Details of DPV and References
DPV NO: 416 March 2007
Family: Geminiviridae
Genus: Mastrevirus
Species: Maize streak virus | Acronym: MSV
This is a revised version of DPV 133
Maize streak virus
D.P. Martin Institute of Infectious Diseases and Molecular Medicine, University of Cape Town, Cape Town, South Africa
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Maize streak disease was first documented in Natal, South Africa by
Fuller (1901)
but its causal agent was only later described as a virus by
Storey (1925).
MSV is a small single stranded DNA virus
(Harrison et al., 1977)
with a twinned quasi-isomeric or "geminate" particle morphology
(Bock et al., 1974).
It is the type member of the genus Mastrevirus in the family Geminiviridae.
Main Diseases
Causes maize streak disease (MSD) of maize (Zea mays), and diseases of varying severity in many other wild and cultivated grasses, including sugarcane and wheat (Fig. 1).
Geographical Distribution
The virus occurs throughout Africa south of the Sahara, on the Indian Ocean islands of Mauritius and Reunion and possibly on the Indian subcontinent.
Host Range and Symptomatology
The virus is restricted to the Gramineae but can infect over 80 grass species (ICTVdb Management, 2006; Damsteegt, 1983; Konate & Traore, 1992). These include species in the tribes Andropogoneae (Cymbopogon, Imperata, Saccharum, Rottboellia), Eragrosteae (Dactyloctenium, Diplachne, Eleusine, Eragrostis, Leptochloa, Setaria), Sporoboleae (Sporobolus), Zoysieae (Tragus), Maydeae (Zea, Euchlaena), Hordeae (Triticum) and Avenae (Avena).
In MSV-susceptible Zea mays (maize) genotypes, first symptoms appear in young seedlings 3-7 days after inoculation as almost circular pale spots, 0.5-2 mm in diameter, in the lowest exposed portions of the youngest leaves (Fig. 1). When the disease is fully developed the plants show narrow veinal streaks which range from a few mm to several cm long, from 0.5-1 mm wide and frequently fuse laterally.
Several grass-adapted strains (MSV-B, C, D, E and F; Martin et al., 2001) produce milder symptoms in MSV-susceptible maize genotypes such as sweetcorn and popcorn than the maize adapted strain. MSV-resistant maize genotypes are often immune to the grass-adapted strains.
Strains
Six strains have been described (MSV-A through F; Fig. 2), only one of which, MSV-A, produces severe symptoms in maize (Martin et al., 2001; Schnippenkoetter et al., 2001; Willment et al., 2002). The whole genome nucleotide sequence divergence of MSV-A isolates obtained from opposite ends of the continent is very low (< 3% divergence; Martin et al., 2001) which may indicate either that the rate at which MSV-A is evolving is fairly slow, or that continent-wide spread and dominance of new MSV genotypes with enhanced fitness is very rapid. Experimental approximations of the rates at which a MSV-A isolate (MSV-Reu) evolves when maintained in a susceptible maize genotype, a resistant maize genotype and a non-maize host (Coix lacryma-jobi) are respectively, 9.5 x 10-5, 17.3 x 10-5, and 26.5 x 10-5 substitutions per site per year (Isnard et al., 1998). These evolutionary rates are relatively low when compared with RNA viruses and imply that genome-wide only one nucleotide becomes fixed every ~1.5 to ~4 years (depending on the selection pressures exerted by host species). Despite these low nucleotide fixation rates, it has been demonstrated that the mutation rate of MSV is unexpectedly high (Shepherd et al., 2005) indicating that the emergence of resistance breaking MSV variants is a possibility should MSV resistant maize genotypes become widespread in Africa.
Transmission by Vectors
At least eight species of cicadellid leafhoppers in the genus Cicadulina (including C. mbila, C. storeyi, C. bipunctella zeae, C. latens and C. parazeae) transmit MSV and related African streak mastreviruses, in a persistent manner (Dabrowski, 1987; Fennah, 1959; Nielson, 1986; Ruppel, 1965; Soto, 1978). Storey (1925; 1928; 1932; 1933; 1938; 1939) studied vector transmission in great detail. The virus may be acquired in less than 1 h (minimum acquisition time 15 sec) and may be inoculated in 5 min; latent period in the vector is 6-12 h at 30°C, which coincides with the first appearance of the virus in the body fluids. Following acquisition of the virus by a leafhopper, it becomes viruliferous within 30 h (Okoth et al., 1987) and is able to transmit for the rest of its life (Reynaud & Peterschmitt, 1992).
Whereas there is some evidence that geminiviruses in the genus Begomovirus (e.g. Tomato yellow leaf curl virus or TYLCV), may be able to replicate within their whitefly vector (Rubinstein & Czosnek, 1997), it is unlikely that MSV replication occurs within leafhoppers since virus titres within an insect decrease over the lifetime of the insect (Reynaud & Peterschmitt, 1992). Another difference between TYLCV in whiteflies and MSV in leafhoppers is that MSV is not transmitted transovarially (Storey, 1928; Van Rensburg & Giliomee, 1990) whereas TYLCV is (Ghanim et al., 1998).
All five nymphal instars are able to acquire and transmit the virus; it is retained during moulting but is not transmitted through the egg. Races that are active (able to transmit) and inactive have been found for four of the Cicadulina vector species (mbila, storeyi, bipunctella zeae and latens); the gut wall of inactive races is impermeable to passage of the virus but inactive races are rendered active by injecting virus into the abdomen or by puncturing the gut wall. The ability to transmit is inherited as a simple dominant gene linked with sex. Employing conventional notation (A = major gene for activity, a for inactivity), the constitutions are active males (AX) (Y); inactive males, (aX) (Y); active females, (AX) (AX) or (AX) (aX); and inactive females, (aX) (aX). Individual insects of an active race are not equally efficient; there is evidence that a second autosomal gene modifies the effect of the major activity gene.
Transmission through Seed
Not seed-transmitted in maize.
Transmission by Dodder
Not reported.
Serology
Maize streak virus is moderately to strongly immunogenic; Bock et al. (1974) obtained an antiserum with titres of 1/256 and 1/1024 in gel-diffusion and tube precipitin tests respectively.
Relationships
MSV is closely related to the other African streak mastreviruses, Panicum streak virus, Sugarcane streak virus, Sugarcane streak Egypt virus and Sugarcane streak Reunion virus with which it shares approximately 65% genome sequence identity. It is, however, most similar to a Digitaria streak virus isolate from the Pacific island of Vanuatu with which it shares ~67% genome sequence identity (Fig. 2) but this is not necessarily of overriding phylogenetic significance.
Stability in Sap
Studied by allowing vectors to feed through membranes on crude sap containing the type (maize) strain and transferring them to maize seedlings. Thermal inactivation point is not less than 60°C; dilution end-point not less than 1/1000; longevity at room temperature not less than 24 h (K. R. Bock & E. J. Guthrie, unpublished data).
Purification
The different virus strains may be purified satisfactorily from their preferred hosts (Bock et al., 1974). Homogenize 100 g infected leaf tissue in 200 ml 0.01 M phosphate buffer containing 0.1 % thioglycollic acid, pH 3.9-4.1. Clarify with n-butanol (7 ml/100 ml extract), concentrate by ultracentrifugation, and resuspend the virus in 0.01 M phosphate, pH 7.7.
Properties of Particles
Sedimentation coefficent, s°20,w: 76 S for paired and 54 S for single particles (Bock et al., 1974).
Particle Structure
The particles consist of 2 incomplete icosohedra with T=1 surface lattice, comprising 22 pentameric capsomers each containing 5 coat protein molecules (Zhang et al., 2001). Particle dimensions are 38 x 22 nm. An atomic model of the CP has been inferred using the CP Cryo-EM structure (Zhang et al., 2001).
Particle Composition
Particles are comprised of 110 coat protein molecules packaged around a ~2690 nucleotide, covalently closed, mostly single-stranded, DNA genome (Zhang et al., 2001). The packaged DNA is double stranded over an ~80 nucleotide sequence that is believed to act as a primer for complementary strand synthesis following infection and uncoating.
Properties of Infective Nucleic Acid
The packaged MSV genome is a circular covalently closed single stranded DNA molecule containing ~2690 nucleotides.
Genome Properties
The genome encodes four proteins: a movement protein (MP), a coat protein (CP), a replication associated protein (Rep), and a regulatory protein (RepA) that is expressed from the same transcript as Rep. MSV genomes also contain two intergenic regions; a short or small one (SIR) and a long or large one (LIR; Fig. 3).
The long intergenic region
Besides containing divergent RNA polymerase II-type promoters and other transcriptional regulatory features
that are necessary for the expression of the complementary and virion sense genes, the LIR also contains
sequence elements that are essential for MSV replication. The most striking of these is an inverted repeat
sequence that is capable of forming a stable hairpin loop structure in duplex DNA
(Fig. 4;
Sunter et al., 1985).
All geminiviruses sequenced to date have the highly conserved nonanucleotide sequence, TAATATT|AC, within
similar hairpin structures: This sequence contains the virion sense strand origin of replication
(|; Heyraud et al., 1993;
Heyraud-Nitschke et al., 1995;
Laufs et al., 1995a;
Stanley, 1995;
Stenger et al., 1991).
A sequence occurring between the TATA box, believed to direct Rep/RepA transcription (Boulton et al., 1991), and the RepA initiation codon is directly repeated in the stem of the V-ori hairpin (Fig. 4) and has been found to occur in all mastreviruses sequenced to date. The repeated sequence ranges in size from 6 to 12 nucleotides in different viruses. By analogy with similar iterated sequences identified as Rep binding sites in other geminiviruses, these sequences are also believed to be involved in Rep and/or RepA binding (Arguello-Astorga et al., 1994a; Choi & Stenger, 1996; Fontes et al., 1994a, 1994b; Castellano et al., 1999; Missich et al., 2000; Sanz-Burgos & Gutierrez, 1998). The MSV Rep and/or RepA may also bind at the stem loop and the virion sense promoter TATA box (Fig. 4; Castellano et al., 1999; Missich et al., 2000)
The virion sense promoter required for optimal CP expression has been localised to a region 530 nucleotides upstream of the CP start codon (Fenoll et al., 1988, 1990). This region, called the upstream activator sequence (UAS), includes the stem loop and two directly repeated GC boxes at the base of the stem proximal to the RepA start codon. One or two GC boxes at this position is a conserved feature of all the geminivirus genomes so far sequenced. In MSV, the region containing the GC boxes binds maize nuclear factors to the UAS and has been named the rightward promoter element (rpe1; Fig. 4; Fenoll et al., 1990). Besides functioning in transcription of virion sense genes, it is also possible that rpe1 may recruit nuclear factors to this region for purposes of replication (Arguello-Astorga et al., 1994b).
The short intergenic region
The MSV SIR exists between the termination codons of the CP and Rep genes of all mastreviruses and is the
origin of complementary strand synthesis. It also contains the polyadenylation and termination signals of
the virion and complementary sense transcripts
(Fig. 5)
but it has not been very thoroughly investigated.
A small complementary sense primer-like molecule ~80 nucleotides in size and of unknown origin is bound to the SIR of enapsidated MSV DNA (Donson et al., 1987; Donson et al., 1984). The 52 3'-nucleotides of the primer binding site and 8 additional nucleotides past its 3'-end are, together with most of the LIR, the only sequences always present in naturally occurring MSV subgenomic DNAs (Fig 5; Billharz, Martin, & Rybicki unpublished). At the onset of an infection, the primer molecule probably enables synthesis of dsDNA replicative forms from newly uncoated virion strand DNA. While it is possible that a similar primer is produced during active geminivirus replication (Gutierrez, 1999), its occurrence in virions may simply be a consequence of encapsidation prematurely arresting complementary strand synthesis (Donson et al., 1984). The exact sequence requirements for the initiation of (-) strand synthesis are almost certainly very lax (Liu et al., 1999b).
The complementary sense genes (Rep and RepA)
The replication initiator/associated protein (Rep) is the only MSVgene product that is absolutely required
for its replication
(Elmer et al., 1988;
Hanley-Bowdoin et al., 1990;
Hayes & Buck, 1989;
Schalk et al., 1989).
In mastreviruses, RepA (the C1 ORF) and the C2 ORF (also called RepB), respectively encode
the amino- and carboxy-terminal parts of Rep
(Fig. 6;
Mullineaux et al., 1990;
Schalk et al., 1989).
Beginning at the same transcription initiation site, two C-sense transcripts (1.5Kb and 1.2Kb;
Fig. 6)
are produced during MSV infections
(Dekker et al., 1991;
Mullineaux et al., 1990).
Splicing of the larger transcript removes an intron which permits expression of full length Rep from the
two ORFs
(Wright et al., 1997).
It is very probable, although as yet unproven in vivo, that RepA, a variant of Rep is translated from both the unspliced 1.5Kb and the 1.2Kb C-sense transcripts (Dekker et al., 1991; Mullineaux et al., 1990; Gutierrez, 1999). If expressed in infected cells, MSV RepA would have the same N-terminal 214 amino acid sequence as Rep, but would have a different C-terminus (Fig. 6). There is evidence that RepA is a multifunctional protein with unique features (Table 1) that are required at different stages during the MSV replicative cycles of mastreviruses (Fig. 6).
Table 1 The characteristics of Rep and RepA
| Activity | Rep | RepA | Reference |
| Rb binding | - | + | Horvath et al., 1998 |
| GRAB binding | - | + | Xie et al., 1999 |
| Down regulation of replication | - | + | Collin et al., 1996 |
| Required for replication | + | - | Schalk et al., 1989 |
| Sequence specific DNA binding | + | + | Castellano et al., 1999; Missich et al., 2000 |
| Oligomerisation | + | + | Horvath et al., 1998; Sanz-Burgos & Gutierrez, 1998 |
| ATPase activity | +a | - | Hanson et al., 1995; Desbiez et al., 1995 |
| Transactivation activity in yeast | +b | + | Horvath et al., 1998 |
| Transactivation of V-sense genes | + | + | Collin et al., 1996; Hofer et al., 1992; Zhan et al., 1993) |
bTransactivation activity required truncation of the protein
The N-terminal portions of Rep and RepA that are common to both proteins contain a number of conserved amino acid sequence motifs with either known, unknown or suspected activities (Fig. 6). Three of these, named rolling circle replication motifs I, II and III, are shared with replication-associated proteins from a diverse group of ssDNA viruses and plasmids that replicate via a rolling circle mechanism (Koonin & Ilyana, 1993). Whereas motif I has an unknown function, motif II is possibly involved in binding Mn2+ or Mg2+ (Koonin & Ilyana, 1992; Laufs et al., 1995a) and motif III participates in DNA cleavage and linkage at the virion sense origin of replication (Laufs et al., 1995b). A motif involved in retinoblastoma protein (Rb) binding is also found in both the Rep and RepA proteins of MSV, although the motif only has Rb binding activity in RepA.
Rep and RepA proteins perform certain of their functions in an oligomerised form (Sanz-Burgos & Gutierrez, 1998; Missich et al., 2000) and a 13 amino acid region between the Rb binding domain and motif III has been implicated in Rep-Rep, RepA-RepA and RepA-Rep interactions (Fig. 6; Horvath et al., 1998).
Sequence-specific DNA recognition and binding domains have been mapped to the N-terminal 116 and 89 amino acids of other geminivirus Rep proteins (Choi & Stenger, 1996, Jupin et al., 1995) and are also likely to reside in this region of MSV Rep and RepA proteins (Gutierrez, 1999).
Conserved amongst geminivirus Rep proteins (but not found in RepA) is a dNTP binding domain (also referred to as motif IV) with A and B motifs similar to those found in proteins with kinase and helicase activities (Fig. 6; Gorbalenya & Koonin, 1989). In other geminiviruses it has been demonstrated that the motif has an associated ATPase activity that is apparently required for efficient replication (Desbiez et al., 1995; Hanson et al., 1995). Although the exact purpose of the ATPase activity is uncertain, it is possible that the motif may have an associated helicase activity that is operational during replication (Breret et al., 1999; Desbiez et al., 1995)
Overlapping the dNTP binding motif of MSV and other geminivirus Reps is a region with similarity to the DNA binding domains of the myb-related class of plant transcription factors (Hofer et al., 1992; Horvath et al., 1998) which may be functional in the induction of virion sense gene transcription (Hofer et al., 1992; Zhan et al., 1993) and/or host genes during an infection (Palmer & Rybicki, 1998). However, full length Rep is unable to activate transcription in yeast reporter gene constructs, implying that the transactivation domain may be masked during protein folding. Such a masking effect has been detected for the ATF-2 transcription factor (Li & Green, 1996) and may regulate the transactivation activity of Rep by ensuring that the domain is only exposed following binding of Rep to appropriate host proteins (Horvath et al., 1998).
Associated with the unique C-terminus of MSV RepA is another transactivation domain with demonstrable activity in yeast (Fig. 6; Horvath et al., 1998). In other geminiviruses there is some evidence to suggest that RepA but not Rep may be responsible for transactivation of V-sense gene transcription (Collin et al., 1996).
Also unique to the C-terminus of mastrevirus RepA proteins is a region that, in other geminiviruses has been found to interact with a group of host GRAB (geminivirus RepA binding) proteins (Xie et al., 1999). These GRAB proteins resemble members of a family of plant proteins containing a NAC (non-apical-meristem, ATAF and CUC2 genes) domain. It appears that NAC proteins are involved in diverse plant developmental pathways and it is possible that RepA-GRAB binding may influence these pathways for the benefit of viral processes (Gutierrez, 2000).
The virion sense genes (MP and CP)
Movement protein (MP) and coat protein (CP) genes are absolutely required for systemic infection of host
plants by MSV
(Boulton et al., 1989;
Lazarowitz et al., 1989;
Woolston et al., 1989).
Transcription of the MSV virion sense genes is probably directed by two TATA boxes 26 and 214
nucleotides upstream from the MP start codon
(Wright et al., 1997).
Two co-terminal bicistronic transcripts are produced – a less abundant transcript initiated ~145
upstream from the MP start within the stem sequence of the V-ori hairpin structure and a more
abundant transcript initiated only 1 nucleotide upstream from the MP start codon
(Fig. 7;
Wright et al., 1997).
While it is probable that MP and CP can be expressed from both virion sense (V-sense) transcripts
(Wright et al., 1997),
CP is generally present in infected cells at much higher concentrations than MP
(Mullineaux et al., 1988).
In MSV and certain other mastreviruses, splicing of an intron within the MP portion of V-sense
transcripts appears to be an important determinant of relative MP and CP expression levels
(Fig. 7;
Wright et al., 1997).
Whereas CP is expressed from all spliced and unspliced V-sense transcripts, MP is only expressed from the
unspliced transcripts.
It is likely that post-translational modification of the MSV MP occurs since its apparent size of 14kDa is larger than that predicted from its sequence (10.9kDa; Dickinson et al., 1996). Mastrevirus MPs have a hydrophobic domain (Fig. 7) that may either facilitate their interaction with host cell membranes or be involved in homo- or hetero-oligomerisation with the CP (Boulton et al., 1993; Kotlizky et al., 2000).
The MSV CP binds both ds and ssDNA in a sequence non-specific manner (Liu et al., 1997). Whereas the binding domain lies within the first 104 CP amino acids, the binding site probably falls between amino acids 5 – 22 (Fig. 7; Liu et al., 1997). In accordance with the identification of potential nuclear localisation signals within the N-terminal regions of all mastrevirus CPs (Fig. 7; Liu et al., 1997), it has been demonstrated that MSV CP is imported into the nuclei of infected maize cells (Kotlizky et al., 2000; Liu et al., 1999a).
Relations with Cells and Tissues
Very little information is available on the stages of MSV infection between leafhopper delivery of virus particles into permissive host plants and the onset of rolling circle replication in the nuclei of permissive cell types. Leafhoppers feed from both the mesophyll and phloem of healthy host plants (Mesfin et al., 1995). Virus particles delivered into the phloem of leaves are apparently transported at > 200 mm h-1 within sieve tubes to regions removed from the leafhopper feeding site (Peterschmitt et al., 1992; Storey, 1938). The ultimate destination of these particles remains to be determined but a sufficient number move out of an inoculated leaf within a few hours to initiate a productive infection (Storey, 1938).
A MSV infection is presumably initiated when at least one virus particle enters a nucleated plant cell such as a phloem companion or mesophyll cell. It is unknown whether a productive infection can be initiated by entry of a particle into any nucleated cell or whether it requires that a particle enters a cell undergoing active division (Hanley-Bowdoin et al., 1999). It is also unknown whether infection of these first cells is by individual particles (completely monoclonal) or by groups of particles (potentially polyclonal).
Once an individual virus particle is within a permissive cell it presumably uncoats either in the nucleus or in the cytoplasm. In the latter case, capsomeres or CP monomers derived from virion disassembly would probably bind to and transport the released DNA into the nucleus (Liu et al., 1999a). DNA released from virions is single-stranded except for an approximately 80 nucleotide region in the SIR which is believed to be vital for priming the initiation of complementary strand DNA synthesis (Donson et al., 1984; Hayes et al., 1988). The short complementary nucleic acid molecule bound to the otherwise ssDNA is primer-like in that it is predominantly DNA but also contains a few ribonucleotides on its 5' end (Donson et al., 1984). While similar primer molecules have also been found in a number of other mastrevirus species (Andersen et al., 1988; Donson et al., 1987; Hayes et al., 1988; Morris et al., 1992), the mechanism of their synthesis remains to be determined (Gutierrez, 1999).
MSV replicates by a rolling circle mechanism within the nuclei of infected cells. It is also possible that replication occurs by a recombination dependent mechanism but this has not been conclusively demonstrated for MSV. The molecular biology of MSV replication is summarised in Fig. 8 and is reviewed in Gutierrez (1999).
Besides being the primary location of replication, the nucleus is also the site of virus particle assembly. It is probable that later in the replication process, when virion sense gene expression is underway, increasing nuclear concentrations of CP result in the accumulation of ssDNA (Boulton et al., 1989; Lazarowitz et al., 1989). CP molecules in the nucleus possibly bind (+) strands released during RCR and arrest the synthesis of new RF DNAs (Donson et al., 1984). There is apparently no encapsidation signal within geminivirus genomes and it is possible that any circular ssDNA of an appropriate size will be encapsidated (Mansoor et al., 1999; Saunders & Stanley, 1999). MSV virion strands, each with an attached ~80 nucleotide primer molecule (Donson et al., 1984), are packaged into particles that congregate to form large paracrystalline nuclear inclusions (Pinner et al., 1993). Crystalline arrays of MSV particles have also been detected outside nuclei within physiologically active phloem companion cells, and inside the vacuoles of dead and dying cells within chlorotic lesions (Pinner et al., 1993).
At some point in the infection process after the onset of replication and probably before the completion of encapsidation, viral DNA must be moved into neighbouring cells. The mechanistic details of this process are still obscure but in MSV it appears to involve an interaction between the CP, MP and viral DNA (Boulton et al., 1989; Kotlizky et al., 2000; Lazarowitz et al., 1989).
Besides requiring the co-ordinated interactions of viral gene products and DNA, the successful movement of MSV genomes from infected to uninfected cells is strongly dependent on the extent of plasmodesmatal connections between neighbouring cells (Lucy et al., 1996). Also, in maize it appears as though certain cell types are more sensitive to MSV infection than others (Lucy et al., 1996; Pinner et al., 1993). For example, in maize leaves the virus infects all photosynthetic cell types (e.g. mesophyll and bundle sheath cells) but despite abundant plasmodesmatal connections between photosynthetic, epidermal and parenchyma cells, MSV is only rarely detectable in the latter two cell types (Lucy et al., 1996).
It is unknown whether systemic movement of geminiviruses within plants simply relies on normal cell to cell movement to deliver genomic DNA into the phloem, or whether viral DNA is specifically packaged for long distance transport. It is possible that cell to cell movement might involve unencapsidated ss or dsDNA but that long distance movement in the phloem might require encapsidation (Palmer & Rybicki, 1998). However, leafhopper feeding experiments have determined that no virus acquisition occurs unless leafhoppers feed directly on symptomatic tissues (Peterschmitt et al., 1992; Storey, 1928). This is interesting because MSV CP is detectable at low levels in the asymptomatic leaf tissues of infected plants (Peterschmitt et al., 1992) indicating that movement of MSV throughout the plant is possibly in a form that is not transmissible. While it may be that leafhopper transmission requires that MSV particles form crystalline complexes to survive the ingestion process, it is also possible that the CP detected in asymptotic tissues is in the form of unencapsidated CP:DNA complexes in the process of movement.
In MSV-infected maize, the correlation between patterns of virus accumulation and translocation pathways between tissues is strong evidence that long distance movement occurs via phloem elements (Lucy et al., 1996). It is believed that MSV is incapable of invading the root apical, shoot apical and reproductive meristems due to the absence of developed vasculature in these tissues (Lucy et al., 1996). It is also noteworthy that in all maize organs except the leaves, MSV infection is almost completely restricted to the vasculature (Lucy et al., 1996; Pinner et al., 1993). There is a degree of correlation between MSV replication and the expression of the mitosis-specific histone gene, H2b, in the developing vasculature and it is therefore possible that infection of host cells in all organs except the leaves requires active cell division (Lucy et al., 1996).
Within the shoot apex, MSV first enters developing leaves at approximately plastochron five. While the virus is restricted to the developing leaf vasculature before plastochron 12, it is likely that the development of metaphloem elements at approximately plastochron 12 provides an opportunity for MSV to escape the vasculature into the photosynthetic cells of the leaf. Metaphloem develops with the abundant plasmodesmatal connections required for efficient loading of photoassimilates once the leaf emerges from the whorl. Before emergence, however, the developing photosynthetic tissues are still net importers of photoassimilates and the virus most likely moves into these cells through their plasmodesmatal connections with the metaphloem (Lucy et al., 1996).
On the leaves, the pattern of chlorotic streak-like lesions that characterises MSV infections is directly correlated with the pattern of virus accumulation within the leaves (Lucy et al., 1996) and the virus can only be acquired by leafhoppers from these lesions (Peterschmitt et al., 1992; Storey, 1928). The degree of chlorosis that occurs within lesions is MSV strain-dependent and is related to different degrees of infection-induced chloroplast malformation (Pinner et al., 1993). It is interesting that while leafhoppers generally feed from the phloem sieve tubes of hosts such as Digitaria sanguinalis, they mainly feed on the mesophyll cells of maize plants (Mesfin et al., 1995). In an infected maize leaf, virus particles are undetectable in phloem sieve tubes whereas they occur at high concentrations within mesophyll cells (Lucy et al., 1996) and it is possible that colonisation by MSV of photosynthetic cells in host species such as maize is an evolutionary adaptation to leafhopper feeding behaviour.
Ecology and Control
As is the case with most virus diseases, maize streak disease (MSD) is erratic: It can be devastating in some years and insignificant in others (Efron et al., 1989). Despite its name, MSD can not be fully understood in terms of the isolated relationship between maize plants, a single maize-adapted MSV strain, and an insect species that transmits viruses between maize plants. It is instead the product of extremely complex interactions between at least 8 species of cicadellid leafhoppers in the Genus Cicadulina (Dabrowski, 1987; Fennah, 1959; Nielson, 1986; Ruppel, 1965; Soto, 1978), over 80 grass species (ICTVdb Management, 2006; Damsteegt, 1983; Konate & Traore, 1992), and an as yet unknown number of virus strains (Konate & Traore, 1994; Mesfin et al., 1992; Martin et al., 2001). Compounding this complexity are environmental factors that influence the species composition of grass and leafhopper populations (Dabrowski, 1987).
It is not surprising therefore that accurate predictive modelling of MSD epidemics has not yet been achieved. However, loose correlations between disease incidence and both environmental conditions and agricultural practices have been noted. Because there is a strong correlation between rainfall and leafhopper population densities (Asanzi et al., 1994; Okoth & Dabrowski, 1987; Rose, 1972), drought conditions followed by irregular rains at the beginning of growing seasons tend to be associated with severe MSD outbreaks (Efron et al., 1989). It has also been noted that in regions with two maize growing seasons a year, the incidence of MSD is higher during the second season (Dabrowski et al., 1991; Efron et al., 1989; Fajemisin & Shoyinka, 1976).
The primary focus of future epidemiological modelling of MSD will probably be the dynamics of MSV transmission by leafhopper populations. Like most other infectious plant diseases with an insect vector, serious MSD outbreaks are absolutely governed by leafhopper acquisition and movement of severe MSV isolates from infected plants (wild grasses or crop plants) to sensitive, uninfected crop plants. Of the 8 Cicadulina species that are capable of transmitting MSV, Cicadulina mbila is considered the most important. It is the most widely distributed species and a larger proportion of C. mbila populations have the ability to transmit MSV, compared with other Cicadulina species (Storey, 1928, 1933; Markham et al., 1984). The relative abundance of the different Cicadulina species in different parts of Africa is influenced by altitude, temperature and rainfall (Dabrowski et al., 1987; Okoth & Dabrowski, 1987)
The virus is acquired by leafhoppers during the course of their feeding on infected host plants. C. mbila can feed on more than 138 grass species, ~70% of which are also known to be potential MSV hosts (ICTVdb Management, 2006; Damsteegt, 1983; Konate & Traore, 1992). When feeding on an infected plant, a leafhopper can only acquire the virus by feeding directly on chlorotic lesions (Peterschmitt et al., 1992; Storey, 1928). It is believed that MSV infected plants displaying substantial chlorosis may actually attract leafhoppers because of their yellow colour and that leafhoppers may preferentially feed on infected cells within chlorotic lesions because they are a richer supply of nutrients (P. Markham, personal communication).
Uptake of virus particles by a leafhopper during feeding on a MSV infected cell does not automatically enable transmission of MSV by the insect. The ability of leafhoppers to transmit MSV is an inherited, dominant sex-linked characteristic (Storey, 1932) and the different Cicadulina species vary enormously in both the proportion of individuals capable of transmitting the virus and in the efficiency with which transmission occurs (Asanzi et al., 1995; Dabrowski, 1987; Markham et al., 1984; Van Rensburg & Giliomee, 1990). Once ingested by a leafhopper, the mechanisms by which MSV particles are first transported into its haemocoel, and then into its salivary glands, are unknown.
The distance that MSV spreads from a source of inoculum is determined by the movement behaviour of leafhoppers. Distinct long and short distance flight morphs have been detected amongst Cicadulina populations in Zimbabwe (Rose, 1972). It is believed that the long flight morphs are a migratory form (Rose, 1972) and as such they may play an important part in the rapid long distance spread of virulent MSV variants (Rose, 1978; Dabrowski et al., 1987). Migratory movement is more common in certain Cicadulina species than in others (Dabrowski et al., 1991) and it is probably influenced by environmental conditions (Downham & Cooter, 1998; Rose, 1972).
The dynamics of primary infection following leafhopper invasion of a susceptible maize crop is influenced by leafhopper population densities, the proportion of viruliferous individuals in populations, and the virus titre within these individuals (Rose, 1978; Van Rensburg & Giliomee, 1990). Disease spread is apparently linear when only a few viruliferous leafhoppers are involved in transmission, but becomes exponential once the number of insects exceeds one individual per three plants (Rose, 1978).
Although attempts to understand the dynamics of MSD epidemics have focused primarily on vector population dynamics and behaviour, an important component of any realistic MSD epidemiological model will be the influence of the grass species that are hosts of both MSV and vector. Because Cicadulina species have favoured hosts for mating and oviposition, the species composition of grass populations in any particular area will directly influence leafhopper population densities in that area (Van Rensburg, 1982). Also, many preferred leafhopper hosts are annual grasses, and seasonal alterations in the species composition of grass populations strongly affect leafhopper population densities and feeding behaviours (Okoth & Dabrowski, 1987).
The species composition and age distribution of grasses (including cultivated crops) in an area may also strongly influence the amount of MSV inoculum available for transfer in that area (Konate & Traore, 1992; Rodier et al., 1995). While MSV infects at least 80 of the 138 grass species that leafhoppers feed on (ICTVdb Management, 2006; Damsteegt, 1983; Konate & Traore, 1992), both the susceptibility of these grasses to MSV infection and the severity of symptoms that occur following their infection, may be strongly influenced by a number of factors. While sensitivity to infection can vary substantially from species to species (Damsteegt, 1983), it can also vary within a species with genotype and plant age at the time of inoculation: For example, plants from many species, including maize, generally become more resistant to MSV infection with age (Damsteegt, 1983; Peterschmitt et al., 1992).
Once a viruliferous leafhopper has successfully transmitted virus to a susceptible plant at a suitable growth stage, the time taken for symptoms to develop is correlated with the virus strain transmitted, the virus titre within the insect (Rose, 1978) and the sex of the insect (females are apparently better transmitters than males; Van Rensburg & Giliomee, 1990). Between 3 and 7 days after leafhopper inoculation of plants with MSV, roughly circular spots (0.5 – 2 mm in diameter), become visible on the lowest exposed portions of the youngest leaves (Bock et al., 1974). Later symptoms range from narrow veinal streaks a fraction of a millimeter in width to complete leaf chlorosis, stunting and sterility, depending on the MSV strain involved, the host plant species and genotype, and the age of the plant at the time of infection.
Whereas effective control of MSD in cultivated crops is possible with the use of carbamate insecticides (Drinkwater et al., 1979; Van Rensburg, 1988; Van Rensburg & Walters, 1978; Van Rensburg & Giliomee, 1991), it is widely believed that the development and use of MSV resistant crop genotypes is the best way to minimise the impact of MSD on African agriculture (Efron et al., 1989; Rodier et al., 1995; Tang & Bjarnason, 1993). Great successes have been achieved in the development of MSV resistant maize genotypes that tolerate infection without significant yield loss (Barrow, 1992; Efron et al., 1989; Rodier et al., 1995; Van Rensburg et al., 1991). Besides providing relatively inexpensive security against crop losses, MSV-infected tolerant maize genotypes contain 10 to 90 times fewer virus particles than infected sensitive genotypes (Peterschmitt et al., 1992) and are therefore much poorer sources of inoculum during secondary disease spread (Rodier et al., 1995).
While efforts are underway to promote the widespread cultivation of MSV resistant maize in Africa, surprisingly little is known about the MSV populations that will confront these new genotypes. Attempts to determine the depth of MSV's genetic diversity have just begun and to date only six major MSV strain groupings have been discovered (Martin et al., 2001; Oworb, Martin, Shepherd & Varsani, unpublished). The other African streak virus species, of which only PanSV and a few sugarcane streak virus species have yet been described (Bigarre et al., 1999; Briddon et al., 1992; Briddon et al., 1996; Hughes et al., 1992), have received even less attention than MSV.
It is unknown whether any MSV strains other than MSV-A play an important role in the epidemiology of MSD. Both MSV-B and -C isolates have been found to produce mild symptoms only in maize genotypes that are very MSV susceptible and are therefore unlikely to pose any significant direct threat to maize production (Schnippenkoetter et al., 2001; Willment et al., 2002). It has been directly demonstrated that mixed MSV-A and -B infections occur in nature (Willment et al., 2001) and there is also strong evidence of recombination between these strains (Martin et al., 2001). It is therefore possible that MSV-B, and other MSV strains may indirectly influence MSD epidemiology through recombination with MSV-A type viruses. Recombination has been linked with the emergence of a number of geminivirus diseases (Umaharan et al., 1998; Zhou et al., 1997; Zhou et al., 1998) and it is quite conceivable that it may also eventually contribute to the evolution of MSV genotypes with elevated virulence.
Figures

Maize streak virus symptoms in different host species. A, MSV-A variant in Maize. MSV-B variants in: B, Wheat; C, Setaria sp.; D, Paspalum sp.; E, Digitaria sp.; F, Rye.
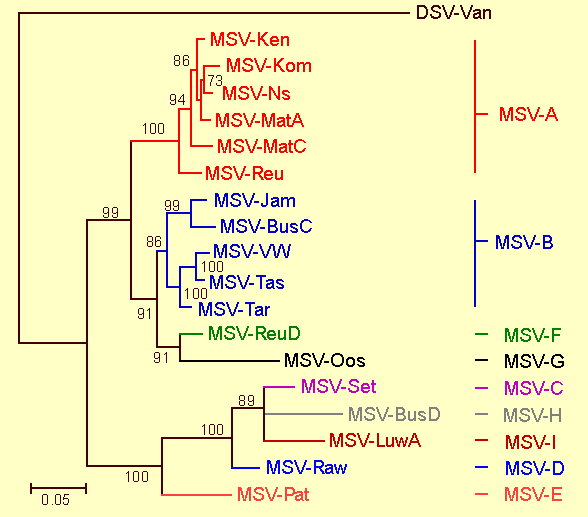
Phylogenetic relationships between the full genome sequences of different MSV strains. The tree is constructed using the maximum likelihood method (HKY model; transition:transversion weight determined from the data; 100 bootstrap replicates) and numbers associated with branches indicate the degree of bootstrap support for them. Branches with less than 70% support have been collapsed and the genome sequence of a Digitaria streak virus (DSV) from Vanuatu is included as an outgroup. Only viruses in the MSV-A group have been isolated from maize; all viruses in the other groups have been isolated from either wheat or wild grass species.

Genomic organisation of Maize streak virus. The origin of virion strand synthesis is indicated by the stem loop symbol at 12 o'clock. LIR, long/large intergenic region; SIR, short/small intergenic region; Rep, replication associated/initiator protein gene. In mastreviruses, Rep is expressed from the post-translationally spliced transcript of ORFs C1 and C2; RepA, variant of Rep potentially expressed from an unspliced complimentary sense transcript and encoding a multifunctional regulatory protein that is unique to the mastreviruses; MP, movement protein gene; CP, coat protein gene.

A schematic representation of the MSV-Ns long intergenic region (LIR). (A) The main landmarks within the LIR. These include a stem-loop structure and highly conserved nonanucleotide at the origin of virion strand replication, iterated sequences (iterons) potentially involved with Rep recognition or binding to the LIR, TATA and GC-boxes involved in gene transcription, and a region of static DNA curvature. (B) Initiation points of complementary and virion sense transcripts. (C) Possible positions of Rep binding within the LIR inferred by analogy to those detected in another mastrevirus, Wheat dwarf virus (WDV). (D) Minimal sequence requirements for MSV replication initiation and termination. The regions that contain replication specificity determinants most likely contain Rep binding sites analagous to those detected in WDV. Numbering is relative to the virion strand replication initiation site.
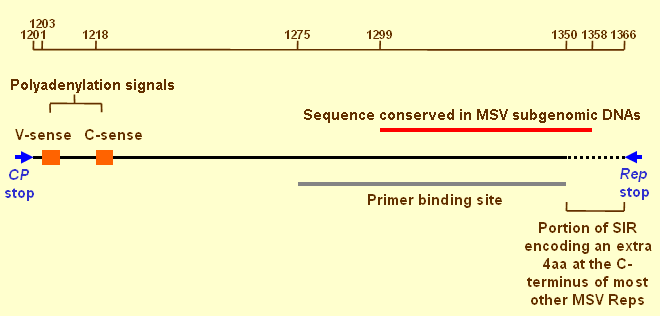
A schematic representation of the short intergenic region (SIR) of MSV-Ns. Orange boxes indicate the positions of polyadenylation signals for both virion and complementary sense transcripts. The primer binding site is the region of the SIR that binds a small complementary DNA molecule encapsidated within virions. The region conserved in MSV subgenomic DNAs is likely to contain the minimal origin of complementary strand replication. Rep terminates at slightly different positions in different MSV isolates. Numbering is relative to the virion strand replication initiation site. References are within the text.

The MSV-Ns RepA (C1) and C2 ORFs, their expression and the functional domains of their products. (A) The C-sense intron (in red) is bounded by donor and acceptor sites (DS and AS respectively) and contains a consensus branch point or lariat sequence (LS). Orange boxes represent potential C-sense transcript polyadenylation signals (PAS). At the 3' end of the C2 ORF are a series of sequences that are conserved between MSV-isolates and are potentially capable of forming stable stem-loop structures (Donson et al., 1984). (B) Small (1) and large (2) C-sense transcripts are produced with 20% of the large transcripts being spliced to produce the mRNAs from which Rep is expressed. RepA is potentially expressed from either long or short unspliced transcripts. (C) The sequence motifs and functional domains that have been detected in mastrevirus Reps (After Gutierrez, 1999). Note that no mastrevirus DNA binding domain has yet been mapped and the region indicated here is inferred by analogy to those mapped for begomovirus and curtovirus Reps (Choi & Stenger, 1995, 1996; Jupin et al., 1995). Also, there is currently no evidence that the GRAB binding activity of WDV's RepA is shared by MSV's RepA. Numbering of DNA is relative to the virion strand replication initiation site.

The MSV-Ns movement protein (MP) and coat protein (CP) genes, their expression and the functional domains of their products. (A) The MP intron is bounded by donor and acceptor sites (DS and AS respectively), and contains both a series of T-tracts and a consensus branch point or lariat sequence (LS). Shown in orange is the V-sense transcript polyadenylation signal (PAS). (B) Large (L) and small (S) V-sense transcripts are produced with the small transcripts being more common. 10% of large and 50% of small transcripts are spliced. CP can be expressed from all transcripts but is most efficiently expressed from the spliced and unspliced small transcripts. MP can be expressed from large or small unspliced transcripts. (C) The known or suspected functional domains of MP and CP. The location of the MP intron corresponds directly with the suspected MP transmembrane domain. Numbering of DNA is relative to the virion strand replication initiation site.

Summary of the MSV infection process. Early during an infection following the synthesis of a double stranded DNA replicative form (RF; 1) RepA is most likely expressed and induces a cellular state in which viral DNA replication can occur (2). The replication associated protein (Rep) is also expressed early and rolling circle replication begins (3). At a later point in the infection process, following genome amplification and possibly Rep and/or RepA induction of the virion sense promoter, movement (MP) and coat protein (CP) expression takes place (4) and movement and encapsidation occur (5). Represented here is movement of unencapsidated ssDNA but it should be noted that it is possible that dsDNA and/or encapsidated ssDNA may also be moved either cell to cell or systemically within the phloem of plants. Whereas the involvement of MSV CP and MP in movement has been demonstrated (Kotlizky et al., 2000; Liu et al., 1997, 1999a), the mechanics of the process are obscure and certain details have been borrowed from a cell to cell movement model proposed for the begomovirus, SqLCV (Lazarowitz & Beachy, 1999; Qin et al., 1998). While the probable timing of events is indicated, it is unlikely, for example, that absolutely no MP and CP expression occurs during the earlier stages of the infection process. Single stranded DNA is represented by blue lines, double stranded DNA by bold black lines and RNA by orange lines.
References list for DPV: Maize streak virus (416)
- Andersen, Richardson, Harbison & Morris, Virology 164: 443, 1988.
- Arguello-Astorga, Guevara-Gonzalez, Herrera-Estrella & Rivera-Bustamante, Virology 203: 90, 1994a.
- Arguello-Astorga, Herrera-Estrella & Rivera-Bustamante, Plant Molecular Biology 26: 553, 1994b.
- Asanzi, Bosque-Perez & Buddenhagen, Plant Pathology 43: 145, 1994.
- Asanzi, Bosque-Perez, Nault, Gordon, & Thottappilly, African Entomology 3: 173, 1995.
- Barrow, Crop Protection 11: 267, 1992.
- Bigarre, Salah, Granier, Frutos, Thouvenel & Peterschmitt Archives of Virology 144: 2331 1999.
- Bock, Guthrie & Woods, Annals of Applied Biology 77: 289, 1974.
- Boulton, Steinkelner, Donson, Markham, King & Davies, Journal of General Virology 70: 2309, 1989.
- Boulton, King, Donson & Davies, Virology 183: 114, 1991.
- Boulton, Pallaghy, Chantani, MacFarlane & Davies, Virology 192: 85, 1993.
- Breret, Clerot & Gronenborn, Proceedings of the XI International Congress of Virology Sydney Australia, 1999.
- Briddon, Lunnes, Chamberlin, Pinner, Brundish & Markham, Journal of General Virology 73: 1041, 1992.
- Briddon, Lunnes, Bedford, Chamberlin, Mesfin & Markham, European Journal of Plant Pathology 102: 379, 1996.
- Castellano, Sanz_Burgos & Gutierrez, Journal of Molecular Biology 290: 639, 1999.
- Choi & Stenger, Virology 206: 904, 1995.
- Choi & Stenger, Virology 226: 122, 1996.
- Collin, Fernandez-Lobato, Gooding, Mullineaux & Fenoll, Virology 219: 324, 1996.
- Dabrowski, Journal of Applied Entomology 103: 489, 1987.
- Dabrowski, Nwilene & Kumar, Insect Science and its Application 12: 249, 1991.
- Damsteegt, Plant Disease 67: 734, 1983.
- Dekker, Woolston, Xue, Cox & Mullineaux, Nucleic Acids Research 19: 4075, 1991.
- Desbiez, David, Metouchi, Laufs & Gronenborn, Proceedings of the National Academy of Sciences USA 92: 5640, 1995.
- Dickinson, Halder & Woolston, Virology 220: 51, 1996.
- Donson, Morris-Krsinich, Mullineaux, Boulton & Davies. EMBO Journal 3: 3069, 1984.
- Donson, Accotto, Boulton, Mullineaux & Davies, Virology 161: 160, 1987.
- Dowham & Cooter, Bulletin of Entomological Research 88: 117, 1998.
- Drinkwater, Walters & Van Rensburg, Phytophylactica 11: 5, 1979.
- Duffus & Gold, Phytopathology 63: 1107, 1973.
- Efron, Kim, Fajemsin, Mareck, Tang, Dabrowski, Rossel, Thottappilay & Buddenhagen, Plant Breeding 103: 1, 1989.
- Elmer, Brand, Sunter, Gardiner, Bisaro & Rogers, Nucleic Acids Research 16: 7043, 1988.
- Fajemisin & Shoyinka, Proceedings of the International Maize Virus Disease Colloquium and Workshop, Wooster Ohio, USA p52, 1976.
- Fennah, Annals and Magazine of Natural History 2: 757, 1959.
- Fenoll, Black & Howell, EMBO Journal 7: 1589, 1988.
- Fenoll, Schwartz, Black, Schneider & Howell, Plant Molecular Biology 15: 865, 1990.
- Fontes, Gladfelter, Schaffer, Pett & Hanley-Bowdoin, Plant Cell 6: 405, 1994a.
- Fontes, Eagle, Sipe, Luckow & Hanley-Bowdoin, Journal of Biological Chemistry 269: 8459, 1994b.
- Fuller, 1st Report of the Government Entomologist, Natal, 1899-1900, 1901.
- Ghanim, Morin, Zeidan & Czosnek, Virology 240: 8022, 1998.
- Gorbalenya & Koonin, Nucleic Acids Research 17: 8413, 1989.
- Gutierrez, Cellular and Molecular Life Sciences 56: 131, 1999.
- Gutierrez, EMBO Journal 19: 792, 2000.
- Hanley-Bowdoin, Elmer & Rogers, Proceedings of the National Academy of Sciences USA 87: 1446, 1990.
- Hanley-Bowdoin, Sellage, Orozco, Nager & Robertson, Critical Reviews in Plant Science 18: 71, 1999.
- Hanson, Hoogstraten, Ahlquist, Gilbertson, Russell & Maxwell, Virology 211: 1, 1995.
- Harrison, Barker, Bock, Guthrie, Merideth & Atkinson, Nature 270: 760, 1977.
- Hayes & Buck, Nucleic Acids Research 17: 10213, 1989.
- Hayes, MacDonald, Coutts & Buck, Journal of General Virology 69: 1345, 1988.
- Heyraud, Matzeit, Kammann, Schaefer, Schell & Gronenborn, Biochemie 75: 605, 1993.
- Heyraud-Nitscke, Schumacher, Laufs, Schaefer, Schell & Gronenborn, Nucleic Acids Research 23: 910, 1995.
- Hofer, Dekker, Reynolds, Woolston, Cox & Mullineaux, Plant Cell 4: 213, 1992.
- Horvath, Pettko-Szandtner, Nikovics, Bilgin, Boulton, Davies, Gutierrez & Dudits, Plant Molecular Biology 38: 699, 1998.
- Hughes, Rybicki & von Wechmar, Journal of General Virology 73: 1031, 1992.
- ICTVdB Management, in ICTVdB - The Universal Virus Database, version 4, 00.029.0.01.001. Maize streak virus, ed. C. Büchen-Osmond, New York: Columbia University, 2006
- Isnard, Garnier, Frutos, Reynaud & Peterschmitt, Journal of General Virology 79: 3091, 1998.
- Jupin, Hericourt, Benz & Gronenborn, Virology 204: 82, 1995.
- Konate & Traore, Phytoprotection 73: 111, 1992.
- Konate & Traore, Phytoprotection 75: 91, 1994.
- Koonin & Ilyana, Journal of General Virology 73: 2763, 1992.
- Koonin & Ilyana, Biosystems 30: 241, 1993.
- Kotlizky, Boulton, Pitaksutheepong, Davies & Epel, Virology 274: 32, 2000.
- Laufs, Traut, Heyraud, Matzeit, Rogers, Schell & Gronenborn, Proceedings of the National Academy of Sciences USA 92: 3879, 1995a.
- Laufs, Schumacher, Geisler, Jupin & Grunenborn, FEBS Letters 377: 258, 1995b.
- Lazarowitz & Beachy Plant Cell 11: 535, 1999.
- Lazarowitz, Pinder, Damsteegt and Rogers, EMBO Journal 8: 1023, 1989.
- Li & Green, Genes and Development 10: 517, 1996.
- Liu, Boulton & Stanley, Journal of General Virology 78: 1265, 1997.
- Liu, Boulton, Thomas, Prior, Oparka & Davies, Molecular Plant-Microbe Interactions 12: 894, 1999a.
- Liu, Pinner, Davies & Stanley, Journal of General Virology 80: 501, 1999b.
- Lucy, Boulton, Davies & Maule, Molecular Plant-Microbe Interactions 9: 22, 1996.
- Mansoor, Khan, Bashir, Saeed, Zafar, Malik, Briddon, Stanley & Markham, Virology 259: 190, 1999.
- Markham, Pinner & Boulton, Maize Virus Disease Newsletter 1: 33, 1984.
- Martin, Willment, Billharz, Velters, Odhiambo, Njuguna, James& Rybicki Virology, 288: 247, 2001.
- Mesfin, Bosque-Perez & Buddenhagen, Plant Disease 76: 789, 1992.
- Mesfin, Den Hollander & Markham, Bulletin of Entomological Research 85: 387, 1995.
- Missich, Ramirez-Parra & Gutierrez, Virology 273: 178, 2000.
- Morris, Richardson, Haley, Zhan & Thomas, Virology 187: 633 1992.
- Mullineaux, Boulton, Bowyer, van der Vlugt, Marks, Donson & Davies, Plant Molecular Biology 11: 57 1988.
- Mullineaux, Guerineau & Accotto, Nucleic Acids Research 18: 7259, 1990.
- Nielson, U.S. Department of Agricultur Technical Bulletin 1382, 1986.
- Okoth & Dabrowski, Acta Oecologica Applicata 8: 191, 1987.
- Okoth, Dabrowski, Thottappilly,Van Emden, Insect Science and its Application 8: 295, 1987.
- Palmer & Rybicki, Advances in Virus Research 50: 183, 1998.
- Peterschmitt, Quiot & Reynaud, Annals of Applied Biology 121: 641, 1992.
- Pinner, Medina & Plaskitt, Plant Pathology 42: 75, 1993.
- Qin, Ward & Lazarowitz, Journal of Virology 72: 9247 1998.
- Reynaud & Peterschmitt, Annals of Applied Biology 121: 85, 1992.
- Rodier, Assio, Marchand & Hervo, Euphytica 81: 57, 1995.
- Rose, Journal of Animal Ecology 41: 495, 1972.
- Rose, Annual Review of Entomology 23: 250, 1978.
- Rubinstein and Czosnek, Journal of General Virology 78: 2683, 1997.
- Ruppel, Publications of the Michigan State University Museum, Biology Series 2: 385, 1965.
- Sanz-Burgos & Gutierrez, Virology 264: 142, 1998.
- Saunders & Stanley, Virology 264: 142, 1999.
- Schalk, Matzeit, Schiller, Schell & Gronenborn, EMBO Journal 8: 359, 1989.
- Schnippenkoetter, Martin, Hughes, Fyvie, Willment, James, von Wegmar & Rybicki, Archives of Virology 146: 1075, 2001.
- Shepherd, Martin, McGivern, Boulton, Thomson & Rybicki, Journal of General Virology 86: 803, 2005.
- Soto, East African Agriculture and Forestry Journal 44: 70, 1978.
- Stanley, Virology 206: 707, 1995.
- Stenger, Revington, Stevenson & Bisaro, Proceedings of the National Academy of Sciences USA 88: 8029, 1991.
- Storey, Annals of Applied Biology 12: 422, 1925.
- Storey, Annals of Applied Biology 15: 1, 1928.
- Storey, Proceedings of the Royal Society Series B 112: 46, 1932.
- Storey, Proceedings of the Royal Society Series B 113: 463, 1933.
- Storey, Proceedings of the Royal Society Series B 125: 455, 1938.
- Storey, Proceedings of the Royal Society Series B 127: 526, 1939.
- Sunter, Buck & Coutts, Nucleic Acids Research 13: 4645, 1985.
- Tang & Bjarnason, Maydica 38: 301, 1993.
- Umaharan, Padidam & Phelps, Phytopathology 88: 1262, 1998.
- Van Rensburg, Proceedings of the 5th South African Maize Breeding Symposium 100, 1982.
- Van Rensburg, Applied Plant Science 2: 63, 1988.
- Van Rensburg & Giliomee, Phytophylactica 22: 241, 1990.
- Van Rensburg & Giliomee, Phytophylactica 23: 141, 1991.
- Van Rensburg & Walters, Phytophlactica 10: 49, 1978.
- Van Rensburg, Gilomee & Pringle, South African Journal of Plant and Soil 8: 38 1991.
- Willment, Martin & Rybicki, Journal of Virological Methods 93: 75, 2001.
- Willment, Martin, Van der Walt & Rybicki, Phytopathology, 92: 81, 2002.
- Woolston, Reynolds, Stacey & Mullineaux, Nucleic Acids Research 17: 6029, 1989.
- Wright, Heckel, Groenendijk, Davies & Boulton, Plant Journal 12: 1285, 1997.
- Xie, Sanz-Burgos, Guo, Garcia & Gutierrez, Plant Molecular Biology 39: 647, 1999.
- Zhan, Richardson, Haley & Morris, Virology 193: 498, 1993.
- Zhang, Olson, Baker, Faulkner, Aqbandje-McKenna, Boulton, Davies & McKenna, Virology, 279: 471, 2001.
- Zhou, Liu, Calvert, Munoz, Otim-Nape, Robinson & Harrison, Journal of General Virology 78: 2101, 1997.
- Zhou, Liu, Robinson & Harrison, Journal of General Virology 79: 915, 1998.