Details of DPV and References
DPV NO: 68 October 1971
Family: Virgaviridae
Genus: Hordeivirus
Species: Barley stripe mosaic virus | Acronym: BSMV
There is a more recent description of this virus: DPV 344
Barley stripe mosaic virus
J. G. Atabekov Laboratory of Bioorganic Chemistry, Moscow State University, USSR
V. K. Novikov Laboratory of Bioorganic Chemistry, Moscow State University, USSR
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
McKinney (1951).
Selected synonym
- Barley false stripe virus (Rev. appl. Mycol. 30: 408)
-
An RNA-containing virus with short tubular particles c. 128 x 20 nm. It has a narrow host range and is seed- and pollen-transmitted. Vector unknown. World-wide distribution.
Main Diseases
Only natural hosts known are wheat (Triticum aestivum) and barley (Hordeum vulgare) in which it causes diseases ranging in severity from very mild stripe mosaic to lethal necrosis (McKinney & Greeley, 1965).
Geographical Distribution
World-wide.
Host Range and Symptomatology
Natural hosts are members of the Gramineae, but species of Chenopodiaceae and Solanaceae can be experimentally infected.
-
Diagnostic species
- Hordeum vulgare
(barley), Triticum aestivum (wheat) and Avena sativa (oat). Systemic stripe mosaics of different types (Fig. 1). -
Chenopodium amaranticolor, C. quinoa, C. album. Large chlorotic yellow lesions;
not systemic
(Fig. 2).
- Beta vulgaris (beet). Chlorotic yellow local lesions; not systemic.
- Some strains produce systemic infection (stripe mosaic) in Zea mays (maize), mosaic in Spinacea oleracca (spinach) and local chlorotic lesions in Nicotiana tabacum cv. Samsun (tobacco).
- Beta vulgaris (beet). Chlorotic yellow local lesions; not systemic.
-
Propagation species
- Hordeum vulgare
and Triticum aestivum are the best sources of virus for purification.Assay species
- Chenopodium amaranticolor
and C. quinoa are useful local lesion hosts.
Strains
Several strains of the virus (McKinney & Greeley, 1965), producing symptoms of different severity in barley, wheat and oat, have been named the latent, mild, moderate, severe, yellow leaf, white-leaf, dwarf and necrotic strains. The mild and moderate strains are the most common. The Russian strain of the virus (Atabekov & Novikov, 1966; Novikov & Atabekov, 1970) does not infect Zea mays. Most isolates have been maintained successfully in culture for many transfers but some variants become difficult or impossible to transfer by sap inoculation (McKinney & Greeley. 1965; Novikov, unpublished results). The reason for this change is not known.
Transmission by Vectors
None reported.
Transmission through Seed
Transmitted through seed to 90% (McKinney, 1953) or even 100% (Gold et al., 1954). The virus is also pollen-borne and infects the pollinated plants (Gold et al., 1954; Gardner, 1967). The virus affects the inheritance of characters by Zea mays, in which it is not seed-borne (Sprague, McKinney & Greeley, 1963).
Serology
The virus is highly immunogenic. In immunodiffusion tests, intact virus usually forms a single precipitin line close to the antigen well. Protein(s) of low molecular weight and antigenically related to the protein in the virus particles occur in extracts of infected leaves (Hamilton & Ball, 1966).
Virus protein in monomeric (fully dissociated) form is serologically related but not identical to that in the intact virus particles but aggregates of the viral protein that have sedimentation coefficients greater than 10 S are antigenically indistinguishable from each other and from intact virus particles. In immunodiffusion tests in the presence of excess antibody, purified preparations of the virus and its protein produce multiple precipitin lines (Atabekov et al., 1968a).
Relationships
The virus is distantly serologically related to lychnis ringspot virus (Gibbs et al., 1963) but is not related to viruses of the tobacco mosaic virus group.
Stability in Sap
In barley sap the thermal inactivation point (10 min) is about 70°C (McKinney, 1951; Kassanis & Slykhuis, 1959). The virus can be stored in dehydrated tissue for as long as 12 years (McKinney & Greeley, 1965). Dilution end-point of sap varies greatly but is usually about 0.5-2.0 x 10-3. Purified preparations frequently have little infectivity (Brakke, 1959) or none (Atabekov & Novikov, 1966). However, when the RNA of completely non-infective preparations is extracted by phenol treatment it is frequently found to be infective (Atabekov & Novikov, 1966; Novikov & Atabekov, 1970).
Purification
(Novikov, 1970). Use fresh systemically infected barley leaves; do not freeze. Homogenize the leaves in 2-5 volumes of 0.05-0.1 M phosphate buffer, pH 7.2. Filter and adjust the pH to 7.5. Clarify the extract by shaking it with 1/8 volume chloroform for 15-20 min and centrifuging it at low speed. Precipitate the virus by adding ammonium sulphate to 22% (w/v), centrifuge and resuspend the pellet in 0.01 M Tris-HCl buffer, pH 7.2. Concentrate the virus by one cycle of differential centrifugation and resuspend the pellet in 0.03 M Tris-HCl buffer, pH 7.0-7.2. Paracrystals can be obtained by adding MgCl2 to 0.05-0.1 M, and centrifuging at low speed. Resuspend the pellet in 0.05 M Tris-HCl buffer, pH 7.5 with 0.02 M disodium ethylene diamine tetraacetate. Further purify the virus by two cycles of differential centrifugation. Yields are 100-200 mg per kg fresh leaf material.
Store the purified virus as paracrystals in 0.05-0.1 M MgCl2 at 5°C. The virus can be crystallized from purified preparations but not from crude plant extracts. Greater concentrations of divalent cations may damage the virus and dissociate it into RNA and protein (Atabekov et al., 1968b). Stability of purified virus is greatest between pH 6.0 and 7.5 (Brakke, 1962). The virus precipitates at its isoelectric point, but much virus is lost at this pH, so that this property should not be used for purifying the virus.
Properties of Particles
Sedimentation coefficient (s20,w) at infinite dilution in 0.01 M phosphate buffer, pH 7.2: about 185 S (Atabekov & Novikov, 1966). Intrinsic viscosity (z) of these preparations: 0.12-0.17 dl/g. Additional components with sedimentation coefficients of 178 and 200 S are found in purified preparations (Harrison, Nixon & Woods, 1965). In 0.1 M phosphate buffer the particles aggregate and this produces a change in the hydrodynamic properties of the preparations (s20,w=263 S; (z)=0.58 dl/g).
Molecular weight: c. 26 x 106 daltons (for 185 S particles).
Isoelectric point: c. pH 4.5.
Electrophoretic mobility: -21.0 x 10-5 cm2 sec-1 volt-1 at pH 7.7 in 0.05 M phosphate buffer (Russian strain).
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 2.6, but a maximum value of 2.7 is obtained at 270-272 nm.
A260/A280: 1.00-1.05.
A270/A280: 1.06-1.12.
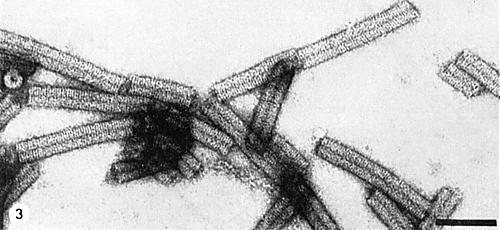
Particle Structure
Particles are tubular rods (130 x 20 nm) (Fig. 3) and are helically constructed with the pitch of the basic helix 2.5-2.6 nm. Diameter of the internal hole: 3.0-4.0 nm (Gibbs et al., 1963; Harrison et al., 1965; Finch, 1966). Particles in crude sap are reported to have a single modal length of 130 nm (Gold et al., 1954; Shikata, 1966), whereas particles in purified preparations have additional maxima at 112 and 150 nm (Harrison et al., 1965; Novikov, unpublished).
Particle Composition
RNA: M. Wt. c. 1 x 106, about 3.8-4.0% of particle weight. Single-stranded. Molar percentage of nucleotides: G20.3; A30.9; C19.4; U29.4 (Novikov & Atabekov, unpublished). Sedimentation coefficient in 0.1 M NaCl is 20 S, but smaller components may be present in heterogeneous preparations.
Protein: Subunits (l.8 S) have M. Wt 21.5 x 103 and contain 187-190 amino acid residues (Atabekov et al., 1968b). Amino acid composition (residues per protein monomer): asp 25-26; thr 9; ser 9; glu 19-20; pro 12-13; gly 8; ala 20; val 10; ile 6; leu 21; tyr 8; phe 7; his 4; lys 7; arg 17; trp 5; cys 0; met 0. Native subunits have little or no a-helix. Isoelectric point: pH 5.3. The viral protein can be repolymerized in vitro (Atabekov et al., 1968b; Gumpf & Hamilton, 1968).
Relations with Cells and Tissues
All tissues are infected. Mature particles accumulate in the cytoplasm and the nuclei; they also occur in the cytoplasm of pollen cells (Gardner, 1967). The virus can survive and, probably, multiply in haploid cells of barley (Gardner, 1967). It induces peripheral vesicles in chloroplasts (Carroll, 1970).
Figures
References list for DPV: Barley stripe mosaic virus (68)
- Atabekov & Novikov, Biokhimia, U.S.S.R. 31: 157, 1966.
- Atabekov, Dementyeva, Schaskolskaya & Sacharovskaya, Virology 36: 601, 1968a.
- Atabekov, Novikov, Kiselev & Kaftanova, Virology 36: 620, 1968b.
- Brakke, Virology 9: 506, 1959.
- Brakke, Virology 17: 131, 1962.
- Carroll, Virology 42: 1015, 1970.
- Finch, Nature, Lond. 212: 349, 1966.
- Gardner, Phytopathology 57: 1315, 1967.
- Gibbs, Kassanis, Nixon & Woods, Virology 20: 194, 1963.
- Gold, Suneson, Houston & Oswald, Phytopathology 44: 115, 1954.
- Gumpf & Hamilton, Virology 35: 87, 1968.
- Hamilton & Ball, Virology 30: 661, 1966.
- Harrison, Nixon & Woods, Virology 26: 284, 1965.
- Kassanis & Slykhuis, Ann. appl. Biol. 47: 254, 1959.
- McKinney, Phytopathology 41: 563, 1951.
- McKinney, Pl. Dis. Reptr 37: 292, 1953.
- McKinney & Greeley, Tech. Bull. U.S. Dep. Agric. 1324, 84 pp., 1965.
- Novikov, Doctoral Dissertation, Moscow State University, 1970.
- Novikov & Atabekov, Virology 41: 101, 1970.
- Shikata, J. Fac. Agric. Hokkaido (imp.) Univ. 55(1), 110 pp., 1966.
- Sprague, McKinney & Greeley, Science, N.Y. 141: 1052, 1963.