Details of DPV and References
DPV NO: 57 June 1971
Family: Unallocated ssRNA+ viruses
Genus: Sobemovirus
Species: Southern bean mosaic virus | Acronym: SBMV
There is a more recent description of this virus: DPV 274
Southern bean mosaic virus
R. J. Shepherd Department of Plant Pathology, University of California, Davis, California 95616, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Zaumeyer & Harter, 1943.
Selected synonyms
- Bean mosaic virus 4 (Rev. appl. Mycol. 21: 478)
- Southern bean mosaic virus 1 (Rev. appl. Mycol. 23: 203)
- Marmor laesiofaciens (Rev. appl. Mycol. 23: 467)
- Phaseolusvirus laedens (Rev. appl. Mycol. 38: 677)
- Bean virus 4 (Rev. appl. Mycol. 31: 163)
- Southern bean mosaic virus 1 (Rev. appl. Mycol. 23: 203)
-
A virus with isometric particles about 28 nm in diameter containing single-stranded RNA. It has a very restricted host range, is transmitted by beetles, by seed and by inoculation of sap. It occurs predominantly in warm temperate and tropical regions.
Main Diseases
Causes mosaic and mottle diseases of beans and cowpeas.
Geographical Distribution
Warm temperate and tropical regions of the Americas and Africa. Recently reported in France (Férault et al., 1969).
Host Range and Symptomatology
Host range is very narrow; only species of Leguminosae are susceptible to infection (Zaumeyer & Harter, 1943; Shepherd & Fulton, 1962). Strains of the virus from bean may not infect cowpea and isolates from cowpea may not infect bean. Readily transmissible by inoculation of sap.
-
Diagnostic species
- Phaseolus vulgaris
(common bean). Some varieties produce only necrotic local lesions (Fig. 2) and do not become systemically infected; others produce no local reactions but become systemically infected (Zaumeyer & Harter, 1943; Grogan & Kimble, 1964). Systemic infections appear as a prominent chlorotic mottle and mosaic in some varieties (Fig. 1) but in others may be so mild as to be readily overlooked. Puckering and vein-banding may be present. - P. lunatus (lima bean). Only small-seeded varieties are susceptible;
these produce only
local lesions.
- P. aureus (mung bean). Small necrotic local lesions.
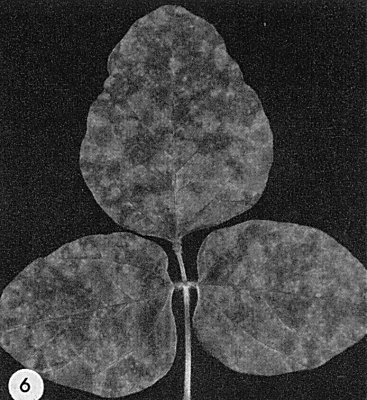
- Glycine max (soybean). Systemic mottle (Fig. 6).
- Vigna sinensis (cowpea). Some varieties develop small necrotic local lesions (Fig. 3) without subsequent systemic infection. Other varieties produce local chlorotic lesions followed by a marked mottle, coarse mosaic (Fig. 5), or green vein-banding.
- P. aureus (mung bean). Small necrotic local lesions.
-
Propagation species
- Systemically susceptible varieties of bean or cowpea are suitable plants for maintaining cultures and providing virus for purification (Shepherd & Fulton, 1962; Grogan & Kimble, 1964).
-
Assay species
- Any of the various varieties of bean or cowpea that produce local necrotic lesions after inoculation are useful for bioassay of the virus (Zaumeyer & Harter, 1943; Kuhn & Brantley, 1963).
Strains
In addition to some minor variants (Zaumeyer & Harter, 1943; Magdoff-Fairchild, 1967), several major variants have been described:
Cowpea strain (Shepherd & Fulton, 1962). Infects cowpea, yardlong bean (Vigna sesquipedalis) and certain other legumes, but not common bean.
Severe bean mosaic strain (Yerkes & Patino, 1960; Grogan & Kimble, 1964). Produces reactions in bean varieties different from those produced by the original isolate described by Zaumeyer & Harter (1943). Many bean varieties produce both local and systemic reactions with systemic necrosis developing in some varieties.
Electrophoretic and buoyant density variants have also been described (Magdoff-Fairchild, 1967).
Transmission by Vectors
Transmissible by the bean leaf beetle (Ceratoma trifurcata) after a 48 hr acquisition feeding period. Retained for at least 5 days by feeding insects (Walters, 1964).
Transmission through Seed
The virus may be seed-borne in beans (Zaumeyer & Harter, 1943); a high percentage of immature seed embryos of bean carry the virus (Cheo, 1955). In cowpea var. Early Wilt Resistant Ramshorn, the virus was transmitted to progeny seedlings through 3-4% of the seed from infected plants (Shepherd & Fulton, 1962).
Serology
The virus is highly immunogenic. Purified virus produces a granular (somatic) type of precipitate in tube-precipitin tests. The virus diffuses well in agar gels to produce a single band of precipitate. Gel-precipitin tests with expressed sap from infected plants provide a rapid and specific diagnostic test.
Relationships
Strains that differ in host range or other properties are closely related serologically in tests with narrow-spectrum antiserum (Shepherd & Fulton, 1962; Grogan & Kimble, 1964). No distantly serologically related viruses have been reported.
Stability in Sap
In bean or cowpea sap: dilution end-point, 10-5-10-6; thermal inactivation point (10 min) 90-95°C; longevity (18-22°C), 20-165 days (Zaumeyer & Harter, 1942; Yerkes & Patino, 1960; Shepherd & Fulton, 1962).
Purification
Homogenize tissue in 0.25 M phosphate buffer (pH 7.5) and clarify by low speed centrifugation after adjusting to pH 4.5, or after stirring in n-butanol to 10% (v/v) to precipitate host chloroplastic material. Give clarified homogenates 2-3 cycles of differential centrifugation (c. 8000 and 100,000 g) with 0.025 M phosphate as solvent (Shepherd & Fulton, 1962). Cautious use of low speed centrifugation with virus suspensions adjusted to pH 4.5 may be needed for further removal of Fraction I protein.
Properties of Particles
Sedimentation coefficient (s20,w) at infinite dilution: 115 S. No accessory particles are detected by analytical centrifugation.
Molecular weight: 6.6 x 106 (Miller & Price, 1946); 6.1 x 106 (Lauffer, Taylor & Wunder, 1952).
Diffusion coefficient: 1.34 x 10-7 cm2/sec (Miller & Price, 1946).
Isoelectric point: 3.9-6.0 depending on the strain (Magdoff-Fairchild, 1967).
Partial specific volume: 0.696-0.700 (Miller & Price, 1946; Lauffer et al., 1952).
Electrophoretic mobility: -4.0 x 10-5 cm2 sec-1 volt-1 at pH 7 and ionic strength of 0.02 for one strain (Hartman & Lauffer, 1953).
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 5.85.
A260/A280: 1.6.
Particle Structure
Particles are isometric, 25 nm diameter when packed in crystals (Price, Williams & Wyckoff, 1946), but appear somewhat larger when suitably stained (Fig. 4).
Particle Composition
RNA: Single-stranded, M. Wt 1.4 x 106 (Diener, 1965). Molar percentages of nucleotides: G27; A22-24; C22-24; U27 (Ghabrial, Shepherd & Grogan, 1967). RNA is 21% of particle weight. Sedimentation coefficient is 25 S (Diener, 1965).
Protein: The particle contains a single species of protein with about 270 amino acid residues and a M. Wt of 29,000 (Tremaine, 1966; Ghabrial et al., 1967).
Relations with Cells and Tissues
Virus particles are found both in the cytoplasm and in the nucleus of infected cells (De Zoeten & Gaard, 1969). The cowpea strain forms prominent crystalline arrays in cytoplasm and nucleus of plants infected for less than 2 weeks (Weintraub & Ragetli, 1970).
Notes
The virus can be distinguished from others common in legumes (except the legume strain of tobacco mosaic virus) by its high thermal inactivation point, its longevity in sap and high dilution end-point. It can be distinguished from the legume strain of tobacco mosaic virus and most other viruses found in legumes by its unusually restricted host range.
Figures
References list for DPV: Southern bean mosaic virus (57)
- Cheo, Phytopathology 45: 17, 1955.
- De Zoeten & Gaard, J. Cell Biol. 40: 814, 1969.
- Diener, Virology 27: 425, 1965.
- Férault, Spire, Bannerot, Bertrandy & Le Tan, Annls Phytopath. 1: 619, 1969.
- Ghabrial, Shepherd & Grogan, Virology 33: 17, 1967.
- Grogan & Kimble, Phytopathology 54: 75, 1964.
- Hartman & Lauffer, J. Am. chem. Soc. 75: 6205, 1953.
- Kuhn & Brantley, Pl. Dis. Reptr 47: 1094, 1963.
- Lauffer, Taylor & Wunder, Archs Biochem. Biophys. 40: 453, 1952.
- Magdoff-Fairchild, Virology 31: 142, 1967.
- Miller & Price, Archs Biochem. 10: 467, 1946.
- Price, Williams & Wyckoff, Archs Biochem. 9: 175, 1946.
- Shepherd & Fulton, Phytopathology 52: 489, 1962.
- Tremaine, Virology 30: 348, 1966.
- Walters, Pl. Dis. Reptr 48: 935, 1964.
- Weintraub & Ragetli, J. Ultrastruct. Res. 32: 167, 1970.
- Yerkes & Patino, Phytopathology 50: 334, 1960.
- Zaumeyer & Harter, Phytopathology 32: 438, 1942.
- Zaumeyer & Harter, J. agric Res. 67: 305, 1943.