Details of DPV and References
DPV NO: 402 November 2003
Family: Pospiviroidae
Genus: Cocadviroid
Species: Coconut cadang-cadang viroid | Acronym: CCCVd
This is a revised version of DPV 287
Coconut cadang-cadang viroid
M. J. B. Rodriguez Philippine Coconut Authority, Guinobatan, Albay, Philippines
J. W. Randles Waite Campus, The University of Adelaide, South Australia, Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
The disease was first described in the Philippines by Ocfemia (1937) and the viroid was first identified by Randles (1975).
A viroid consisting of circular or linear single-stranded RNA with a basic size of 246 or 247 nucleotides, occurring as monomeric and dimeric forms in Cocos nucifera. Larger molecular forms appear as the disease progresses. Mechanically transmissible under laboratory conditions using nucleic acid inocula. Natural transmission occurs, possibly by mechanical damage and pollen. Host range is limited to the Palmae. Found only in the Philippines, where the disease has been responsible for lost production with the premature death of more than 40 million coconut palms. Annual losses are 200,000-400,000 palms and cost the industry around $40 million (Randles & Rodriguez, 2003)
Main Diseases
Cadang-cadang is a lethal disease of coconut (Cocos nucifera), recognised by a generalised yellow-bronze coloration of the lower two-thirds of fronds in the crown, persistence of stipules at the base of fronds, cessation of nut production, crown diminution and death (Fig.1). The third or fourth fronds below the spear leaf show small chlorotic leaf spots which expand and coalesce during ageing of the frond to produce a chlorotic mottle. As the spots appear, the nuts become smaller and rounded (Mohamed et al., 1982), and many of them develop equatorial scarification. Inflorescences later become necrotic and infertile, and nut production ceases. Frond production and size gradually decline (Randles et al., 1977), leaflets become brittle, and death of the crown ensues. Palms die between 8 and 16 years after the onset of symptoms (Zelazny & Niven, 1980). Cadang-cadang differs from the coconut Tinangaja disease in both symptomatology (Boccado et al., 1981) and in the nucleotide sequence of the associated Coconut tinangaja viroid.
Naturally-infected oil palm (Elaeis guineensis) shows a progression from general chlorosis on younger fronds to development of translucent orange leaf spots and cessation of inflorescence production (Randles et al., 1980).
Geographical Distribution
Cadang-cadang disease is limited to the central eastern Philippines (Quezon, Bicol provinces, Samar provinces and Biliran). Distribution is unaffected by water barriers, but incidence is negatively correlated with altitude (Zelazny, 1980). It has a slow rate of spread with new sites of incidence occurring within the confines of the above-mentioned areas (M.J.B. Rodriguez, E.P. Pacumbaba, J.C. Orense, A.R. Alfiler & G.G. Manalo, unpublished data).
Host Range and Symptomatology
Known hosts are confined to the Palmae. It is transmitted experimentally by high pressure injection of seedlings (two to three months old) with concentrated or partially-purified viroid, combined with pricking (Randles et al., 1977). For coconut, high pressure injection alone is more efficient when one to seven day old sprouts are used (Fig. 2) (Imperial et al. 1986).
- Diagnostic species
- Cocos nucifera (coconut). Inoculated seedlings show spotting one to two years after
inoculation, on leaflets of fronds that are at least three positions below the unexpanded spear
leaf. There is an associated reduction in growth rate, frond size and production and failure to bear
nuts at the expected time.
A more severe type of symptom first appeared in 1987, in a significant number of artificially inoculated coconut palms (Rodriguez & Randles 1993; Rodriguez 1998). Its main feature is a greater reduction in the width of the leaf lamina, giving the palm a 'broomed' appearance, and more rapid death (Fig. 3 & Fig. 4). Occasionally palms showing similar symptoms are observed in plantations, suggesting that 'brooming' occurs naturally.
Chlorotic or orange spotting of leaflets is characteristic of other inoculated species of palm, such as betel nut palm (Areca catechu), buri palm (Corypha elata), Manila palm (Adonidia merrillii), oil palm (Elaies guineensis), palmera (Chrysalidocarpus lutescens) and royal palm (Oreodoxa regia) (Imperial et al. 1985)
- Propagation species
- Naturally-infected Cocos nucifera.
- Assay species
- Cocos nucifera seedlings inoculated between seven days to three months after germination are used as the systemic assay host. Infection is confirmed by detection of the viroid in leaf extracts by gel electrophoresis (Fig. 5) or molecular hybridisation, one to two years after inoculation.
Strains
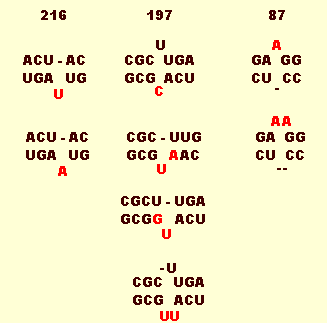
Coconut cadang-cadang viroid shows only minor variation in the minimal nucleotide sequence of 246 nucleotides (Fig. 6) (Haseloff et al., 1982; Rodriguez & Randles, 1993). Modifications to the right terminus are related to stage of disease development (Imperial et al. 1981) and are not strain differences because they revert to the minimal form when inoculated to new host seedlings. Mutations at positions 87, 197 and 216 are correlated with the expression of the severe lamina-depleting 'brooming' symptom (Fig. 6 & Fig. 7) (Rodriguez & Randles, 1993).
Transmission by Vectors
Natural transmission does occur, but no vector has been identified.
Transmission through Seed
Occurs at a low rate, with studies showing that only one out of 320 seedlings derived from infected material tested positive for CCCVd (Anon., 1982); low rate of pollen transmission (Pacumbaba et al., 1994).
Serology
No viroid-specific antibodies were detected in rabbits injected with the viroid-containing polyethylene glycol-insoluble fraction from coconut leaves (J.W. Randles & O.W. Barnett, unpublished data).
Relationships
Coconut cadang-cadang viroid is the smallest known nucleic acid containing pathogen. It is biologically and structurally distinct from all other viroids and is the type member of the genus Cocadviroid (Family Pospiviroidae). It has about 64% overall sequence identity with Coconut tinangaja viroid. In nucleic acid hybridisation assays, it shows some cross-hybridisation with Coconut tinangaja viroid, Potato spindle tuber viroid and Citrus exocortis viroid (Pospiviroidae) but not with Avocado sunblotch viroid (Avsunviroidae) (Hanold & Randles, 1991; J.W. Randles, R.A. Owens & J.A. Daros, unpublished data).
Stability in Sap
No information on survival of infectivity in sap. The structure of the viroid is more stable than that of cell RNA species in incubated polyethylene glycol concentrates of coconut leaf sap (Randles, 1975; Randles et al., 1976).
Purification
Chop leaflets from fronds four or more below the spear leaf and blend with 3 volumes of 0.1 M Na2SO3. Filter the extract through cloth mesh, clarify by centrifugation at 10,000 g for 10 min and add polyethlene glycol (PEG), M. Wt 6000, to a final concentration of 5% (w/v). After 1-2 h incubation at 4°C, centrifuge at low speed and extract nucleic acids from the precipitate by protease, phenol-sodium dodecyl sulphate, or chloroform procedures (Randles, 1975; Randles et al., 1976). The viroid may also be recovered directly from the PEG precipitate by resuspending it in 1 mM EDTA and precipitating the viroid with 3 vol. ethanol (J.W. Randles & J.S. Imperial, unpublished data). Subject extracts to electrophoresis in 5% polyacrylamide gels and detect the bands by staining in aqueous toluidine blue or ethidium bromide. Excise the bands and lay them over a gel containing 8 M urea (Randles et al., 1982). A second electrophoresis under these partially denaturing conditions separates circular and linear forms of the viroid monomer and dimer. Again excise the bands and extract the RNA by maceration and extraction of gel followed by sucrose density gradient centrifugation (Randles & Palukaitis, 1979), or by preparative gel electrophoresis, or by electrophoresis into agarose gel (Randles et al., 1982) followed by gel maceration and extraction. RNA obtained by either method is precipitated with ethanol, dried, resuspended in 0.2 M NaCl, then reprecipitated with 1% cetyl trimethyl ammonium bromide to remove gel residue which remains in solution.
Properties of Particles
Occurs as both linear and circular monomers and dimers, with sedimentation coefficients of 7 S and 10 S (Riesner et al., 1982). Monomers and dimers are infectious.
Monomer M. Wt is 83,000.
A260/A280 ratio: 2.1.
Buoyant density in Cs2SO4: approximately 1.60 g/cm3 (Randles et al., 1976).
Molecular Structure
Both the monomer and dimer are single-stranded RNAs, occurring as either covalently closed circular molecules or as linear molecules (Randles & Hatta, 1979). In the earliest stages of infection, CCCVd is detected as small forms, with monomeric sizes of either 246 (Accession No. J02049) or 247 nucleotides; the latter formed by the addition of a cytosine residue (Haseloff et al. 1982). The 246 form is more frequently observed as the initial infecting agent (Rodriguez et al., 1995). The dimer is detected simultaneously with the monomer. As infection develops these small forms are replaced by larger forms which arise through reiteration of a sequence at the 'right hand' end of the native molecule to give molecules 287-297 nucleotides in size (Accession No. J02051).
The native monomer shows two thermal transitions in 10 mM Na+, at 49° and 58°C; the first represents cooperative melting of the double-stranded regions to allow the formation of a stable intermediate, the second represents melting of the intermediate to yield a covalently linked open circle (Randles et al., 1982). The native viroid has 70% GC pairs, the half-width of the first thermal transition (DT1/2) is 1.4°C (Randles et al., 1982).
The reversible melting of the viroid to an open circle is the basis of the 2-dimensional polyacrylamide gel electrophoresis assay used for diagnosis (Schumacher et al., 1983).
Relations with Cells and Tissues
The viroid has been detected by molecular hybridisation in the inflorescence, in the meristems, in fronds of various ages and in roots (J.W. Randles & G. Boccardo, unpublished data). In infected oil palm, CCCVd is localised in the vascular bundles and in the nuclei of mesophyll cells. Within the nucleus both the viroid and its minus sense form are mainly concentrated in the nucleolus (Bonfiglioli et al. 1994, 1996). Light microscopy of yellow leaf spots shows hypoplasia and loss of chloroplast pigments, while electron microscopy shows the accumulation of dark structures (which are presumed to be tannin bodies), but no disease-specific cytoplasmic changes have been recognised (Randles, 1985).
Ecology and Control
Disease boundaries are quite stable. Epidemics occur at different times in different places with examples of formerly high incidence areas reverting to low incidence areas and new epidemics occurring in formerly low incidence areas. Patterns of disease increase vary from site-to-site. Sources of infection have not been identified. A negative correlation has been observed between incidence and altitude (Zelazny, 1980) but a positive correlation has been observed between palm age and incidence. Thus, palms less than 10 years old are rarely affected and the incidence increases nearly linearly up to palm ages of about 40 years, thereafter remaining constant (Zelazny et al., 1982). The beetles, Oryctes rhinoceros, Plesispa reichei and Hemipeplus sp. are more abundant in areas of high incidence (Zelazny & Pacumbaba, 1982) but no insects have been shown to transmit Cadang-cadang.
Eradication where only late stage palms were removed has failed to control the disease and in the absence of other management strategies, it is recommended that losses can be minimised by removing and replanting diseased trees as soon as early symptoms are recognised. To date, no resistance has been identified (Orolfo et al., 2000).
Notes
Coconut cadang-cadang is the most serious of all the viroid diseases because of its lethality and has not been found outside the Philippines. The viroid is not associated with Kerala wilt or Tatipaka disease of coconut palm in India, nor with coconut foliar decay in Vanuatu or coconut rapid decline in Sri Lanka (A.A.L. Perera & J.W. Randles, unpublished data). The similar but distinct Tinangaja disease of coconut, which is associated with Coconut tinangaja viroid infection, is reported only from Guam (Wall & Randles, 2003).
Figures

A stand of Cadang-cadang affected old palms in the Philippines together with a 10 year-old infected palm (indicated by the arrow).

The unusual 'brooming' symptom observed in an inoculated coconut palm with severe stunting and persistence of stipules at the base of leaf fronds. In the background is a healthy palm of the same age.

Reduced laminae from a palm with 'brooming' (lower) compared with leaflets from typical cadang-cadang infected (middle) and healthy palms (upper).

20% non-denaturing polyacrylamide gel electrophoresis-silver stain assay of leaf extracts from palms showing varying intensities of the 'brooming' symptom (lanes 1-5 and a mixture of their isolates in lane no. 6; M denotes Coconut cadang-cadang viroid marker consisting of the commonly detected variants 246, 247, 296 and 297 nucleotides.
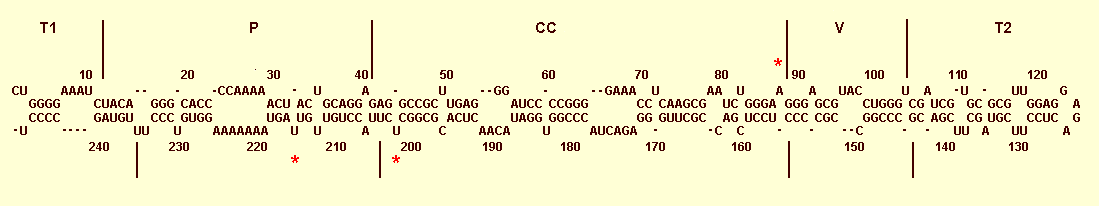
The primary and secondary structure of Coconut cadang-cadang viroid 246 with demarcation according to the proposed terminal (T1 and T2), pathogenicity (P), central conserved (CC) and variable (V) domains of Keese et al. (1988). The sites of mutations associated with the brooming symptom are denoted by red asterisks.
References list for DPV: Coconut cadang-cadang viroid (402)
- Anonymous, Cadang-cadang research, 1971-1982. Tech. Rep. F.A.O./U.N. Dev. Proj. AG:DP/ PH1/523, 75 pp., 1982.
- Boccardo, Beaver, Randles & Imperial, Phytopathology 71: 1104, 1981.
- Bonfiglioli, McFadden & Symons, Plant Journal 6: 99, 1994.
- Bonfiglioli, Webb & Symons, Plant Journal 9: 457, 1996.
- Hanold & Randles, Annals of Applied Biology 118:139, 1991.
- Haseloff, Mohamed & Symons, Nature 299: 316, 1982.
- Imperial, Bautista & Randles, Plant Pathology 34: 391, 1985.
- Imperial, Olfato & Rodriguez, PCA-ARDB Annual Report 1985: 7, 1986.
- Imperial, Rodriguez & Randles, Journal of General Virology 56: 77, 1981.
- Keese, Visvader & Symons, in RNA Genetics, Variability of RNA Genomes, ed. E. Domingo, J.J. Holland & P. Ahlquist, Boca Raton, CRC Press, Vol III, p. 71, 1988.
- Mohamed, Haseloff, Imperial & Symons, Journal of General Virology 63: 181, 1982.
- Ocfemia, Philipp. Agric. The Philippine Agriculturist 26: 338, 1937.
- Orolfo, Estioko & Rodriguez, Report, Philippine Coconut Authority, 2000.
- Pacumbaba, Zelazny, Orense & Rillo, Phytopathology 142: 37, 1994.
- Randles, Phytopathology 65: 163, 1975.
- Randles, in Subviral Pathogens of Plants and Animals: Viroids and Prions, ed. K. Maramorosch & J.J. McKelvey, New York, Academic Press, p. 39, 1985.
- Randles & Hatta, Virology 96: 47, 1979.
- Randles & Palukaitis, Journal of General Virology 43: 649, 1979.
- Randles & Rodriguez, in Viroids, ed. A. Hadidi, R. Flores, J.W. Randles & J.S.Semancik, Collingwood, Australia, CSIRO Publishing, p. 233, 2003.
- Randles, Rillo & Diener, Virology 74: 128, 1976.
- Randles, Boccardo, Retuerma & Rillo, Phytopathology 67: 1211, 1977.
- Randles, Boccardo & Imperial, Phytopathology 70: 185, 1980.
- Randles, Steger & Riesner, Nucleic Acids Research 10: 5569, 1982.
- Riesner, Kaper & Randles, Nucleic Acids Research 10: 5587, 1982.
- Rodriguez, in Report on ACIAR-funded research on viroids and viruses of coconut palm and other tropical monocotyledons 1985-1993, ed. D. Hanold & J.W. Randles, ACIAR Monograph No. 45, p. 90, 1998.
- Rodriguez, Estioko & Manalo, The Philippine Journal of Science 124; 1, 1995.
- Rodriguez & Randles, Nucleic Acids Research 21: 2771, 1993.
- Schumacher, Randles & Riesner, Analytical Biochemistry 135: 288, 1983.
- Wall & Randles, in Viroids, ed. A. Hadidi, R. Flores, J.W. Randles & J.S.Semancik, Collingwood, Australia, CSIRO Publishing, p. 242, 2003.
- Zelazny, Phytopathology 70: 700, 1980.
- Zelazny & Niven, Plant Disease 64: 841, 1980.
- Zelazny & Pacumbaba, Ecological Entomology 7: 113. 1982.
- Zelazny, Randles, Boccardo & Imperial, Scientia Filipinas 2: 45, 1982.