Details of DPV and References
DPV NO: 394 February 2003
Family: Secoviridae
Genus: Sequivirus
Species: Parsnip yellow fleck virus | Acronym: PYFV
This is a revised version of DPV 129
Parsnip yellow fleck virus
A. F. Murant Scottish Crop Research Institute, Invergowrie, Dundee DD2 5DA, Scotland, UK
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Murant & Goold (1968) and Hemida & Murant (1989a).
- Synonyms
- Celery yellow net virus (Hollings, 1964) is probably synonymous with the parsnip serotype of PYFV.
A virus with isometric particles c. 31 nm in diameter forming two centrifugal components: 'empty' protein shells, and 'full' nucleoprotein particles. The virus possesses a single genomic ssRNA molecule of 9871 nt and three coat proteins of Mr (x10-3) 30-31, 26 and 22.5-24. The RNA encodes a large polyprotein from which the mature proteins are probably cleaved by protease action. Two major serotypes are known, with slightly different host ranges; each serotype infects systemically several species of umbelliferous plant, and a few species in other families. Transmission by the aphid Cavariella aegopodii occurs in a semi-persistent manner and is dependent on a helper virus, Anthriscus yellows virus (genus Waikavirus, family Sequiviridae). The virus is also transmitted by Cavariella pastinacae and, inefficiently, by C. theobaldi. Found in western Europe.
Main Diseases
The parsnip serotype causes vein-yellowing, yellow flecks and yellow/green mosaic symptoms in parsnip (Pastinaca sativa) (Fig. 1; Fig. 2) (Murant & Goold, 1968), and 'yellow net', followed by yellow spots and leaf distortion in celery (Apium dulce) (Hollings, 1964; Singh & Frost, 1980). It also occurs in the wild plant Heracleum sphondylium (hogweed).
The anthriscus serotype occurs naturally in the wild plant Anthriscus sylvestris (cow parsley). It causes necrosis, severe stunting and 'viral dieback' in carrot (Daucus carota) plants infected when young (Fig. 3), and mottle, yellow flecks and mosaic in plants infected later, with the roots often becoming distorted at the crown (`cigar-shaped') and exhibiting internal necrosis (Fig. 4) (Murant & Spence, 2002). In the Netherlands, the anthriscus serotype was found to be responsible for early season dieback of carrot seed crops, and also caused dieback symptoms in chervil (Anthriscus cerefolium), coriander (Coriandrum sativum) and dill (Anethum graveolens) (Van Dijk & Bos, 1985).
Geographical Distribution
Reported from the UK (Murant & Goold, 1968), Germany (Wolf & Schmelzer, 1972) and the Netherlands (Van Dijk & Bos, 1985; 1989).
Host Range and Symptomatology
PYFV is easily transmitted by manual inoculation. Isolates of both serotypes infect many species of Umbelliferae, and several species in other families, especially in the Chenopodiaceae and Solanaceae (Murant & Goold, 1968; Van Dijk & Bos, 1985; Hemida & Murant, 1989b). However, isolates of the anthriscus serotype, unlike those of the parsnip serotype, do not infect celery or parsnip, and rarely or never give systemic infection in common solanaceous test plants, notably Datura stramonium, Nicotiana clevelandii, N. glutinosa, N. rustica, N. tabacum, Petunia hybrida and Physalis floridana. An exception to this generalization is Nicotiana benthamiana, which is an important systemic host of isolates of both serotypes.
- Diagnostic hosts
In hosts that they have in common, isolates of both serotypes give very similar symptoms. The reactions of the most important experimental hosts in glasshouse conditions are as follows (symptoms are for both serotypes unless otherwise stated):
Anthriscus cerefolium (chervil) and Coriandrum sativum (coriander). Local necrotic spots. Within about 4 days of inoculation, young systemically infected leaves blacken and shrivel, at first distally. The necrosis spreads along the petiole into the crown and the plant dies, usually within 2 weeks of inoculation (Fig. 5).
Chenopodium amaranticolor. Minute necrotic local lesions; not systemic (Fig. 6).
C. quinoa. Necrotic local lesions 2-3 mm in diameter in 4 days (Fig. 7), sometimes followed by systemic necrotic flecks and distortion.
Nicotiana benthamiana. Plants inoculated in winter show necrotic local lesions up to 6 mm in diameter, and the inoculated leaf usually shrivels and dies within 5 days. Plants inoculated in summer develop symptomless local infection. In all seasons, the plants quickly develop a characteristic systemic epinasty (Fig. 8), with irregular chlorotic areas and crinkling of the leaves. The axillary shoots become necrotic and the plant dies after about 10 days.
Nicotiana clevelandii. Isolates of the parsnip serotype induce local chlorotic or necrotic spots or rings followed by systemic veinal necrosis (Fig. 9); plants later partially recover and show a light and dark green mottle. Plants inoculated with isolates of the anthriscus serotype may develop a symptomless or mild infection in inoculated leaves but rarely become infected systemically.
Spinacia oleracea (spinach). Local chlorotic spots, followed by systemic bright yellow flecks, yellow/green mottle, or yellowing of the whole leaf (Fig. 10).
- Propagation species
Spinacia oleracea is the best source of virus for purification. This species and (for isolates of the parsnip serotype only) Nicotiana clevelandii are useful for maintaining isolates.
- Assay species
Chenopodium quinoa is a good local lesion host. Anthriscus cerefolium is a good test plant for use in aphid-transmission experiments.
Strains
All isolates studied so far belong to one or other of two serotypes (Murant & Goold, 1968; Hemida & Murant, 1989b): (i) the parsnip serotype (type isolate P-121), which occurs in parsnip, celery and Heracleum sphondylium, and (ii) the anthriscus serotype (type isolate A-421), which occurs in carrot and Anthriscus sylvestris. Minor differences in immunological and host reactions were observed between isolates within each serotype (Hemida & Murant, 1989b).
Transmission by Vectors
PYFV is transmitted by the aphids Cavariella aegopodii and C. pastinacae, but not by Myzus persicae (Murant & Goold, 1968). Bem & Murant (1979) found that C. theobaldi did not transmit, but Van Dijk & Bos (1989) reported that this species is an inefficient vector. However, aphid-transmission depends on the presence of a helper virus. The helper virus found in Anthriscus sylvestris is called Anthriscus yellows virus (AYV) (Murant & Goold, 1968) and, experimentally, this virus can assist the transmission by C. aegopodii of PYFV isolates of both serotypes (Elnagar & Murant, 1976a). Because PYFV is transmissible by aphids from Heracleum sphondylium (Bem & Murant, 1979), a helper virus probably exists in this species but it has been little studied and has not been characterised. AYV, which resembles PYFV in particle properties but has distinct biological properties (Hemida et al., 1989), is classified in the genus Waikavirus, family Sequiviridae. Carrot red leaf virus, which assists transmission of Carrot mottle virus by C. aegopodii, does not act as a helper for PYFV (Murant & Goold, 1971; Elnagar & Murant, 1976b).
AYV, unlike PYFV, is confined to the phloem tissue of infected plants and is not transmissible by inoculation of sap; aphid-transmissibility of a PYFV/AYV culture is therefore lost after a manual transmission. Aphids are also unable to transmit PYFV from plant species, such as parsnip or carrot, that are immune to AYV (Murant & Goold, 1968). However, experimentally, aphids that have already acquired AYV from another plant can acquire PYFV from plants infected with PYFV alone (Elnagar & Murant, 1976a), or by feeding through membranes on plant extracts or purified preparations containing PYFV alone (Elnagar & Murant, 1976b). The aphids can then transmit both viruses to susceptible test plants, such as chervil or coriander. Aphids that feed first on a source of PYFV, and then on a source of AYV, transmit only AYV.
Both AYV and PYFV are transmitted by C. aegopodii in the semi-persistent manner (Elnagar & Murant, 1976a). C. aegopodii can acquire both dependent and helper viruses after minimum acquisition feeding times of 10-15 min and can inoculate in 2 min, with no latent period. However, aphids already carrying AYV can acquire PYFV in 2 min. Transmission efficiency increases with increasing acquisition and inoculation feeding times up to 24 h. Starving the aphids for 3 h before acquisition of either virus, or both viruses together, does not improve the efficiency of transmission. Infective adult aphids retain the ability to inoculate both viruses for up to 4 days (usually only 1 day), but infective larvae lose ability to inoculate the viruses on moulting (Murant & Goold, 1968; Elnagar & Murant, 1976a).
Virus-like particles 20-28 nm in diameter were found by electron microscopy in the pharynx of aphids (C. aegopodii) that were carrying AYV or the AYV/PYFV complex but not in aphids that were not carrying the viruses (Murant et al., 1976). The particles were thought to be those of AYV but the possibility could not be ruled out that some of the particles in aphids carrying the virus complex were those of PYFV.
Transmission through Seed
PYFV is probably not seed-transmitted.
Serology
PYFV is moderately to highly immunogenic. Antisera to isolate A-421 (anthriscus serotype) have immunodiffusion titres of 1/1024 to 1/4096, whereas those to isolate P-121 (parsnip serotype) usually have titres of about 1/256 (Murant & Goold, 1968). The concentration of antigen in sap of glasshouse-grown plants is often too low to give reactions in immunodiffusion tests, and such tests are best done with sap from spinach grown in the conditions mentioned below (see Purification), or with purified preparations. Isolates of both serotypes are readily detected by ELISA.
Relationships
In immunodiffusion tests with eight isolates of PYFV, Hemida & Murant (1989b) distinguished two major serotypes, one (here called the parsnip serotype) containing isolates from parsnip, celery and Heracleum sphondylium, and the other (here called the anthriscus serotype) containing isolates from carrot and Anthriscus sylvestris. When placed in wells adjacent to each other in immunodiffusion tests, isolates within each serotype formed no spurs or only weak spurs, whereas isolates from different serotypes gave very strong spurs.
Stability in Sap
In Spinacia oleracea sap, the virus lost infectivity after 10 min at 57.5-65 °C, storage at room temperature for 4-7 days, or dilution to 10-3-10-4 (Murant & Goold, 1968). Dilution end-points in sap may be only 10-1 in glasshouse-grown plants in summer. However, they can be up to 10-4-10-5 in sap from spinach plants kept in growth chambers under the conditions specified below (see Purification).
Purification
Yields of virus are very dependent on the environmental conditions in which the source plants are kept. The best virus yields are obtained by placing the plants after inoculation in growth chambers at 15 °C, with light at 10000 lux for 8 h per day, and harvesting whole systemically infected plants 19 days after inoculation (Abu Salih, 1968). Extract the tissue in 60 mM phosphate buffer, pH 7.0, containing 10 mM EDTA and 0.1% thioglycollic acid, then clarify by either (i) adding an equal volume of diethyl ether, shaking the mixture well, and after 30 min at room temperature collecting the virus-containing aqueous layer, or (ii) adding butan-1-ol to give a concentration of 8% (v/v) (Hemida & Murant, 1989a). Following either treatment, clarify the resulting extract by low-speed centrifugation, then precipitate the virus by adding polyethylene glycol, Mr 6000, to 10% (w/v) and NaCl to 1% (w/v), stirring for 60 min at 4 °C, then centrifuging at low speed. Resuspend the pellet in 60 mM phosphate buffer, pH 7.0, and concentrate the virus further by differential centrifugation. The ether method is the more reliable with isolates of the anthriscus serotype, or with either serotype if the maximum amount of top component is required. Yields of bottom component particles are up to 1 mg per 100 g leaf tissue; yields of top component may be up to 0.6 mg (isolate A-421) or 5.5 mg (isolate P-121) per 100 g leaf tissue.
Properties of Particles
The particles sediment as two components (Hemida & Murant, 1989a). Top component particles (T) have sedimentation coefficients (s°20) of about 56 S (isolate A-421) or 60 S (isolate P-121). Bottom component particles (B) have sedimentation coefficients (s°20,w) of about 148 S (isolate A-421) or 152 S (isolate P-121).
A260/A280: 0.8-0.9 (T); 1.9 (B) (Hemida & Murant, 1989a).
Buoyant density in CsCl (g/cm3): T: 1.297; B: 1.490 (isolate P-121) or 1.520 (isolate A-421) (Hemida & Murant, 1989a).
In immunoelectrophoresis, using 0.025 ionic strength barbitone acetate buffer, pH 8.6, the particles migrate heterogeneously towards the anode, producing a single precipitin line, without spurs, trailing to the origin.
Particle Structure
Particles are isometric, c. 31 nm in diameter (Fig. 11). T particles are stain-penetrable protein shells (Fig. 11b), which are very fragile. They are better preserved by purification with diethyl ether than with butan-1-ol, and do not survive the butanol/chloroform purification procedure. B particles are not penetrable by negative stain (Fig. 11a).
Particle Composition
Nucleic acid: B particles contain one ssRNA molecule, of Mr about 3.4 × 106, constituting 40% of the particle weight (Hemida & Murant, 1989a).
Protein: The protein shell consists of three polypeptides, of Mr (× 10-3) about 31, 26 and 23 (isolate P-121) or 30, 26 and 24 (isolate A-421) (Hemida & Murant, 1989a).
Genome Properties
The nucleotide sequence and genome structure of isolate P-121 (Fig. 12) was determined by Turnbull-Ross et al. (1992, 1993) (Accession No. D14066). The genome is a positive-sense ssRNA of 9871 nt. The 3' end is not polyadenylated but terminates in COH and may form a base-paired structure with some resemblance to that deduced for the genomic RNA of flaviviruses (Turnbull-Ross et al., 1992). Loss of infectivity after treatment with pronase or proteinase K suggests that PYFV RNA may possess a genome-linked protein (Murant et al., 1987).
The genomic RNA contains a large ORF between nts 279 and 9362, which encodes a polyprotein of 3027 amino acids, with a calculated Mr of 336K (Turnbull-Ross et al., 1992). The mature viral proteins, including the three coat proteins, are presumably cleaved from the polyprotein by protease action. The 22.5K, 26K and 31K coat proteins are encoded in that order in the 5' half of the genomic RNA (Turnbull-Ross et al., 1992), and are found in the N-terminal half of the polyprotein, beginning at amino acid positions 395, 589 and 811, respectively (Turnbull-Ross et al., 1993). There are polypeptide(s) of up to 43K in total on the N-terminal side of the particle protein sequences. A region within the 26K particle protein has sequence similarity to the VP3 particle protein of picornaviruses. Three other regions in the PYFV polyprotein have sequence similarity to regions thought to have RNA polymerase, nucleoside triphosphate (NTP)-binding and protease functions in the polyproteins of picornaviruses, comoviruses and nepoviruses.
Relations with Cells and Tissues
In spinach, Nicotiana clevelandii and Anthriscus cerefolium, the virus induces the formation, adjacent to the nucleus, of inclusion bodies composed of vesicles, straight tubules about 30 nm in diameter, and 'matrix material' (Fig. 13; Fig. 14) (Murant et al., 1975; Fasseas et al., 1989). Infected cells also possess plasmodesmatal and cytoplasmic tubules about 45 nm in diameter containing virus particles. The plasmodesmatal tubules are sheathed by cell wall outgrowths. Tubules containing virus particles also occur in the lumen of sieve tubes.
Immunogold labelling studies (Fasseas et al., 1989) showed the presence of PYFV particle antigen in the 'matrix material' but not in the other components of the inclusion bodies (Fig. 13), and it was suggested that the 'matrix material' may represent the site of virus protein synthesis. Specific but less dense labelling was observed over the cell wall outgrowths, and over other areas of cell wall.
Ecology and Control
Because PYFV depends on a helper virus for aphid-transmission, only those plant species that are hosts of both viruses are effective sources in nature. Aphids can transmit PYFV from the wild hedgerow plants Anthriscus sylvestris (anthriscus serotype of PYFV) and Heracleum sphondylium (parsnip serotype of PYFV). The helper virus in A. sylvestris is Anthriscus yellows virus (AYV) but that in H. sphondylium has been little studied and may differ from AYV, at least in host range. Although some cultivated umbelliferous species, such as chervil and coriander, are hosts of both AYV and PYFV, the three major crop hosts of PYFV in the UK (carrot, celery and parsnip) are immune to AYV, so that PYFV does not spread from plant to plant within these crops. Nevertheless, PYFV (parsnip serotype) was the most common virus found in parsnip in England in the 1960s (Murant & Goold, 1968) (although incidence seems now to be low), and in recent years the anthriscus serotype has been the most common virus in ware crops of carrot in the UK and Ireland. These high incidences of what must be primary PYFV infections probably reflect the very high populations of Cavariella spp. that develop in some seasons.
In the UK, severe PYFV outbreaks have occurred annually in carrot in recent years. In 1998, up to 30% of plants were killed, resulting in overgrowth of surviving plants and consequent further grading-out losses of 20% for early crops (N. Spence, personal communication). Estimates of average losses in 1998 were around 4%, equivalent to nearly £5 million (N. Spence, personal communication). In the Netherlands, PYFV-induced early season 'dieback' of carrot seed crops is common in most years and sometimes causes severe losses in seed production (Van Dijk & Bos, 1985).
In the UK, insecticide usage has hitherto been intensive in carrot crops, mainly for the control of carrot fly (Psila rosae), but undoubtedly also affecting aphid-borne viruses such as PYFV. The trend towards decreased use of insecticides and the spread of 'organic' methods of farming, may lead to increased virus incidence. Tests in the UK suggested that systemic insecticide sprays could prevent epidemics of PYFV in carrot if applied at the optimal time (N. Spence, personal communication), but in the Netherlands such treatment gave only limited control of the spread of PYFV in carrot seed crops (Van Dijk & Bos, 1985). Anthriscus sylvestris and Heracleum sphondylium are so widespread in hedgerows and waste land in western European countries that control of PYFV by the removal of sources of infection is impractical.
Notes
PYFV is readily distinguished from other viruses found in carrot, celery and parsnip in the UK, including Carrot mottle virus, Carrot red leaf virus, Celery mosaic virus, Parsnip mosaic virus and parsnip mottle virus (Watson et al., 1964), by its characteristic symptoms in Anthriscus cerefolium, Chenopodium amaranticolor, C. quinoa, Nicotiana benthamiana, N. clevelandii and Spinacia oleracea.
The only other known plant virus with properties similar to those of PYFV is Dandelion yellow mosaic virus (Bos et al., 1983; Murant, 1988). This virus, like PYFV, is transmitted by aphids in what may be a semi-persistent manner, and has isometric particles ca. 30 nm in diameter, with three particle proteins and a single genomic RNA, all of which are similar in Mr to those of PYFV. Murant (1988, 1991) proposed that these two viruses should be placed in a new taxonomic group, with PYFV as the type member. The group was named Sequivirus (Murant, 1993), from the Latin sequi (to follow, accompany or attend), in reference to the dependent aphid-transmission of PYFV. Reavy et al. (1993) noted that PYFV resembled the leafhopper-transmitted Rice tungro spherical virus (RTSV) in particle properties and genome structure, and that the disposition of the sequences of the coat proteins, NTP-binding, protease and polymerase motifs in the polyproteins of the two viruses resembled those of picornaviruses such as Poliovirus. Reavy et al. (1993) proposed that the two viruses should be placed together in a new family, but within separate genera. The viruses are now classified in the family Sequiviridae (Mayo et al., 2000), with PYFV as the type species of the genus Sequivirus (additional species: Dandelion yellow mosaic virus) and RTSV as the type species of the genus Waikavirus (additional species: Anthriscus yellows virus and Maize chlorotic dwarf virus).
Acknowledgements
I am indebted to Dr Nicola Spence for information about recent PYFV outbreaks in carrot, and for providing Fig. 3 and Fig. 4.
Figures

Fine vein-yellowing symptom caused by the parsnip serotype of PYFV in leaf of naturally infected Pastinaca sativa (parsnip). (Photograph: Scottish Crop Research Institute).

Yellow fleck symptom caused by the parsnip serotype of PYFV in leaf of naturally infected Pastinaca sativa (parsnip). (Photograph: Scottish Crop Research Institute).

`Viral dieback' symptom caused by infection of young carrot seedlings with the anthriscus serotype of PYFV. (Photograph: Horticulture Research International).

Cigar-shaped roots of carrot infected with the anthriscus serotype of PYFV; the root at the bottom shows the internal necrosis which is often associated with PYFV infection. (Photograph: Horticulture Research International).

Systemic necrosis of youngest leaf of Anthriscus cerefolium (chervil) aphid-inoculated with an isolate of the anthriscus serotype. (Photograph: Scottish Crop Research Institute).

Local lesions in Chenopodium amaranticolor 6 days after inoculation with a purified preparation of isolate A-421 (anthriscus serotype). (Photograph: Scottish Crop Research Institute).

Local lesions in Chenopodium quinoa 6 days after inoculation with isolate P-121 (parsnip serotype). (Photograph: Scottish Crop Research Institute).

Systemic symptoms in Nicotiana benthamiana 1 week after inoculation with isolate A-421 (anthriscus serotype). (Photograph: Scottish Crop Research Institute).

Systemic symptoms in Nicotiana clevelandii 2 weeks after inoculation with isolate P-121 (parsnip serotype). (Photograph: Scottish Crop Research Institute).

Local and systemic symptoms in Spinacia oleracea (spinach) 3 weeks after inoculation with isolate A-421 (anthriscus serotype). (Photograph: Scottish Crop Research Institute).

Particles mounted in 2% uranyl acetate. a) B component; b) T component. Bar represents 100 nm. (Photograph: Scottish Crop Research Institute).
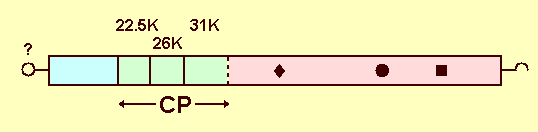
Genome stucture of the parsnip serotype of PYFV (isolate P-121). The box represents the polypeptide (3027 amino acids) encoded by the large ORF. The vertical lines show where cleavages are known (solid lines) or thought (dotted line) to occur. CP denotes the three coat proteins. Conserved domains are shown as: u, NTP-binding; l, protease; and n, polymerase. A VPg (¡) probably exists at the 5'-terminus, and there is a stem loop structure at the 3'-terminus [After Turnbull-Ross et al., 1993].

Portion of leaf cell of Spinacia oleracea infected with PYFV isolate P-121 (parsnip serotype), showing an inclusion body adjacent to the nucleus (N). The tissue was fixed with glutaraldehyde, treated with rabbit antibodies to isolate P-121, and labelled with goat anti-rabbit gold. The inclusion comprises granular bodies (G), tubules (T), and `matrix material' (M). The `matrix material' is heavily labelled and the nucleus (N) lightly labelled. Bar represents 2 µm. [From Fasseas et al., 1989].
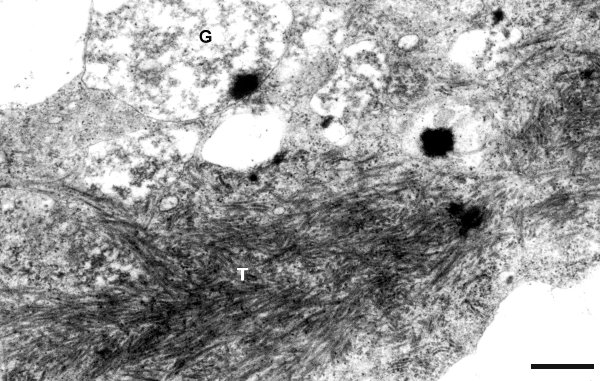
Detailed structure of an inclusion body from a leaf cell of Spinacia oleracea infected with isolate A-421 (anthriscus serotype), showing masses of tubules (T) and some granular bodies (G). Fixed with glutaraldehyde and OsO4, not gold-labelled. Bar represents 200 nm. [From Fasseas et al., 1989].
References list for DPV: Parsnip yellow fleck virus (394)
- Abu Salih, Ph.D. Thesis, University of St. Andrews, 1968.
- Bem & Murant, Annals of Applied Biology 92: 237, 1979.
- Bos, Huijberts, Huttinga & Maat, Netherlands Journal of Plant Pathology 89: 207, 1983.
- Elnagar & Murant, Annals of Applied Biology 84: 153, 1976a.
- Elnagar & Murant, Annals of Applied Biology 84: 169, 1976b.
- Fasseas, Roberts & Murant, Journal of General Virology 70: 2471, 1989.
- Hemida & Murant, Annals of Applied Biology 114: 87,1989a.
- Hemida & Murant, Annals of Applied Biology 114: 101, 1989b.
- Hemida, Murant & Duncan, Annals of Applied Biology 114: 71, 1989.
- Hollings, Journal of Horticultural Science 39: 130, 1964.
- Mayo, Murant, Reavy, Vetten & Reddick, in Virus Taxonomy. 7th Report of the International Committee on Taxonomy of Viruses, p. 685, eds M.H.V. van Regenmortel et al., New York: Academic Press, 2000.
- Murant, in The Plant Viruses. Vol. 3. Polyhedral Viruses with Monopartite RNA Genomes, p. 273, ed. R. Koenig, New York: Plenum, 1988.
- Murant, in Classification and Nomenclature of Viruses. Fifth Report of the International Committee on Taxonomy of Viruses, p. 318, eds R.I.B. Francki, C.M. Fauquet, D.L. Knudson & F. Brown, Vienna: Springer-Verlag, 1991.
- Murant, in Encyclopedia of Virology, p. 1042, eds R. G. Webster & A. Granoff, New York: Academic Press, 1993.
- Murant & Goold, Annals of Applied Biology 62: 123, 1968.
- Murant & Goold, Report of the Scottish Horticultural Research Institute for 1970: 53, 1971.
- Murant & Spence, in Compendium of Umbelliferous Crop Diseases, p. 56, eds R. M. Davis & R. N. Raid, St Paul, Minnesota: American Phytopathological Society, 2002.
- Murant, Roberts & Hutcheson, Journal of General Virology 26: 277, 1975.
- Murant, Roberts & Elnagar, Journal of General Virology 31: 47, 1976.
- Murant, Hemida & Mayo, Abstracts, 7th International Congress of Virology, Edmonton, Canada, p. 183, 1987.
- Reavy, Mayo, Turnbull-Ross & Murant, Archives of Virology 131: 441, 1993.
- Singh & Frost, Plant Pathology 36: 415, 1980.
- Turnbull-Ross, Reavy, Mayo & Murant, Journal of General Virology 73: 3203, 1992.
- Turnbull-Ross, Mayo, Reavy & Murant, Journal of General Virology 74: 555, 1993.
- Van Dijk & Bos, Netherlands Journal of Plant Pathology 91: 169, 1985.
- Van Dijk & Bos, Netherlands Journal of Plant Pathology 95 Supplement 2, 1989.
- Watson, Serjeant & Lennon, Annals of Applied Biology 54: 153, 1964.
- Wolf & Schmelzer, Zentralblatt für Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, Abteilung II 127: 665, 1972.