Details of DPV and References
DPV NO: 80 October 1971
Family: Secoviridae
Genus: Nepovirus
Species: Cherry leaf roll virus | Acronym: CLRV
There is a more recent description of this virus: DPV 306
Cherry leaf roll virus
R. Cropley East Malling Research Station, Maidstone, Kent, England
J. A. Tomlinson National Vegetable Research Station, Wellesbourne, Warwickshire, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by
Posnette & Cropley (1955) and
Cropley (1961).
- Synonyms
- The following names refer to serologically related strains
- Synonyms
- Elm mosaic virus (Rev. appl. Mycol. 20: 236; 23:
244)
- Golden elderberry virus (Rev. appl. Mycol. 46: 3634d)
- Golden elderberry virus (Rev. appl. Mycol. 46: 3634d)
- A virus with isometric particles about 30 nm in diameter occurring in Europe and North America. It is readily sap-transmissible and has a wide host range. The virus is seed- and pollen-transmitted in several host plants and is reported to be transmitted by nematodes (Xiphinema spp.).
Main Diseases
Causes leaf roll of cherry (Fig. 1), ringspot of Sambucus racemosa, yellow net of S. nigra, mosaic of elm, and stunting, leaf pattern and death of blackberry (P. J. Ormerod, pers. comm.). It frequently occurs in certain rhubarb cultivars, but its effects on rhubarb plants have not been assessed.
Geographical Distribution
Europe and North America.
Host Range and Symptomatology
Natural infection has been detected in cherry, rhubarb, Sambucus spp., American elm, American dogwood (H. E. Waterworth & R. Lawson, pers. comm.) and blackberry. Schmelzer (1966) transmitted a Sambucus isolate to 61 species in 23 dicotyledonous families and to one monocotyledonous species, Commelina coelestris. Hansen & Stace-Smith (in press) reported that another Sambucus isolate infected 44 out of 55 herbaceous species.
Diagnostic species
- Chenopodium amaranticolor and C. quinoa. Chlorotic and
necrotic primary lesions; systemic mottle, distortion and necrosis
(Fig. 4).
- Cucumis sativus (cucumber). Chlorotic primary lesions in cotyledons. In plants grown in glasshouses in winter, distortion and mottling of true leaves, stunting and premature death; in summer, chlorotic rings and lines with little effect on growth.
- Nicotiana tabacum cv. White Burley (tobacco). Primary necrotic spots after 2-3 days; under some conditions concentric necrotic rings develop (Fig. 2). Systemic necrotic and chlorotic rings and lines (Fig. 3); leaves produced later are symptomless.
- N. rustica. Primary chlorotic and necrotic spots and rings; systemic necrotic and chlorotic rings.
- Phaseolus vulgaris cv. The Prince (French bean). Primary necrotic lesions in primary leaves; brown spots and rings; vein necrosis in true leaves; death of apical shoot in winter.
- Cucumis sativus (cucumber). Chlorotic primary lesions in cotyledons. In plants grown in glasshouses in winter, distortion and mottling of true leaves, stunting and premature death; in summer, chlorotic rings and lines with little effect on growth.
- Propagation species
- Nicotiana tabacum or (with cherry isolates at least) Prunus
avium are suitable hosts for maintaining the virus; N. tabacum and
Chenopodium quinoa are useful for propagating the virus for
purification.
- Assay species
- Nicotiana tabacum cv. White Burley is a good primary lesion host.
Strains
Isolates differ in virulence in cherry and in herbaceous plants. The following strains differ serologically (see Relationships) and perhaps in other ways:
Type (cherry) strain of Cropley (1961)
Rhubarb strain of Tomlinson & Walkey (1967)
Elm mosaic strain of Varney & Moore (1952)
Golden elderberry strain of Hansen (1967) and Hansen & Stace-Smith (in press).
Transmission by Vectors
Transmission of cherry isolates by the soil-inhabiting nematodes Xiphinema coxi, X. diversicaudatum and X. vuittenezi has been reported (Fritzsche & Kegler, 1964; Flegg, 1969). The elm mosaic strain was not transmitted by X. americanum (Fulton & Fulton, 1970).
Transmission through Seed
Seed-borne in herbaceous hosts, the proportion of seedlings infected ranging from 0.6 to 100% (Schmelzer, 1966; Lister & Murant, 1967; Tomlinson & Walkey, 1967; Hansen & Stace-Smith, in press). The elm mosaic strain was transmitted through elm pollen and ovules to the seed in controlled pollination experiments, but was not transmitted to the parent tree (Callahan, 1957a, 1957b).
Transmission by Dodder
No transmission by Cuscuta californica, C. campestris or C. subinclusa (Schmelzer, 1966).
Serology
Antiserum titres usually low, between 1/64 and 1/640. The virus produces one band of precipitate in gel double-diffusion tests. During the spring, crude cherry sap can be used in gel-diffusion tests (Cropley, 1960).
Relationships
In plant protection and serological tests, no relationship was found between a cherry isolate of cherry leaf roll virus and arabis mosaic, tomato black ring, raspberry ringspot, tomato ringspot or tobacco ringspot viruses (Cropley, 1961). One-way protection between elm mosaic and tomato ringspot viruses led Varney & Moore (1952) to suspect a relationship between them but serological tests (Fulton & Fulton, 1970) did not confirm this. Jones & Murant (in press) established that elm mosaic virus is a strain of cherry leaf roll virus; they found that isolates of cherry leaf roll virus from cherry, rhubarb, elm (= elm mosaic virus) and elderberry (= golden elderberry virus) were serologically related but not identical. English and Dutch cherry isolates were serologically identical as were Scottish and North American isolates from elderberry. Stefanac (1969) found that a Yugoslavian isolate from elderberry was also serologically distinguishable from cherry and rhubarb isolates.
Stability in Sap
In tobacco sap, cherry and elderberry isolates of the virus lost infectivity after 10 min at 52-55°C, after 5-10 days at 20°C and after dilution between 10-3 and 10-4 (Cropley, 1961; Hansen & Stace-Smith, in press). Sap from cherry buds was more infective than tobacco sap when tested on tobacco (Cropley, 1964).
Purification
The following method is effective (Tomlinson & Walkey, 1967). Homogenize tobacco leaves 10 days after inoculation, in cold 0.1 M, pH 7.5 potassium phosphate buffer, 1.5 ml/g tissue. Squeeze through muslin and freeze for 23 weeks. Thaw, add 0.25 volumes chloroform. Shake 5 min and centrifuge at low speed. Centrifuge clear aqueous phase at high speed, resuspend pellets in 0.05 M phosphate, pH 7.5, for 2 h and clarify by low speed centrifugation. To supernatant fluid add 1.0 volumes 50% saturated ammonium sulphate and store at 20°C for 1 h. Sediment the precipitate by low speed centrifugation and resuspend in 0.05 M phosphate, pH 7.5. Centrifuge at low speed and withdraw clear, infective supernatant fluid. Do all steps at 3°C. Preparations are infective to 10-3.
A similar procedure was used by Hansen & Stace-Smith (in press) for the golden elderberry strain. The method of Stace-Smith (1966) for tomato ringspot virus has also proved useful for the golden elderberry strain (A. T. Jones, pers. comm.) but the elm mosaic strain seems more difficult to purify, even with this method (Fulton & Fulton, 1970).
Properties of Particles
Purified preparations of the golden elderberry strain sediment as two components, T and B, each containing infectious nucleoprotein particles (Hansen & Stace-Smith, in press).
Sedimentation coefficients (s20,w) at infinite dilution
(svedbergs): 114 (T), 132 (B).
A260/A280: 1.77 (T), 1.78 (B).
Particle Structure
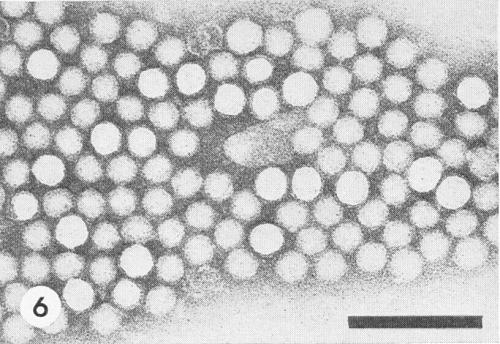
Isometric, approximately 30 nm in diameter, angular in outline (Fig. 6).
Particle Composition
Nucleic acid is RNA; protein M. Wt c. 54,000 (A. T. Jones & M. A. Mayo, pers. comm.).
Relations with Cells and Tissues
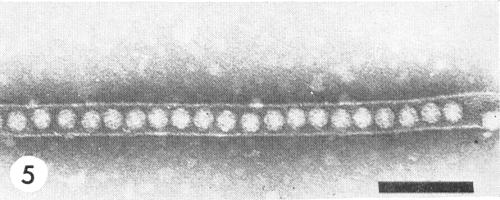
Inclusion bodies consisting of tubules containing single rows of particles (Fig. 5) were observed by electron microscopy in apical meristems, root tips, pollen, ovules and mature seeds, but not in leaf tissue of Nicotiana rustica plants infected with a rhubarb isolate (Walkey & Webb, 1970). However, the virus can be eliminated from rhubarb by meristem-tip culture (Walkey, Fitzpatrick & Woolfitt, 1969).
Notes
Infected cherry trees are stunted, and often die within a few years after infection. Symptoms in cherry are similar to those caused by branch cankers caused by Pseudomonas mors-prunorum or by fungal root pathogens. Although nematode-transmission is reported, it is not established that this is the main mode of spread in the field. In herbaceous test plants cherry leaf roll virus causes symptoms resembling those produced by many nepoviruses and can only be identified with certainty by serological tests.
Acknowledgements
Fig. 1 courtesy of East Malling Research Station; Figs 2-7 courtesy of the National Vegetable Research Station.
Figures
References list for DPV: Cherry leaf roll virus (80)
- Callahan, Phytopathology 47: 5, 1957a.
- Callahan, Diss. Abstr. 17: 1861, 1957b.
- Cropley, Nature, Lond. 188: 875, 1960.
- Cropley, Ann. appl. Biol. 49: 524, 1961.
- Cropley, Ann. appl. Biol. 53: 333, 1964.
- Flegg, Rep. E. Malling Res. Stn for 1968: 155, 1969.
- Fritzsche & Kegler, Naturwissenschaften 51: 299, 1964.
- Fulton & Fulton, Phytopathology 60: 114, 1970.
- Hansen, Phytopathology 57: 813, 1967.
- Hansen & Stace-Smith, Phytopathology (in press).
- Jones & Murant, Ann. appl. Biol. (in press).
- Lister & Murant, Ann. appl. Biol. 59: 49, 1967.
- Posnette & Cropley, Rep. E. Malling Res. Stn for 1954: 126, 1955.
- Schmelzer, Phytopath. Z. 55: 317, 1966.
- Stace-Smith, Virology 29: 240, 1966.
- Stefanac, Acta bot. croat. 28: 373, 1969.
- Tomlinson & Walkey, Ann. appl. Biol. 59: 415, 1967.
- Varney & Moore, Phytopathology 42: 476, 1952.
- Walkey, Fitzpatrick & Woolfitt, J. gen. Virol. 5: 237, 1969.
- Walkey & Webb, J. gen. Virol. 7: 159, 1970.