Details of DPV and References
DPV NO: 405 April 2004
Family: Bromoviridae
Genus: Bromovirus
Species: Brome mosaic virus | Acronym: BMV
This is a revised version of DPV 180
Brome mosaic virus
Rachel S. Wooley Department of Biochemistry and Biophysics, Texas A&M University, College Station, TX 88743-2128, USA.
C. Cheng Kao Department of Biochemistry and Biophysics, Texas A&M University, College Station, TX 88743-2128, USA.
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by McKinney et al. (1942).
- Selected synonyms:
- Marmor graminis (McKinney, 1944)
- Weidelgrasmosaik-Virus (Ohmann-Kreutzberg et al., 1960)
- Ryegrass streak virus (Proll & Schmidt, 1964)
This positive-sense single-stranded RNA virus has been extensively studied for decades and serves as a model of viral RNA replication, recombination, gene expression and virion assembly in plus-stranded RNA viruses.
Main Diseases
BMV is not considered an economically important plant pathogen. It can cause brown streaks in monocots and may decrease seed yield.
Geographical Distribution
BMV is found in Africa, North America, Europe and Asia.
Host Range and Symptomatology
BMV infects many monocots in the family Poaceae, including Bromus inermis, from which the virus was first isolated, Zea mays and Hordeum vulgare. In barley, BMV causes yellow to tan coloured streaks (Fig. 1). BMV can also infect the dicots Nicotiana benthamiana and several Chenopodium species. In Chenopodium species, the infection is localized and appears as chlorotic spots (Fig. 2). A complete host range has been tabulated (Lane, 1974).
- Diagnostic species:
- Zea mays (maize). Seedlings of most varieties show primary lesions or streaks (Fig. 3) followed by necrosis and death (Fig. 4).
- Chenopodium spp. BMV is one of the few grass viruses that produce local lesions in C. amaranticolor, C. hybridum and C. quinoa (Fig. 2).
- Propagation species:
- Hordeum vulgare (barley).
- Assay species:
- Chenopodium hybridum (Rochow, 1959) and C. quinoa (Fig. 2) are local lesion hosts.
Strains
The isolate of McKinney et al. (1942) is regarded as the type strain. Clearly distinct natural isolates have not been found, although slight differences in stability and other properties have been noted (Lane, 1974).
Transmission by Vectors
BMV is transmitted primarily by mechanical inoculation, although dagger nematodes (genus Xiphinema) have been shown to transmit the virus in the laboratory (Schmidt et al., 1963).
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
Antisera with dilution end-points greater than 1/1000 have been prepared but antibody titres drop sharply after reaching a maximum (Moorhead, 1956). Serological studies are complicated by the instability of the virus above pH 6 and its tendency to interact ionically with the sulphate groups of agar and agarose below this pH. Under appropriate conditions, antisera specific to intact virus particles or to the dissociated coat protein can be prepared (von Wechmar & van Regenmortel, 1968).
Relationships
Two strains of BMV originally isolated from Russia and Japan are more than 97% identical in nucleotide sequence. BMV is distantly serologically related to other members of the genus Bromovirus, with which it also shares similar physical properties and coat protein amino acid sequences. The ability of RNA-1 and RNA-2 from BMV and RNA-3 from Cowpea chlorotic mottle virus to form a viable genetic hybrid, albeit with a much reduced growth rate and host range (Bancroft, 1972), also indicates a relationship.
Stability in Sap
The properties of the virus in sap vary. On the average, infectivity survives 10 min at 80 °C or dilution to 10-4 or 10-5 (Lane, 1977).
Purification
Easily purified with or without an ultracentrifuge. A simple method is to grind barley tissue (roughly 2 weeks after infection) with an equal weight of 0.5 M sodium acetate, 8 mM magnesium acetate, pH 4.5 and then emulsify with a small amount of chloroform. Centrifuge for 10 min at 5000 rev/min. Filter the supernatant fluid and precipitate the virus by adding polyethylene glycol M.Wt 6000 to 6 % (w/v). After 15 min of stirring on ice, collect the precipitate by centrifuging for 15 min at 5000 rev/min. Resuspend the pellet in distilled water to about 1/10 the original volume and emulsify with a few ml of chloroform. Centrifuge the emulsion for 10 min at 5000 rev/min and precipitate virus from the upper phase by adding polyethylene glycol to 6% (w/v) and mixing with 1/10 volume of 5 M NaCl. After 15 min of stirring on ice, collect the precipitate by centrifugation and resuspend in 50 mM sodium acetate, 1 mM magnesium acetate, pH 4.5 (buffer A). Virus may be further purified by differential centrifugation (90 min at 40,000 rev/min and 5 min at 20,000 rev/min) (Lane, 1977). An alternative method is to sediment the virions through a 10% (w/v) sucrose cushion. Briefly, the virus solution is placed in an SW28 or SW41 tube (with capacities of 8 and 20 ml, respectively) and underlayed with 1 to 3 ml of 10% sucrose (made in buffer A). The virus pellet obtained after centrifugation for 1 h at 100,000 g can be suspended in the desired volume of buffer A.
Purified virus may be stored frozen by adding 1/20 volume of ethylene glycol as a cryopreservative. Contaminating nucleases gradually degrade RNA within the virus. Quality of virus preparations is best judged by analyzing the integrity of the RNA by gel electrophoresis (Lane, 1974).
Properties of Particles
BMV is stable in acetate buffer containing Mg2+ at a pH between 3.0 and 6.0 (Lucas et al., 2002). Above pH 7 it swells and is degraded by contaminating nucleases (Incardona & Kaesberg, 1964).
Sedimentation coefficient (s20, w): (87.3-0.47 c) S at pH 3-6 and (78.7-0.64 c) S at pH 7 and above (Incardona & Kaesberg, 1964), where c is the virus concentration in mg/ml.
Molecular Weight: 4.6 x 106 (Bockstahler & Kaesberg, 1962).
The isoelectric point varies considerably with ionic strength; it is pH 6.8 by isoelectric focusing (Rice & Horst, 1972).
Partial specific volume: 0.71 cm3/g (estimated, Bockstahler & Kaesberg, 1962).
Electrophoretic mobilities as a function of pH are given by Bockstahler & Kaesberg (1962) and Johnson et al. (1973).
Extinction coefficient (E(0.1%, 1cm)) at 260 nm, uncorrected for light-scattering is 5.15 (Bockstahler & Kaesberg, 1962).
A260/A280 is 1.75. Amax/min is 1.53 (Lane, 1977).
The buoyant density in CsCl is roughly 1.35 g/cm3 (Bancroft, 1971).
Particle Structure
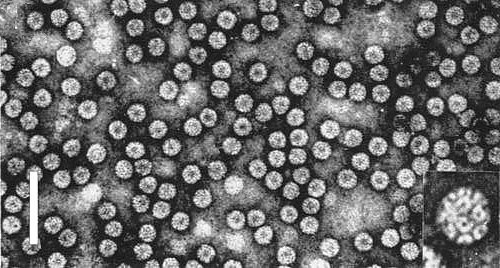
The BMV virion is composed of 180 identical subunits of the capsid protein arranged in a T=3 icosahedral geometry (Fig. 5). The virion structure was determined by X-ray crystallography to a resolution of 0.34 nm (Lucas et al., 2002). The capsid subunits exist in three different arrangements that help form 12 pentameric capsomeres and 20 hexameric capsomeres. At the center of each capsomere is a channel of 0.5-0.6 nm diameter. The outer and inner diameters of the capsid measure approximately 28 nm and 18 nm, respectively. The capsid itself has a thickness of 5-6 nm and weighs roughly 3.6 x 106 kDa (Casselyn et al., 2001). Mg2+ stabilizes the capsid by binding the centres of trimers. Within the virion, two concentric rings of density have been observed, possibly representing viral RNA. RNA-1 and RNA-2 are separately encapsidated. RNA-3 and RNA-4 are usually co-packaged into one particle. RNA-1, -2 and -3 are required to infect (Lane & Kaesberg, 1971), and therefore three different viral particles are needed for successful infection.
Particle Composition
Virus particles are approximately 22% nucleic acid and 78% protein (Bockstahler & Kaesberg, 1962). The four single-stranded RNA species have M.Wts of 1.1 x 106 (RNA-1), 1.0 x 106 (RNA-2), 0.7 x 106 (RNA-3), and 0.3 x 106 (RNA-4). The protein weighs 20,300 daltons. The virus particles contain no polyamines (Nickerson & Lane, 1977).
Genome Properties
The BMV RNAs are capped at the 5' termini and contain 3' sequences of approximately 200 nts that mimic tRNA structures. Schematics of the three BMV RNAs are shown in Fig. 6. The following refers to the Russian strain, but other strains are very similar.
RNA-1 (3234 nt; Acc. No. X02380). RNA-1 codes for one known protein, 1a (961 amino acids). This protein has been shown to be involved in RNA capping and in binding BMV RNA, probably for the formation of membrane-associated replication structures (Schwartz et al., 2002). In addition, 1a contains helicase-like motifs, but RNA helicase activity remains to be demonstrated.
RNA-2 (2865 nt; Acc. No. X01678). RNA-2 codes for one known protein, 2a (822 amino acids). This protein is present in the BMV replicase and is probably the RNA-dependent RNA polymerase responsible for nucleotide polymerization.
RNA-3 (2117 nt; Acc. No. V00099). RNA-3 codes for two known proteins: 3a (303 amino acids), which is required for virus movement in the plant, and the capsid protein (189 amino acids), which is required for encapsidation of BMV RNAs and cell-to-cell movement (Rao, 1997). The latter protein is translated from a subgenomic RNA, RNA-4, whose synthesis is regulated by a noncoding region (nt 1004 - 1250) between the protein-coding sequences (Fig. 6).
Sequences required for packaging of BMV RNA-3 and RNA-4 have been investigated (Choi et al., 2002; Choi and Rao, 2003; Damayanti et al., 2003). However, the two laboratories have identified different packaging signals in BMV RNA-3, and thus the requirements for RNA packaging remain to be fully elucidated.
BMV replication requires the synthesis of three classes of RNA: genomic minus-, genomic plus-, and subgenomic plus-strand RNAs. The sequences that bind the replicase and direct viral replication are reviewed in Kao (2002). The synthesis of all BMV RNAs is initiated by a de novo mechanism using GTP as the first nucleotide (Kao et al., 2001). BMV RNA recombination is achieved primarily by a copy-choice mechanism, reviewed by Bujarski et al. (1994).
BMV can replicate in the budding yeast, S. cerevisiae (Janda & Ahlquist, 1993), allowing the identification of the host genes required for BMV replication in a genetically amenable host. S. cerevisiae genes that affect BMV replication and transcription are identified in Kushner et al. (2003). In S. cerevisiae, the BMV replicase complex is localized in shallow cave-like structures in the nuclear membrane (Schwartz et al., 2002).
Relations with Cells and Tissues
In tobacco protoplasts, the endoplasmic reticulum around the nucleus proliferates during the first 6 h of infection. Later, virus particles are found scattered throughout the cytoplasm. Some virus particles associate in a helical array around the outside of membranous tubules which are about 30 nm in diameter (Burgess et al., 1974). In oat and barley leaves, photosynthetic tissue (mesophyll cells) is more severely affected than other tissues, and virus is often seen in chloroplast invaginations. Both hosts contain virus crystals at late stages in the disease. Chloroplasts degenerate in yellowed areas of the leaves (Paliwal, 1970).
Notes
BMV is distinguishable from most other viruses of Poaceae by its symptoms in maize and by its ability to infect several non-graminaceous hosts. In view of the ease of purification (it can be purified in high yield from naturally infected bromegrass), cursory physical characterization is advisable for purposes of identification.
Figures
References list for DPV: Brome mosaic virus (405)
- Bancroft, Virology 45: 830, 1971.
- Bancroft, Journal of General Virology 14: 223, 1972.
- Bockstahler & Kaesberg, Biophysical Journal 2: 1, 1962.
- Bujarski, Nagy & Flasinski, Advances in Virus Research 43: 275, 1994.
- Burgess, Motoyoshi & Fleming, Planta 117: 133, 1974.
- Casselyn, Perez, Tardieu, Vachette, Witz & Delacroix, Acta Crystalographica 57: 1799, 2001.
- Choi & Rao, Journal of Virology 77: 9750, 2003.
- Choi, Dreher & Rao, Proceedings of the National Academy of Sciences, USA 99: 655, 2002.
- Damayanti, Tsukaguchi, Mise & Okuno, Journal of Virology 77: 9979, 2003.
- Incardona & Kaesberg, Biophysical Journal 4: 11, 1964.
- Janda & Ahlquist, Cell 72: 961, 1993.
- Johnson, Wagner & Bancroft, Journal of General Virology 19: 263, 1973.
- Kao, Molecular Plant Pathology 3: 55, 2002.
- Kao, Sighn & Ecker, Virology 287: 251, 2001.
- Kushner, Lindenbach, Grdzelishvili, Noueiry, Paul & Ahlquist, Proceedings of the National Academy of Sciences, USA 100: 15764, 2003.
- Lane, Advances in Virus Research 19: 151, 1974.
- Lane, CMI/AAB Descriptions of Plant Viruses 180, 1977.
- Lane & Kaesberg, Nature New Biology 232: 40, 1971.
- Lucas, Larson & McPherson, Journal of Molecular Biology 317: 95, 2002.
- McKinney, Journal of the Washington Academy of Sciences 34: 139, 1944.
- McKinney, Fellows & Johnston, Phytopathology 32: 331, 1942.
- Moorhead, Phytopathology 46: 498, 1956.
- Nickerson & Lane, Virology 81: 455, 1977.
- Ohmann-Kreutzberg, Pawlitscher & Schmidt, Phytopathologische Zeitschrift 38: 13, 1960.
- Paliwal, Journal of Ultrastructure Research 30: 491, 1970.
- Proll & Schmidt, Virology 23: 103, 1964.
- Rao, Virology 232: 385, 1997.
- Rice & Horst, Virology 49: 602, 1972.
- Rochow, Phytopathology 49: 126, 1959.
- Schmidt, Fritsche & Lehmann, Naturwissenschaften 50: 386, 1963.
- Schwartz, Chen, Janda, Sullivan, den Boon & Ahlquist, Molecular Cell 9: 505, 2002.
- von Wechmar & van Regenmortel, Virology 34: 36, 1968.