Details of DPV and References
DPV NO: 73 October 1971
Family: Potyviridae
Genus: Potyvirus
Species: Bean common mosaic virus | Acronym: BCMV
There is a more recent description of this virus: DPV 337
Bean common mosaic virus
L. Bos Institute of Phytopathological Research, Wageningen, The Netherlands
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Stewart & Reddick (1917) and
Pierce (1934).
Selected synonyms
- Bean mosaic virus (Stewart & Reddick, 1917)
- Bean virus 1 (Rev. appl. Mycol. 13: 488)
- Phaseolus virus 1 (Rev. appl. Mycol. 17: 52)
- Bean virus 1 (Rev. appl. Mycol. 13: 488)
-
A virus with flexuous filamentous particles c. 750 nm long, transmitted readily by inoculation of sap, by several aphid species in the non-persistent manner, through a high proportion of seed, and through pollen. In nature it is mainly restricted to Phaseolus species and is probably common wherever these crops are grown.
Main Diseases
Causes ‘common mosaic’ (usually associated with leaf malformation and rolling) and ‘black-root’ (characterized by vascular necrosis and plant death) in Phaseolus spp., the nature and severity of symptoms greatly depending on cultivar, time of infection and environmental conditions. French bean (Phaseolus vulgaris) is the common natural host, but runner bean (P. coccineus) is also naturally infected (Ainsworth, 1940).
Geographical Distribution
World-wide.
Host Range and Symptomatology
Readily transmissible by inoculation of sap to many French bean cultivars. A German isolate infected 43 leguminous species and produced local lesions in Chenopodium quinoa (Quantz, 1961). Two typical strains caused local infection of C. quinoa and Gomphrena globosa and systemic infection of Nicotiana clevelandii (Bos, 1970a).
-
Diagnostic species
- Phaseolus vulgaris
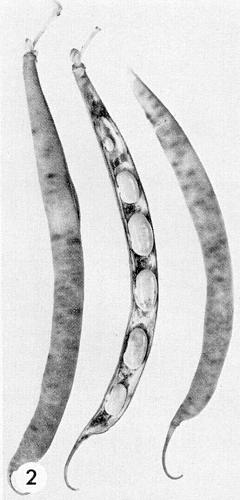
(French bean). Bean cultivars may be tolerant, sensitive or hypersensitive, but the kind of reaction depends greatly on the virus strain. Tolerant cultivars may show only slight narrowing of systemically infected leaves. Sensitive cultivars may show a reddish rugose mosaic in lower leaves, a rolling or curling mosaic with dark green areas along the main veins in the upper leaves (Fig. 1) and mottled or malformed pods. Severe stunting and ‘black-root’ symptoms occur with typical strains at temperatures above 30°C (Grogan & Walker, 1948a) and with virulent strains at 20°C and above (Fig. 5) (Harrison, 1935; Hubbeling, 1955); the vascular necrosis often extends into surrounding tissues, for example in stems and pods (Fig. 2) or in leaves around the sites of infection (Fig. 4, bottom). Hypersensitive cultivars react with necrotic local lesions at normal temperatures (Fig. 4, top) and with ‘black-root’ symptoms at high temperature; this type of reaction gives field resistance, is genetically dominant and is derived from the cultivar Corbett Refugee.Propagation species
- Phaseolus vulgaris
(French bean). Slightly sensitive cultivars are suitable for propagating the virus. It can also be maintained in seed from infected plants. Field-infected plants are unsuitable sources because they contain little virus (Bercks, 1959); in glasshouse-grown plants the inoculated leaves contain more virus than do systemically infected leaves (Scott, 1962).Assay species
- Phaseolus vulgaris.
Sensitive cultivars usually develop a characteristic systemic mosaic (Fig. 1). Some cultivars develop necrotic local lesions with certain strains of the virus (Zaumeyer & Goth, 1963). Leaves of hypersensitive cultivars (e.g. Topcrop) detached after inoculation and incubated on moist filter paper in closed Petri dishes under artificial light at 30-32°C may develop necrotic local lesions after 2-3 days (Quantz, 1957, 1958), but this test is not always reliable, and depends on leaf age and external conditions. - Phaseolus lathyroides gives necrotic local lesions
(Quantz, 1961)
and Chenopodium quinoa
may give diffuse chlorotic local lesions 2-3 mm in diameter.
- For aphid transmission tests Phaseolus vulgaris has been used.
Strains
Various strains have been described, especially in connection with research on
breeding for resistance
in Phaseolus; they differ in the symptoms they induce and in the range of
cultivars they infect.
Some virus isolates described as separate entities are perhaps strains of bean common
mosaic virus
(see Relationships). Well known or well characterized strains include the following:
Type strain of
Reddick & Stewart (1918).
The most prevalent strain in USA.
New York 15 or Burkholder strain of
Richards & Burkholder (1943).
Infects cultivars
Robust, Michelite and Great Northern U.I. Nr. 15, which are resistant to the type strain.
Florida strain of
Zaumeyer & Goth (1964).
More virulent than the type and New York
strains and has a slightly different host range. It does not give the
hypersensitive reaction at
34°C.
Other strains reported from North America include the B strain of Dean & Wilson (1959) and the Mexican strain of Silbernagel (1969); strains from Europe include the Voldagsen and Marienau strains of Frandsen (1952), the Rolmozaïek and Westlandia strains of van der Want (1954) and the Michelite, Imuna and Great Northern strains of Hubbeling (1963).
Transmission by Vectors
Several aphid species have been reported to transmit the virus in the non-persistent manner (Kennedy, Day & Eastop, 1962; Zettler, 1966), notably Acyrthosiphon pisum, Aphis fabae and Myzus persicae. Acquisition and inoculation thresholds are less than 1 min and there is no latent period (van der Want, 1954; Zettler & Wilkinson, 1966). Aphids transmit virus more readily from chlorotic leaf areas than from dark green areas (Zettler, 1969).
Transmission through Seed
Rather common and probably the most important source of initial crop infection. Up to 83% (Reddick & Stewart, 1919; Crowley, 1957; Schippers, 1963) of the seeds of diseased plants may be infected. Plants infected after flowering do not give infected seed (Nelson, 1932; Schippers, 1963), and the distribution of infected seeds in pods is erratic (EI-Attar, Sabet & Nour-Eldin, 1964). Virus is found in the embryo and cotyledons but seldom in the seed-coat (Quantz, 1962); ground seeds may be a good source of inoculum (Cafati, 1968). Active virus has been recovered from seed stored for 30 years (Pierce & Hungerford, 1929). Virus enters the seed, and perhaps also the plant pollinated, from infected pollen (Reddick, 1931).
Transmission by Dodder
Not reported.
Serology
It is hard to obtain sufficiently pure and concentrated virus suspensions for serological tests, and in field-infected plants virus concentration may be too low to detect serologically (Bercks, 1959). Highest reported antiserum titre is 1/2048 (Bercks, 1959; Zaumeyer & Goth, 1964).
Precipitin-tube and micro-precipitin tests are most commonly used because intact particles do not diffuse in agar gel. Jermoljev & Chod (1966) described a serological method for detecting the virus in germinated seeds.
Relationships
Zaumeyer & Goth (1964) found that antiserum to the type strain had the same titre to both the type strain and the Florida strain. Bercks (1959) found complete cross-protection between two German strains, but Silbernagel (1969) found that cross-protection between the Florida, New York 15 and Mexican strains was usually only partial, although the Florida strain did protect Bountiful bean from infection with the Mexican strain.
In particle morphology, mode of transmission and other biological and biophysical properties, bean common mosaic virus resembles members of the potato virus Y group of viruses and is serologically related to several of them. Of these, other seed-borne viruses of legumes, such as soybean mosaic, cowpea aphid-borne mosaic and pea leafroll mosaic (pea seed-borne mosaic) viruses, seem to be close relatives. Bean yellow mosaic (Beemster & van der Want, 1951; Bercks, 1960), pea necrosis and wisteria vein mosaic viruses (Bos, 1970a, 1970b) have wider host ranges and may be less closely related. Partial or complete cross-protection in plants has sometimes been found between bean common mosaic and bean yellow mosaic viruses (Grogan & Walker, 1948b; Quantz, 1961) and between each of these viruses and soybean mosaic virus (Quantz, 1961).
Bean common mosaic virus may also be closely related to the following incompletely described viruses: adzuki bean (Phaseolus radiatus var. aurea) mosaic virus (Matsumoto, 1922), asparagus bean (Vigna sesquipedalis) mosaic virus (Snyder, 1942; Inouye, 1969), and mung bean (Phaseolus lunatus) mosaic virus (Kaiser et al., 1968). The seed-borne bean western mosaic virus, described as a separate virus because of its wide host range (Skotland & Burke, 1961), is also now considered a strain of bean common mosaic virus (Silbernagel, 1969), although, like an isolate from gladiolus described by Snow (1955), it might well be intermediate between bean common mosaic and bean yellow mosaic viruses.
Stability in Sap
The thermal inactivation point (10 min) is usually around 60°C but, depending on virus source, virus strain and environmental conditions, may range between 50 and 65°C; the dilution end-point is usually between l0-3 and l0-4, and sap retains infectivity at room temperature for 1-4 days.
Purification
In sap, much of the virus sediments with the chloroplasts on low speed centrifugation (van der Want, 1954). Partially pure preparations suitable for serological tests have been obtained by one cycle of differential centrifugation followed by low speed centrifugation, clarification with ether, another low speed centrifugation and dialysis against 0.85% NaCl (Bercks, 1959).
Scott (1962) also reported that chloroform-butanol clarification was useful.
Properties of Particles
No data.
Particle Structure
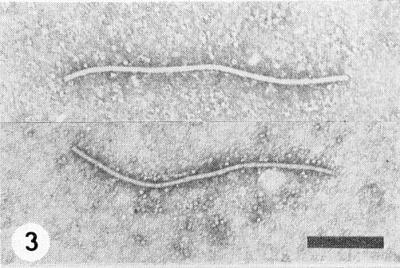
Particles are flexuous filaments (Fig. 3), c. 750 nm long and c. 15 nm wide (Brandes & Quantz, 1955). Zaumeyer & Goth (1964) reported that the particles of the Florida strain were 738 nm long.
Particle Composition
No data.
Relations with Cells and Tissues
Zettler (1969) found cytoplasmic inclusions in epidermal strips, especially from the chlorotic areas of bean leaves infected with a New York isolate, after staining with calcomine orange and luxol brilliant green.
Notes
Bean common mosaic virus can be confused with bean yellow mosaic virus but has a more limited host range. It usually gives distinctive symptoms in Phaseolus but, unlike bean yellow mosaic virus, produces no obvious local reaction in Chenopodium amaranticolor and C. quinoa. In Phaseolus, it causes vein-banding and leaf-rolling somewhat similar to that induced by bean southern mosaic virus, which is also seed-borne. However, the latter virus has a high thermal inactivation point (95°C), has spherical particles easily seen in the electron microscope after negative staining, is not transmitted by aphids, and induces systemic symptoms in bean varieties hypersensitive to bean common mosaic virus.
Acknowledgements
Photographs courtesy of Institute of Phytopathological Research, Wageningen.
Figures
References list for DPV: Bean common mosaic virus (73)
- Ainsworth, Ann. appl. Biol. 27: 218, 1940.
- Beemster & van der Want, Antonie van Leeuwenhoek 17: 15, 1951.
- Bercks, Phytopath. Z. 35: 105, 1959.
- Bercks, Phytopath. Z. 39: 120, 1960.
- Bos, Neth. J. Pl. Path. 76: 8, 1970a.
- Bos, CMI/AAB Descriptions of Plant Viruses 40, 4 pp., 1970b.
- Brandes & Quantz, Naturwissenschaften 42: 588, 1955.
- Cafati, Agricultura téc. 28: 130, 1968.
- Crowley, Aust. J. biol. Sci. 10: 449, 1957.
- Dean & Wilson, Pl. Dis. Reptr 43: 1108, 1959.
- El-Attar, Sabet & Nour-Eldin, J. Bot. Un. Arab. Repub. 7: 77, 1964.
- Frandsen, Z. PflZücht. 31: 381, 1952.
- Grogan & Walker, J. agric. Res. 77: 315, 1948a.
- Grogan & Walker, Phytopathology 38: 489, 1948b.
- Harrison, Tech. Bull. N.Y.St. agric. Exp. Stn 235, 48 pp., 1935.
- Hubbeling, Meded. Inst. plziektenk. Onderz. 83, 80 pp., 1955.
- Hubbeling, Meded. Landb- Hoogesch. OpzoekStns Gent 28: 1025, 1963.
- Inouye, Rev. Pl. Prot. Res., Tokyo 2: 42, 1969.
- Jermoljev & Chod, Ochr. Rost. 2: 145, 1966.
- Kaiser, Danesh, Okhovat & Mossahebi, Pl. Dis. Reptr 52: 687, 1968.
- Kennedy, Day & Eastop, A conspectus of aphids as vectors of plant viruses, London, Commonwealth Institute of Entomology, 1962.
- Matsumoto, Phytopathology 12: 295, 1922.
- Nelson, Tech. Bull. Mich. (St. Coll.) agric. Exp. Stn 118, 71 pp., 1932.
- Pierce, Phytopathology 24: 87, 1934.
- Pierce & Hungerford, Phytopathology 19: 605, 1929.
- Quantz, NachrBl. dt. PflSchutzdienst. Stuttg. 9: 71, 1957.
- Quantz, Phytopath. Z. 31: 319, 1958.
- Quantz, Phytopath. Z. 43: 79, 1961.
- Quantz NachrBl. dt. PflSchutzdienst. Stuttg. 14: 49, 1962.
- Reddick, Deux. Congr. int. Path. Comp. : 363, 1931.
- Reddick & Stewart, Phytopathology 8: 530, 1918.
- Reddick & Stewart, Phytopathology 9: 445, 1919.
- Richards & Burkholder, Phytopathology 33: 1215, 1943.
- Schippers, Acta. bot. neerl. 12: 433, 1963.
- Scott, Phytopathology 52: 166, 1962.
- Silbernagel, Phytopathology 59: 1809, 1969.
- Skotland & Burke, Phytopathology 51: 565, 1961.
- Snow, Phytopathology 45: 696, 1955.
- Snyder, Phytopathology 32: 518, 1942.
- Stewart & Reddick, Phytopathology 7: 61, 1917.
- van der Want, Meded. Inst. plziektenk. Onderz. 85, 84 pp., 1954.
- Zaumeyer & Goth, Phytopathology 53: 490, 1963.
- Zaumeyer & Goth, Phytopathology 54: 1378, 1964.
- Zettler, Diss. Abstr. Sect. B. 27: 1696, 1966.
- Zettler, Phytopathology 59: 1109, 1969.
- Zettler & Wilkinson, Phytopathology 56: 1079, 1966.