Details of DPV and References
DPV NO: 406 December 2004
Family: Caulimoviridae
Genus: Tungrovirus
Species: Rice tungro bacilliform virus | Acronym: RTBV
This is a revised version of DPV 67
Rice tungro bacilliform virus
Roger Hull Department of Disease and Stress Biology, John Innes Centre, Norwich Research Park, Colney, Norwich NR4 7UH, UK
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Rice tungro disease was first described by Anon. (1964) and Rivera and Ou (1965). Rice tungro bacilliform virus (RTBV) was first described by Saito et al. (1975). The role of RTBV as a member of the virus complex causing tungro disease was first reported by Hibino et al. (1978).
- Selected synonyms (these refer to rice tungro disease)
- Accep na pula (Philippines) (Serrano, 1957)
- Mentek (Indonesia) (Ou, 1965)
- Penyakit merah (Malaysia) (Singh,1967)
- Yellow orange leaf (Thailand) (Lamey et al., 1967)
- Leaf yellowing (India) (Raychaudhuri et al., 1967)
A virus with bacilliform particles about 30 nm in diameter and a usual length of 130 nm. The particles contain a single molecule of circular dsDNA of 8 kbp, and a single coat protein species of Mr 33,000 –37,000. The virus occurs in Oryza species in South and South-East Asia. RTBV is not mechanically transmissible but, in a dependent association with Rice tungro spherical virus (RTSV), it is transmitted in the semi-persistent manner by leafhoppers. The virus complex causes a severe disease of rice (tungro), RTBV being responsible for most of the symptoms and RTSV providing the leafhopper transmissibility.
Main Diseases
On its own, or in mixed infections with RTSV, RTBV induces tungro disease, a severe and economically important disease of rice throughout South and South-East Asia. Annual losses due to tungro disease in South-East Asia have been estimated to exceed $1.5 billion (Herdt, 1991).
Geographical Distribution
The virus is found in most, if not all, rice growing countries of South and South-East Asia. Its distribution is likely to be related to that of the major leafhopper vectors of RTSV.
Host Range and Symptomatology
Early host range studies on RTBV are unreliable as its association with RTSV was not recognized. Host range studies are also constrained by the ability of the vector species to transmit the virus to the plant species tested. RTBV appears to have a restricted host range limited to members of the Poaceae and Cyperaceae (Echinochloa crus-galli, E. glabescens, E. colona, Eleusine indica, Leptochloa chinensis, Leersia hexandra, Oryza sativa, Panicum repens, Cyperus rotundus) (Khan et al., 1991).
Symptoms induced by RTBV alone in rice vary with cultivar but, in most, infection causes stunting of the plant, red or orange colouring of the leaves and a reduction in tiller number (Fig. 1). Early infection can result in the death of some cultivars. Joint infection with RTSV enhances the symptoms somewhat. Field infection is characterised by a patchy distribution (Fig. 2).
The virus can be propagated in rice cultivar Taichung Native 1 (TN1).
Strains
The type strain can be considered to be the Philippines isolate that was the first sequenced (Hay et al., 1991). Four biological variants of RTBV (L, G1, G2 and Ic) have been described based on their characteristic symptoms in rice cultivars FK 135 and TN1 (Cabauatan et al., 1995).
RTBV isolates from South Asia (India, Sri Lanka and Bangladesh) differ in sequence from those from South-East Asia (Fan et al., 1996; Druka & Hull, 1998). There is microvariation in the sequences of isolates from the Philippines and other countries (Villegas et al., 1997; Azzam et al., 2000).
Transmission by Vectors
RTBV is transmitted by leafhoppers only when RTSV was acquired previously or at the same time suggesting that RTSV may have a helper component (Cabauatan & Hibino, 1985); leafhoppers carrying RTSV alone can acquire RTBV from singly-infected plants. Thus, in tungro disease, it is transmitted in the semi-persistent manner by several species of leafhopper. The major vector over much of South-East Asia is the rice green leafhopper, Nephotettix virescens (synonym N. impicticeps); it is also transmitted by N. cincticeps, N. nigropictus, N. malayanus and Recilia dorsalis. These other vectors may be of relative importance in certain situations. N. virescens can acquire RTBV (in the presence of RTSV) in 30 min and can retain infectivity for up to 4 days (Anjaneyulu et al., 1995). Nymphs transmit more efficiently than adults and females more efficiently than males. Transmission efficiency is also affected by temperature, increasing as the temperature rises from 10 to 34°C.
Cloned RTBV genome can also be transmitted by agroinoculation (Dasgupta et al., 1991; Sta. Cruz et al., 1999).
Transmission through Seed
No evidence for seed transmission.
Serology
RTBV is moderately immunogenic and the antisera have been used for ELISA, western blots and immunogold electron microscopy (Bajet et al., 1985; Cabauatan & Hibino, 1988; Hay et al., 1994). An antiserum has also been raised to part of the coat protein expressed in E. coli (Hay et al., 1994).
Relationships
Most of the physical and chemical properties of RTBV are similar to those of members of the genus Badnavirus; however, its genome contains four open reading frames whereas those of badnaviruses have only three. No relationships have been shown between RTBV and other viruses.
Biological strains of RTBV can be differentiated by their symptom production in rice cultivars FK 135 and TN1 (Cabauatan et al., 1995). Geographical strains are differentiated by hybridisation, by restriction endonuclease mapping of the genomic DNA, and by the polymerase chain reaction targeted at a deletion in the genome of the South Asia strain (Fan et al., 1996; Druka & Hull, 1998).
Stability in Sap
As the virus is not sap transmissible, there have been no studies on its stability in sap.
Purification
1. Method of Cabauatan & Hibino (1988):
Harvest about 500 g rice plants 50 days after infection and homogenize in 1 L 0.01 M sodium
ethylene-diamine-tetra-acetate (EDTA), pH 8.0. Incubate the extract with 3 g Driselase
(Kyowa Hakko Kogyo Co. Ltd., Tokyo) for 1 h at room temperature, 1 h at 40 °C, and then centrifuge for
10 min at 15,000g. Take the supernatant fluid, add polyethylene glycol (mol. wt 8000), NaCl
and Triton X-100 to 7%, 0.2 M and 1% respectively, and stir the mixture for 1 h at room temperature.
Centrifuge at 30,000g for 30 min, resuspend the pellet in 20 ml cold 0.01 M EDTA,
pH 8.0, and centrifuge at 11,000g for 10 min; centrifuge the supernatant fluid at
100,000g for 60 min. Resuspend the pellet in 2 ml cold 0.01 M phosphate buffer (PB),
pH 7.4, layer it on 10-50% sucrose density gradients (in PB) and centrifuge in a Beckman SW27 rotor at
25,000 rev./min for 2.5 h at 4 °C. Recover the virus-containing band, dilute in 0.01 M PB and centrifuge
at 130,000g for 1 h. Suspend the resulting pellet in 1 ml PB and after
centrifugation at 11,000g for 10 min the virus is found in the supernatant fluid.
2. Method of Jones et al. (1991):
Freeze infected rice leaves in liquid N2, grind and thaw each 100 g tissue into 400 ml 0.1 M
sodium citrate, pH 5.9. Add Celluclast (Novo Enzymes) to 5% (v/v), incubate the mixture at 30 °C for
2 h and centrifuge at 10,000 rev./min for 10 min. Resuspend the pellet in 200 ml citrate buffer and
5% Celluclast, incubate for 1 h at 30 °C and centrifuge as above. Pool supernatant fractions from both
centrifugations and add polyethylene glycol (mol. wt 6000), NaCl and Triton X-100 to give 7%, 0.2 M and
1% respectively. Incubate the mixture for 2 h at room temperature, then centrifuge at 12,000 rev./min for
10 min. Resuspend the pellet in 20 ml citrate buffer and centrifuge through a 5% sucrose cushion at
36,000 rev./min for 2.5 h in a Beckman Ti40 rotor at 4 °C. Resuspend the pellet in 4 ml citrate buffer.
Further purification and, if necessary, separation from RTSV can be effected by rate zonal or isopycnic
gradient centrifugation.
Properties of Particles
The particles sediment with an so20,w of about 200 S and have a buoyant density in CsCl of approximately 1.36 g ml-1 (Hull, 1996). Other physical properties have not been determined.
Particle Structure
The virus particles are bacilliform of diameter 30 nm and predominant length 130 nm (Fig. 3); longer particles, up to 300 nm, are found in some isolates. The particle structure is based on a T = 3 icosahedron cut across its three-fold axis, the tubular portion being made up of rings of hexamer subunits with a repeat distance of about 10 nm (Hull, 1996).
Particle Composition
Nucleic acid:
The virus particles contain a single species of circular dsDNA that has one discontinuity at a specific
position in each strand
(Bao & Hull, 1992).
The complete nucleotide sequences have been determined for six isolates from the Philippines: 8002 bp
(accession number X57924)
for an infectious clone
(Hay et al., 1991), 8000 bp
(accession number M65026) for an unnamed isolate
(Qu et al., 1991), 8002 bp
(accession number D10774) for the WG isolate
(Kano et al., 1992), and 8006, 8001 and 8005 bp
(accession numbers AF113830,
AF113831,
AF113832) for the G1, G2 and Ic isolates respectively
(Cabauatan et al., 1999).
Complete sequences have also been determined for three isolates from other countries: 8010 bp
(accession number AF220561) for the Chainat isolate from Thailand
(Nathwong, B., Ratankarn, W. & Chettanachit S., unpublished data),
8016 bp (accession number AF076470) for the Serdang isolate from Malaysia
(Marmey et al., 1999), and 7934 bp
(accession number AF314596) for the West Bengal, India isolate
(Nath et al., 2002).
Coat protein:
Polyacrylamide gel electrophoresis of viral coat protein reveals a major coat protein species of
Mr 37,000; this may be processed to give a product of Mr about 33,000 and
possibly other bands
(see Hull, 1996).
The molecular mass of the coat protein has been measured by mass spectrometry and the N- and C-terminal
sequences have been determined
(Marmey et al., 1999).
This protein contains two characteristic amino acid sequence motifs:
CXCX2CX4HX4C, characteristic of reverse transcribing virus
gag proteins, and CX2CX11CX2CX4CX2C,
unique to "badnaviruses".
Genome Properties
The RTBV genome contains four open reading frames (ORFs) (reviewed in Hull, 1996) (Fig. 4). The functions of the products of ORFs 1 and 2, of Mr 24,000 and 12,000 respectively, are unknown. ORF 3 encodes a polyprotein that is processed by an aspartate protease to give: N-terminal protein(s) thought possibly to be cell-to-cell movement protein(s), viral coat protein, aspartate protease and the replicase comprising reverse transcriptase and RNaseH activities. The ORF 4 product of Mr 46,000 has no known function.
The viral genome is transcribed asymmetrically to give a "more-than-genome-length" RNA that is the template for the reverse transcription phase of replication and for the expression of ORFs 1 - 3; ORF 4 is expressed from a spliced version of this RNA (reviewed in Hull, 1996).
Relations with Cells and Tissues
RTBV is restricted to vascular tissue and is found in sieve tubes, phloem and xylem parenchyma cells and companion cells (Sta. Cruz et al., 1993); particles are more abundant in phloem cells than in xylem cells. Virus particles appear as single particles or aggregates of tubules of varying length in the cytoplasm (Fig. 5). No inclusion bodies are observed in RTBV infections.
Ecology and Control
RTBV causes no significant damage in most rice cultivars. However, it relies on RTSV for its transmission in the economically important tungro complex and thus RTSV has also to be considered in relation to the disease. The complex spreads from old to seedling plants in overlapping cropping systems and is likely to spread from latent infections in weeds harbouring the leafhopper, Nephotettix virescens, surrounding rice paddies. Tungro infection occurs in periodic epidemics (Savary et al., 1993; Anjaneyulu et al., 1995).
Tungro disease is controlled either by use of insecticides (see Anjaneyulu et al., 1995) or by resistance breeding. Rice breeding programmes have attempted to introduce resistance to both tungro viruses into elite cultivars. This resistance can be to the vector, which is not very durable (Dahal et al., 1990), or resistance to RTBV found in varieties such as Utri Merah (Hibino et al., 1988). Resistance to RTSV can limit the spread of tungro disease.
Figures

Genome organization of RTBV. The inner two circles represent the dsDNA with arrow showing the position of the promoter for the major transcript. The positions of the four open reading frames (ORFs) are indicated by the arrowed boxes.
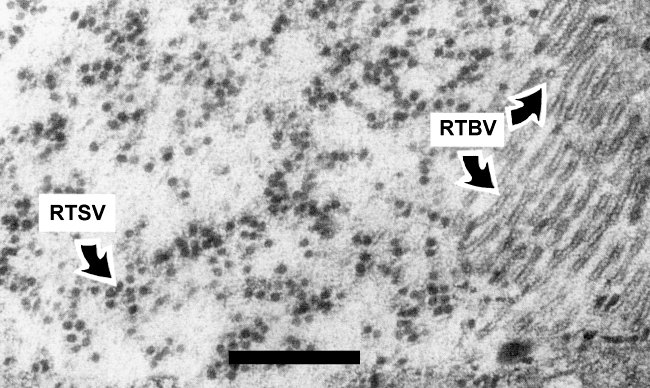
Thin section of rice plant, cultivar TN1, infected with RTBV and RTSV, showing particles of both viruses in the cytoplasm. Bar represents 400 nm. (From Sta. Cruz et al., 1993).
References list for DPV: Rice tungro bacilliform virus (406)
- Anjaneyulu, Satapathy & Shukla, Rice tungro. Science Publishers, Inc., Lebanon, New Hampshire, 228 pp., 1995.
- Anon., Report of the International Rice Research Institute, 1963, 1964.
- Azzam, Arboleda, Umadhay, de los Reyes, Cruz, Mackenzie & McNally, Archives of Virology 145: 2643, 2000.
- Bajet, Daquioag & Hibino, Journal of Plant Protection in the Tropics 2: 125, 1985.
- Bao & Hull, Journal of General Virology 73: 1297, 1992.
- Cabauatan & Hibino, Philippine Phytopathology 21: 31, 1985.
- Cabauatan & Hibino, Plant Disease 72: 526, 1988.
- Cabauatan, Cabunagan & Koganezawa, Phytopathology 85: 77, 1995.
- Cabauatan, Melcher, Ishikawa, Omura, Hibino, Koganezawa & Azzam, Journal of General Virology 80: 2229, 1999.
- Dahal, Hibino & Saxena, Phytopathology 80: 371, 1990.
- Dasgupta, Hull, Eastop, Poggi-Pollini, Blakebrough, Boulton & Davies, Journal of General Virology 72: 1215, 1991.
- Druka & Hull, Journal of Phytopathology 146: 175, 1998.
- Fan, Dahal, Dasgupta, Hay & Hull, Journal of General Virology 77: 847, 1996.
- Hay, Jones, Blakebrough, Dasgupta, Davies & Hull, Nucleic Acids Research 19: 2615, 1991.
- Hay, Grieco, Druka, Pinner, Lee & Hull, Virology 205: 430, 1994.
- Herdt, Rice Biotechnology, Eds Khush & Toenniessen, pp. 19-54, CABI, Wallingford, UK, 1991.
- Hibino, Roechan & Sudarisman, Phytopathology 68: 1412, 1978.
- Hibino, Daquioag, Cabauatan & Dahal, Plant Disease 72: 843, 1988.
- Hull, Annual Review of Phytopathology 34: 275, 1996.
- Jones, Gough, Dasgupta, Subba Rao, Cliffe, Qu, Shen, Kaniewska, Blakebrough, Davies, Beachy & Hull, Journal of General Virology 72: 757, 1991.
- Kano, Koizumi, Noda, Hibino, Ishikawa, Omura, Cabauatan & Koganezawa, Archives of Virology 124: 157, 1992.
- Khan, Hibino, Aguiero & Daquioag, Plant Disease 75: 926, 1991.
- Lamey, Surin & Leewangh, International Rice Communication Newsletter 16: 15, 1967.
- Marmey, Bothner, de Kochko, Ong, Yot, Siuzdak, Beachy & Fauquet, Virology 253: 319, 1999.
- Nath, Mathur & Dasgupta, Archives of Virology 147: 1173, 2002.
- Ou, International Rice Communication Newsletter 14: 4, 1965.
- Qu, Bhattacharyya, Laco, de Kochko, Subba Rao, Kaniewska, Elmer, Rochester, Smith & Beachy, Virology 185: 354, 1991.
- Raychaudhuri, Mishra & Ghosh, Plant Disease Reporter 51: 300, 1967.
- Rivera & Ou, Plant Disease Reporter 49: 127, 1965.
- Saito, Roechan, Tantera & Iwaki, Phytopathology 65: 793, 1975.
- Savary, Fabellar, Tiongco & Teng, Plant Disease 77: 376, 1993.
- Serrano, Philippine Journal of Science 86: 203, 1957.
- Singh, In Proceedings of Symposium of the Virus Diseases of Rice Plants, Los Banos, Philippines, p. 322, 1967.
- Sta. Cruz, Koganezawa & Hibino, Journal of Phytopathology 138: 274, 1993.
- Sta. Cruz, Boulton, Hull & Azzam, Journal of Phytopathology 147: 653, 1999.
- Villegas, Druka, Bajet & Hull, Virus Genes 15: 195, 1997.


