Details of DPV and References
DPV NO: 43 June 1971
Family: Secoviridae
Genus: Comovirus
Species: Squash mosaic virus | Acronym: SqMV
Squash mosaic virus
R. N. Campbell Dept. of Plant Pathology, University of California, Davis, California 95616, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Freitag (1941,
1956) and
Lindberg, Hall & Walker (1956).
Selected synonyms
- Cucurbitavirus maculans
(Rev. appl. Mycol. 38: 677) - Cucurbit ring mosaic virus (Rev. appl. Mycol. 20: 189)
- Muskmelon mosaic virus (Rev. appl. Mycol. 27: 271)
- Marmor melonis (Rev. appl. Mycol. 27: 271)
- Muskmelon mosaic virus (Rev. appl. Mycol. 27: 271)
-
An RNA-containing virus possessing three types of isometric particles all about 30 nm in diameter. Transmitted by mechanical inoculation, by beetles and by embryo infection of seeds. Most hosts are in the Cucurbitaceae. Common in Western Hemisphere; one report from Israel.
Main Diseases
One of several viruses causing mosaic diseases of cucurbits. Infected plants may be symptomless or may show ring patterns, severe blister mottle, deformation and, occasionally, enations. Fruit symptoms range from small chlorotic areas to severe deformation with raised dark green areas.
Geographical Distribution
Widely distributed in Western Hemisphere, one report from Israel. Because it is seed-borne it has probably been introduced wherever seeds have been imported from the Western Hemisphere. Unless insect vectors are present, it probably will not spread in new localities.
Host Range and Symptomatology
Transmissible by inoculation of sap. Natural host range limited to Cucurbitaceae, in which most species are susceptible. Experimentally, it also infects plants in other families: Amaranthaceae, Chenopodiaceae, Hydrophyllaceae, Leguminosae and Umbelliferae (Freitag, 1956).
-
Diagnostic species
- Cucurbita pepo
(summer squash). Severe systemic mosaic with leaf deformation (Fig. 1, Fig. 2) and ring patterns. Fruits often deformed (Fig. 4). - Citrullus lanatus (C. vulgaris). Immune, or may develop necrotic
local lesions.
- Cucumis sativus (cucumber). Systemic vein-clearing, yellow vein-banding and yellow spots (Fig. 3).
-
Propagation species
- Cucurbita pepo
cvs. Small Sugar pumpkin or Early Prolific Straight-neck squash have been generally used.Assay species
- The virus may be assayed by determining the proportion of C. pepo plants that become systemically infected. Apparently, no local lesion host has been used but Early Yellow Summer Crookneck squash was reported to give chlorotic local lesions consistently (Demski, 1969).
Strains
Many strains have been distinguished by host reaction. Freitag (1941, 1956) distinguished the type virus, which caused severe mottle in squash and occurred in coastal areas of California, and the cucurbit ring mosaic virus, which caused ring patterns in squash (Fig. 5) and occurred in the central valleys of California. Lindberg et al. (1956) showed that the latter virus is a strain of squash mosaic virus, as also are typical muskmelon mosaic virus and latent muskmelon mosaic virus (Anderson, 1954). The watermelon stunt strain, infecting Citrullus lanatus systemically (Fig. 6, Fig. 7), has been reported from Arizona, USA (Nelson, Matejka & McDonald, 1965). A virus causing mild yellow mosaic of Ecballium elaterium in Israel has also been identified as squash mosaic virus (Cohen & Nitzany, 1963).
Transmission by Vectors
Transmissible by chrysomelid beetles (Diabrotica spp. and Acalymma spp.) (Freitag, 1956; Lastra, 1968; Sitterly, 1960; Stoner, 1963), a coccinellid beetle (Cohen & Nitzany, 1963) and a grasshopper (Stoner, 1963). Starved beetles acquire the virus in 5 min access periods. The latent period, if any, is less than 10 h. Inoculation access period is less than 24 h; shorter times have not been tested. Virus is retained up to 20 days following a 10-15 day acquisition access period; shorter retention periods result from shorter acquisition access periods. Virus multiplication in the vector has not been reported. Virus has been recovered from regurgitation fluid, faeces and haemolymph.
Transmission through Seed
Virus is frequently transmitted through seed of Cucurbita moschata, C. pepo, C. maxima, C. mixta and Cucumis melo. Commercial and experimental seed lots generally yield about 1% of infected seedlings (Grogan, Hall & Kimble, 1959) but up to 94% transmission has been reported (Rader, Fitzpatrick & Hildebrand, 1947). The watermelon stunt strain is reported to be seed-transmitted more frequently than the common strain (Nelson & Knuhtsen, 1969).
Transmission by Dodder
Apparently not tested.
Serology
The virus is strongly immunogenic. Antisera have been prepared in rabbits or guinea pigs. Reactions may be done by tube-precipitin or agar double-diffusion tests. Double-diffusion tests yield a single band of precipitate.
Relationships
Slight serological differences exist between the common and watermelon stunt strains (Knuhtsen & Nelson, 1968). Cross protection has been reported among 3 strains of squash mosaic virus but squash mosaic virus did not protect against cucumber mosaic, watermelon mosaic or tobacco ringspot viruses (Demski, 1969).
Squash mosaic virus belongs to the cowpea mosaic virus group which also includes bean pod mottle, broad bean stain, Echtes Ackerbohnemosaik, red clover mottle and radish mosaic viruses. Serological relationships have been shown among many of these viruses (Campbell, 1964; Gibbs et al., 1966), and their base ratios, types of particles and vectors are similar. No cross protection tests have been reported between squash mosaic virus and other viruses in the cowpea mosaic virus group.
Stability in Sap
In sap of various cucurbits, the thermal inactivation point (10 min) ranges from 70 to 80°C, and the longevity in vitro at about 20°C is greater than 4 weeks. The dilution end-point ranges from 10-4 to 10-6.
Purification
The virus has been purified by two methods. Yields are 50 to 400 mg/l of sap.
1. Mazzone, Incardona & Kaesberg (1962).
Grind frozen leaves, extract sap,
adjust it to pH 5.0 and incubate
for 4 h. After low speed centrifugation, adjust supernatant fluid to pH 7.0 and
make 45% saturated with (NH4)2SO4. After 5 h
sediment the precipitate and dissolve it in phosphate buffer (0.02 ionic strength,
pH 7.0). Then use two cycles of differential centrifugation. Others have omitted
the (NH4)2SO4 precipitation step. Sucrose density
gradient centrifugation can be used.
2. Lastra & Munz (1969). Homogenize infected leaves with 2 vol (w/v) of 0.1 M phosphate buffer, pH 7.5. Clarify by low speed centrifugation and to the supernatant fluid add polyethylene glycol (M. Wt = 6000) to 6% final concentration and NaCl to 0.2 M. Stir for 1 h at 4°C, collect the precipitate by centrifugation and resuspend in 0.01 M phosphate buffer, pH 7.2. Use two cycles of differential centrifugation. Further purification by molecular exclusion chromatography in columns of agarose beads or by zone electrophoresis is necessary to remove 17 S host components.
Properties of Particles
Purified preparations contain three kinds of particles (T, M, and B) with sedimentation coefficients (s20,w) at infinite dilution (svedbergs) of 57 (T), 95 (M) and 118 (B) (Mazzone et al., 1962).
Molecular weights (daltons): 4.5 x 106 (T), 6.1 x 106 (M), 6.9 x 106 (B).
Absorbances at 260 nm (1 mg/ml, 1 cm light path): 6.8 (M), 8.65 (B).
Diffusion coefficients (D20,w): 1.31 x 10-7 cm2 sec-1 (those of individual components do not differ significantly).
There is evidence from centrifuging in RbCl density gradients that bottom component contains particles of two slightly different buoyant densities (Mazzone et al., 1962).
During electrophoresis in several 0.01 ionic strength buffers (pH 4.75 to 8.2) only one peak was observed; the isoelectric point was pH 4.65 (Rice et al., 1955).
The bottom component, when separated by a single density gradient fractionation, is almost as infective as the unfractionated preparation (Mazzone et al., 1962; Lastra & Munz, 1969). However, further experiments are needed in view of results reported for cowpea mosaic and bean pod mottle viruses, with which a mixture of middle and bottom components is necessary for infection.
Particle Structure
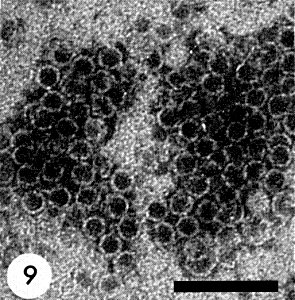
All three components are isometric with a diameter of about 30 nm and 5-6 sided angular outlines (Fig. 8, Fig. 9, Fig. 10). No regularly arranged subunits can be seen in electron micrographs of negatively stained preparations. Particles of top components are penetrated by negative stains (Fig. 9) (Lastra & Munz, 1969).
Particle Composition
RNA: presumably single-stranded, about 26.8% (M) and 34.8% (B) of the particle weight. Molecular weight (daltons): 1.6 x 106 (M), 2.4 x 106 (B). Molar percentages of nucleotides, averaged for middle and bottom components (which were not significantly different): G23; A32; C16; U30 (Mazzone et al., 1962).
Protein: About 100% (T), 73.2% (M) and 65.2% (B) of the particle weight. Amino acid composition given by Mazzone et al. (1962).
Relations with Cells and Tissues
No information.
Notes
Because it is seed-borne, squash mosaic is a virus to beware of when cucurbits are used as hosts for other viruses. It is readily distinguishable from two other viruses that occur commonly in cucurbits: cucumber mosaic virus has a wide host range (including Nicotiana glutinosa and Vigna sinensis) and is aphid-borne; watermelon mosaic virus has flexuous filamentous particles and is aphid-borne. Squash mosaic virus is rapidly and easily identified by gel-diffusion serological tests using crude sap from infected plants.
Figures

Systemic symptoms in leaves of Zucchini squash. (Courtesy J. H. Freitag, University of California, Berkeley.)

Systemic symptoms in leaves of Early White Bush Scallop squash. (Courtesy J. H. Freitag, University of California, Berkeley.)

Systemic symptoms in leaves of Snow’s pickling cucumber. (Courtesy J. H. Freitag, University of California, Berkeley.)

Systemic symptoms on fruit of Black Zucchini squash. (Courtesy J. H. Freitag, University of California, Berkeley.)

Systemic symptoms of cucurbit ring mosaic strain in leaf of Early White Bush Scallop squash. (Courtesy J. H. Freitag.)

Systemic symptoms of watermelon stunt strain in watermelon leaves. (Courtesy M. R. Nelson, University of Arizona, Tucson.)

Systemic symptoms of watermelon stunt strain in watermelon leaves. (Courtesy M. R. Nelson, University of Arizona, Tucson.)

Electron micrograph of particles in sodium phosphotungstate; particles packed in hexagonal array. Bar represents 100 nm. (Courtesy R. Lastra, Instituto Venezolano de Investigaciones Cientificas, Caracas.)
References list for DPV: Squash mosaic virus (43)
- Anderson, Phytopathology 44: 371, 1954.
- Campbell, Phytopathology 54: 1418, 1964.
- Cohen & Nitzany, Phytopathology 53: 193, 1963.
- Demski, Phytopathology 59: 251, 1969.
- Freitag, Phytopathology 31: 8, 1941.
- Freitag, Phytopathology 46: 73, 1956.
- Gibbs, Hecht-Poinar, Woods & McKee, J. gen. Microbiol. 44: 177, 1966.
- Grogan, Hall & Kimble, Phytopathology 49: 366, 1959.
- Knuhtsen & Nelson, Phytopathology 58: 345, 1968.
- Lastra, Pl. Dis. Reptr 52: 171, 1968.
- Lastra & Munz, Phytopathology 59: 1429, 1969.
- Lindberg, Hall & Walker, Phytopathology 46: 489, 1956.
- Mazzone, Incardona & Kaesberg, Biochim. biophys. Acta 55: 164, 1962.
- Nelson & Knuhtsen, Phytopathology 59: 1042, 1969.
- Nelson, Matejka & McDonald, Phytopathology 55: 1362, 1965.
- Rader, Fitzpatrick & Hildebrand, Phytopathology 37: 809, 1947.
- Rice, Lindberg, Kaesberg, Walker & Stahmann, Phytopathology 45: 145, 1955.
- Sitterly, Pl. Dis. Reptr 44: 134, 1960.
- Stoner, Phytopathology 53: 890, 1963.