrifuge at 10,000 g for 10 min. Pellet the virus by centrifugation at 78,000 g for 90 min and resuspend in a minimal volume of 20 mM phosphate buffer, pH 7.4. Repeat this step twice more. When highly purified virus is required, centrifuge the preparation in a 5-25% sucrose gradient at 26,000 rev/min for 2 h in a Beckman SW 27 rotor. Recover the single virus band, dilute with 10 ml of 20 mM potassium phosphate buffer and concentrate by centrifugation at 230,000 g for 2 h. Resuspend the pellet in 10 ml of 20 mM potassium phosphate buffer pH 7.4 and store at 4 °C.
Properties of Particles
In sucrose density gradients, SCMoV particles form a single band that co-sediments with particles of southern bean mosaic virus, which have a sedimentation coefficient (s20,w) of 115S. Purified preparations have an ultraviolet absorption spectrum characteristic of nucleoprotein with an absorption coefficient (A260(0.1%,1cm)) of about 5 and an A260/A280 ratio of about 1.5 (Francki et al., 1983, 1988; Tremaine & Hamilton, 1983; Njeru, 1996).
Particle Structure
SCMoV virions, negatively stained in phosphotungstate, appear isometric, about 25 nm in diameter, with icosahedral symmetry (Fig. 11). They are indistinguishable from virions of southern bean mosaic and velvet tobacco mottle viruses (Francki et al., 1983, 1988; Njeru, 1996).
Particle Composition
Nucleic acid. The particles contain single-stranded linear RNA (RNA1) of M.Wt. about 1.5 x 106, which comprises the positive-sense genome of 4,258 nucleotides (Dwyer et al., 2002). Particles of most but not all field isolates also contain either one or two satellite RNA species of 332 (accession no. M33000) or 388 (accession no. M33001) nucleotides respectively, each of which exists as both circular (RNA2) and linear (RNA3) forms (Francki et al., 1988; Davies et al., 1990). The two satellite RNAs have about 95% nucleotide sequence identity and are capable of extensive base pairing (Davies et al., 1990). The satellites replicate by a rolling circle method and possess a hammerhead type ribozyme activity (Steinecke et al., 1994). An uncharacterised RNA molecule of about 700 nucleotides has also been reported (Njeru, 1996). Virus particle preparations with A260/A280 ratios up to 1.67 are found indicating that particles have a nucleic acid content of about 20% (Francki et al., 1983).
Protein: Dissociated virus particles yield polypeptides of 29 kDa (major band) and 26 kDa (minor band) in polyacrylamide gels (Francki et al., 1983). The significance of the minor band is unknown but it may be a degradation product of the major polypeptide similar to that found with viruses such as velvet tobacco mottle and solanum nodiflorum mottle (Chu & Francki, 1983).
Genome Properties
SCMoV has a small monopartite genome that is comprised of a ssRNA species of 4,258 nucleotides. The complete nucleotide sequence of a single isolate has been determined (accession no. AF208001) and sequence analysis has permitted the identification of open reading frames (ORF) and predictions for function of some of the encoded proteins (Dwyer et al., 2002). The RNA has a base composition of adenine 27.85%, guanine 25.46%, uracil 24.05% and cytosine 22.64%, with an overall G+C content of 48.1%. The 5' untranslatable region is 68 nucleotides in length and the 3' untranslatable region is 177 nucleotides. The 5' terminus has a VPg attached. The 3' terminus is not polyadenylated and does not possess any obvious structural features.
The genome encodes four overlapping ORFs (Fig. 10). ORF1 and ORF3 are predicted to encode single proteins while ORF2 encodes two proteins linked by a -1 ribosomal frameshift (ORF2a and ORF2b). ORF1 spans nucleotides 69-605. It encodes a polypeptide of 179 amino acids with a calculated Mr of 20,320, whose function is unknown. ORF2a spans nucleotides 605-2344 and encodes a polypeptide of 580 amino acids with a calculated Mr of 63,760. The polypeptide has a motif characteristic of chymotrypsin-like serine proteases and is likely to include the VPg near its C-terminal end. ORF2b spans nucleotides 1849-3501. The encoded polypeptide comprises 551 amino acids, and contains motifs typical of positive-stranded RNA-dependent RNA polymerases. This ORF is translated via a -1 ribosomal frameshift, which is predicted to give rise to a large polyprotein (966 amino acids) that consists of the ORF2a gene product, as far as the -1 frameshifting site, and the entire ORF2b polypeptide, with a calculated Mr of 107,460. ORF3 spans nucleotides 3323-4081 and encodes a protein of 253 amino acids with a calculated Mr of 27,300, which is the viral coat protein (Dwyer et al., 2002).
Published gene expression data for other sobemoviruses provide useful information on the strategies likely to be used by SCMoV to express its genes (Fig. 12). Translation initiation of ORF1 occurs via a 5' end-dependent ribosomal scanning mechanism (Hacker & Sivakumaran, 1997). The ORF1 initiation codon has suboptimal context and, when it is bypassed by the ribosomal scanning mechanism, translation of ORF2 is initiated (Sivakumaran & Hacker, 1998). ORF2b is expressed via a -1 ribosomal frameshifting mechanism (Makenin et al., 1995), and the individual mature ORF2a and ORF2b proteins are released through proteolytic digestion by the protease encoded by ORF2a (Tamm et al., 1999). ORF 3 is expressed from a subgenomic RNA molecule via a 5' end-dependent ribosomal scanning mechanism (Hacker and Sivakumaran, 1997).
Relations with Cells and Tissues
Numerous electron-dense particles about 22 nm in diameter are observed in cells of infected T. subterraneum tissues that have been treated with ribonuclease to remove densely staining ribosomes (Hatta & Francki, 1981). They are distributed in nuclei, cytoplasm and cytoplasmic vacuoles of all types of cells. In the cytoplasm and nucleus they are always seen aggregated in distinct groups (Francki et al., 1983, 1985).
Ecology and Control
SCMoV is a very stable virus that reaches high concentrations within infected plants. It is therefore readily contact-transmissible from infected to healthy clover plants within pastures. This transmission occurs by trampling of stock and on wheels of vehicles, and, to a lesser extent, by grazing and by mowing of pastures for hay production or to prevent pasture growth becoming rank. The virus is seed-borne in annual clover species and the pastures involved are annually self-regenerating. The seeds germinate each year in autumn following opening rains, grow over winter and produce seeds before dying from lack of moisture in spring. The virus survives the dry summer period within dormant clover seeds. The primary infection sources in autumn are plants infected through seed. SCMoV spread is polycyclic and occurs from these primary infection sources to neighbouring plants, which in turn create a larger secondary infection source. If spread starts early and the stocking rate is high, further cycles of virus spread can accelerate the rate of increase in virus incidence so much that virtually all susceptible plants are infected by the end of the annual growing period. Other factors favouring increased virus spread and high final incidences include warmer than normal temperatures in winter and growing seasons extended by late rains. High final incidences result in heavy losses in clover herbage and seed production, and preponderance of pasture grasses and weeds over clover (Wroth & Jones, 1992b; Ferris & Jones, 1994, 1995; Jones, 1996; McKirdy et al., 1998).
An integrated disease management approach is appropriate to control spread of SCMoV in infected pastures. A key control measure is sowing SCMoV-resistant clover cultivars in pastures. Clover cultivars are available that are partially or highly resistant. The latter are more effective as with the former resistance breaks down under heavy stocking pressure (Wroth & Jones, 1992a; Njeru et al., 1995; Ferris et al., 1996). Other control measures include: avoiding new virus introductions by sowing new pastures with tested clover seed stocks that have no contamination with SCMoV; avoiding introduction of SCMoV to healthy pastures on farm machinery or stock moving from older infected pastures; sowing pastures with admixtures of susceptible clover cultivars with resistant ones or with alternative pasture species that are non-hosts (e.g. grasses); relaxing grazing pressure to (i) minimise contact spread by stock and (ii) allow swards to grow up in spring so that healthy plants shade out the stunted SCMoV-infected source plants; avoiding driving vehicles over pastures to prevent SCMoV spread on wheels; decreasing the seed bank of SCMoV-infected clover seeds in the soil by growing crops instead of pastures in affected fields for several years in extended (phased) rotations; and avoiding sowing new pastures in fields next to old infected ones (Ferris & Jones, 1994, 1995; Jones, 1996; McKirdy et al., 1998; Barbetti and Jones, 1999).
Notes
In the field, T. subterraneum plants infected with the virus exhibit symptoms similar to those induced by bean yellow mosaic and subterranean clover stunt viruses (Francki et al., 1985, 1988; Ferris & Jones, 1994). However, when SCMoV is present, inspection of tracks made by stock or vehicle wheels and of heavily trampled areas normally reveals a concentration of infected plants in such areas. Also, SCMoV is readily distinguished in leaf samples by its particle morphology, serological specificity (e.g. in ELISA) and nucleic acid-based methods (e.g. RT-PCR) (Ferris & Jones, 1994, 1995; Jones, 1996; Njeru et al., 1997; McKirdy et al., 1998).
Acknowledgements
The authors are participants in the Centre for Legumes in Mediterranean Agriculture, University of Western Australia, Crawley, WA 6009, Australia. The support provided by CLIMA for research on SCMoV is gratefully acknowledged.
Figures

Leaflets of Trifolium subterraneum cv. Woogenellup infected systemically with SCMoV showing symptoms of vein clearing, mottle, leaf deformation and decreased leaf size. Uninfected leaf on right.

SCMoV-infected patch (centre) with unaffected plants on either side in a sward of Trifolium subterraneum cv. Woogenellup. Plants within infected area show leaf mottle and pallor, leaf deformation, decreased leaf size, and dwarfing.

Close up of sheep track through a sward of Trifolium subterraneum. Note severely dwarfed plants infected with SCMoV in base of track with unaffected plants outside it.

Five week old seedlings of Trifolium subterraneum cv. Woogenellup: healthy (left) and infected with SCMoV via seed (right). Infected seedling shows leaf deformation, decreased leaf size and severe stunting.

Heavily grazed rows of Trifolium subterraneum cultivars Denmark (left) and Woogenellup (right). Note vigorous healthy growth of partially SCMoV resistant cv. Denmark and SCMoV symptoms of chlorosis and stunting in susceptible cv. Woogenellup.

Obvious mottle and necrotic streaking in leaf of Trifolium clypeatum infected systemically with SCMoV.

Local chlorotic and necrotic spots caused by infection with SCMoV in inoculated leaf of Chenopodium quinoa.

Spreading local necrotic spots caused by infection with SCMoV in inoculated leaf of Pisum sativum cv. Greenfeast.

Genome organisation of SCMoV. The ORFs are labelled and shown as dark boxes. VPg attached to the 5' terminus is shown as a filled circle. The position and length of 5' & 3' UTRs are indicated. Location of the chymotrypsin-like serine protease domain, RNA-dependent RNA polymerase domain (RdRp) and coat protein are indicated.
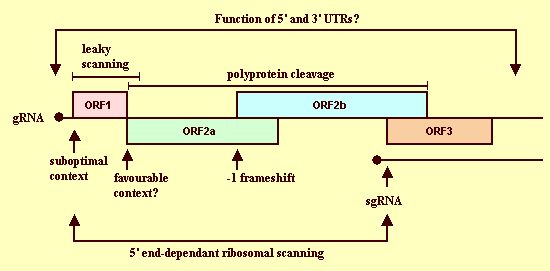
Proposed gene expression strategy of SCMoV, based on data for other sobemoviruses. Translation initiation from genomic (gRNA) and subgenomic RNA (sgRNA) occurs via a 5' end-dependent ribosomal scanning mechanism. The ORF1 initiation codon has suboptimal context and translation of ORF2 occurs via a leaky ribosomal scanning mechanism. ORF2b is expressed via a -1 ribosomal frameshifting mechanism and the individual mature proteins are released through proteolytic digestion by the protease encoded by ORF2a. ORF 3 is expressed from a subgenomic RNA molecule.
References list for DPV: Subterranean clover mottle virus (387)
- Barbetti & Jones, in Proceedings of the Fourth International Herbage Seed Conference - Herbage seed as a key factor for improving production and environmental quality, p. 69. Perugia, Italy: University of Perugia Publishers, 1999.
- Campbell & Guy, Australasian Plant Pathology 30: 217, 2001.
- Chu & Francki, Virology 129: 350, 1983.
- Collins, Gellatly, Sehgal & Abouhaidar, Virology 241: 269, 1998.
- Dall, Graddon, Randles & Francki, Journal of General Virology 71: 1873, 1990.
- Davies, Haseloff & Symons, Virology 177: 216, 1990.
- Dwyer, Njeru, Williamson, Fosu-Nyarko, Hopkins, Jones, Waterhouse & Jones, Archives of Virology (submitted).
- Ferris & Jones, Western Australian Journal of Agriculture, Fourth Series 35: 40, 1994.
- Ferris & Jones, Australian Journal of Agricultural Research 46: 755, 1995.
- Ferris, Jones & Wroth, Annals of Applied Biology 128: 303, 1996.
- Fosu-Nyarko, Jones, Smith, Jones & Dwyer, Australasian Plant Pathology (submitted).
- Francki, Randles, Hatta, Davies & Chu, Plant Pathology 32: 47, 1983.
- Francki, Randles, Chu, Rohozinski & Hatta, in Subviral Pathogens of Plants and Animals: Viroids and Prions, p. 265, ed. K. Maramorosch & J.J. McKelvey, New York: Academic Press, 1985.
- Francki, Randles & Graddon, CMI/AAB Descriptions of Plant Viruses 329: 1988.
- Gould, Francki, Hatta & Hollings, Virology 108: 499, 1981.
- Graddon & Randles, Journal of Virological Methods 13: 63, 1986.
- Hacker & Sivakumaran, Virology 234: 317, 1997.
- Hatta & Francki, Journal of Ultrastructural Research 74: 1, 1981.
- Helms, Muller & Waterhouse, Australian Journal of Agricultural Research 44: 1837, 1993.
- Johnstone & McLean, Annals of Applied Biology 110: 421, 1987.
- Jones, in Pasture and Forage Crop Pathology, p. 303, eds S. Chackraborty, K. Leath, R. Skipp, G. Pederson, R. Bray, G. Latch & F. Nutter, Madison, Wisconsin: American Society of Agronomy, 1996.
- Makenin, Tamm, Naess, Truve, Puurand, Munthe & Saarma, Journal of General Virology 76: 2817, 1995.
- McKirdy, Jones & Sivasithamparam, Annals of Applied Biology 132: 91, 1998.
- McLean, Western Australian Journal of Agriculture, Fourth Series 24: 12, 1983.
- McLean & Price, Western Australian Department of Agriculture Technical Bulletin No. 68, p. 22, 1984.
- Njeru, PhD Thesis, Murdoch University, Perth, Western Australia, 1996.
- Njeru, Jones, Sivasithamparam & Jones, Australian Journal of Agricultural Research 46: 633, 1995.
- Njeru, Ferris, Jones & Jones, Australian Journal of Agricultural Research 48: 343, 1997.
- Ryabov, Krutov, Novikov, Zheleznikova, Morozov & Zavriev, Molecular Plant Pathology 86: 391, 1996.
- Sivakumaran & Hacker, Virology 246: 34, 1998.
- Steinecke, Steger & Schreier, Gene 149: 47, 1994.
- Tamm, Makenin & Truve, Archives of Virology 144: 1557, 1999.
- Tremaine & Hamilton, CMI/AAB Descriptions of Plant Viruses 274: 1983.
- Wroth & Jones, Annals of Applied Biology 121: 329, 1992a.
- Wroth & Jones, Australian Journal of Agricultural Research 43: 1597, 1992b.

