Details of DPV and References
DPV NO: 83 June 1972
Family: Bromoviridae
Genus: Ilarvirus
Species: Apple mosaic virus | Acronym: ApMV
Apple mosaic virus
R. W. Fulton Department of Plant Pathology, University of Wisconsin, Madison, Wisconsin, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Bradford & Joley (1933), Christoff (1934) and White (1928).
- Selected synonyms
- Apple infectious variegation virus (Rev. appl. Mycol. 12: 636)
- Rose infectious chlorosis virus (Rev. appl. Mycol. 7: 721)
- Rose mosaic virus (Rev. appl. Mycol. 19: 409)
- Plum line pattern virus (Rev. appl. Mycol. 35: 686)
- European plum line pattern virus (Rev. Pl. Path. 49: 1725)
- Rose infectious chlorosis virus (Rev. appl. Mycol. 7: 721)
An isometric virus with particles about 26 nm in diameter. Mechanically transmissible, with difficulty, from young leaves of apple and rose to herbaceous plants. Transmitted readily from numerous herbaceous hosts. The virus is very labile in extracts. It has a wide experimental host range, but no known vector. Distribution world-wide.
Main Diseases
Causes mosaic in apple (Fig. 1), and line-pattern in plum (Gilmer, 1956), although other viruses cause similar symptoms in plum. In rose it causes mosaic symptoms (chlorotic bands and lines) (Fig. 5) which are not readily differentiated from those caused by prunus necrotic ringspot virus (Fulton, 1967). It occurs naturally in hop (Bock, 1967) and in birch (A. R. Gotlieb & J. G. Berbee, pers. comm.) in leaves of which it causes chlorotic lines and rings. It may be transmitted by natural root grafts (Hunter, Chamberlain & Atkinson, 1958). Apple mosaic was one of the first ‘variegations’ shown to be transmissible by grafting. Bradford & Joley (1933) review early records from the 19th century.
Geographical Distribution
World-wide where its hosts are cultivated.
Host Range and Symptomatology
The virus has a wide host range. Experimentally or naturally, it has infected over 65 species in 19 families (Fulton, 1952, 1965 and unpublished; Kristensen & Thomsen, 1963). There is much variation in symptoms with different isolates, at least some of which differ in host range. Mechanically transmissible with difficulty from young leaves of apple and rose to herbaceous plants. Transmitted somewhat more readily from petals or from graft-inoculated Prunus persica and Chaenomeles japonica, and readily from numerous herbaceous hosts.
-
Diagnostic species
- Cucumis sativus
(cucumber). Prominent chlorotic primary lesions on cotyledons (Fig. 2); systemic invasion results in extreme stunting. - Torenia fournieri. Bright yellow mosaic
(Fig. 4).
- Vinca rosea (periwinkle). Chlorotic rings and lines on systemically invaded leaves (Fig. 3). Symptoms become indistinct.
- Vigna sinensis (cowpea). Isolates from rose usually cause chlorotic lines and rings on trifoliolate leaves. Some isolates from apple do not infect this host.
- Malus sylvestris (apple) cv. Lord Lambourne and Jonathan develop a prominent mosaic and are recommended as woody indicators.
- Vinca rosea (periwinkle). Chlorotic rings and lines on systemically invaded leaves (Fig. 3). Symptoms become indistinct.
-
Propagation species
- Vinca rosea
and Chaenomeles japonica are suitable for maintaining cultures. Cucumber cotyledons 4-6 days after inoculation are a suitable source of virus for purification.Assay species
- Most isolates induce distinct necrotic lesions in Cyamopsis tetragonoloba (Fig. 7).
Strains
Strains ranging from mild to virulent in apple are described (Posnette & Cropley, 1956; Kristensen & Thomsen, 1963). Differences also exist among isolates in ability to infect particular herbaceous species.
Transmission by Vectors
No vectors are known.
Transmission through Seed
No evidence was found for seed transmission in apple (Campbell, 1964).
Transmission by Dodder
Not transmitted by Cuscuta campestris, C. gronovii, C. subinclusa (Fulton, 1952; Gilmer, 1958) or C. reflexa (Nagaich & Vashishth, 1963).
Serology
The virus is moderately immunogenic; rabbits receiving twice-weekly intramuscular injections of about 1 mg virus emulsified in Freund’s incomplete adjuvant developed antibody titres of 1/1280 or more after 4 weeks (Fulton, 1967). The virus reacts well in agar diffusion tests; in liquid precipitin tests precipitates are granular.
Relationships
Mild strains of the virus protect against more virulent strains in apple (Posnette & Cropley, 1956). Similarities among apple mosaic, plum line pattern and rose mosaic viruses were demonstrated by interspecific inoculations: inoculum from rose produced line pattern in plum (Gualaccini, 1960), inoculum from plum with line pattern produced mosaic in apple, and inoculum from apple with mosaic produced line pattern in plum (Gilmer, 1956). Serological tests confirmed that apple mosaic, rose mosaic and European plum line pattern diseases are caused by the same or closely related viruses (Fulton, 1968; Seneviratne & Posnette, 1970). Apple mosaic and rose mosaic viruses completely absorb each other's antiserum. These two viruses also cross-react with prunus necrotic ringspot virus antiserum (De Sequeira, 1967); the proportion of common antigen is low, however (Fulton, 1968).
Stability in Sap
The virus is very unstable, losing most of its infectivity in a few minutes in undiluted cucumber sap, and within a few hours in buffer extracts. Infectivity is stabilized, at least for several hours, in buffer containing 0.02 M 2-mercaptoethanol (Fulton, 1968). In stabilized extracts the thermal inactivation point (10 min) is 54°C. Dilution end-point, 2 x 10-3.
Purification
The following method is effective (Fulton, 1967). Homogenize heavily infected cucumber cotyledons cold in 1.5 ml buffer/g tissue. The buffer is 0.02 M phosphate, pH 8.0, and is 0.02 M with respect to 2-mercaptoethanol and sodium diethyldithiocarbamate. After low speed centrifugation, mix the supernatant liquid thoroughly with 0.9 volumes of hydrated calcium phosphate and centrifuge at low speed for 20 min. Sediment the virus from the supernatant liquid by centrifuging 3 hr at 78,000 g. Resuspend pellets in phosphate buffer and centrifuge at low speed for 20 min. Dilute the supernatant liquid with phosphate buffer and repeat the high speed centrifugation and pellet resuspension steps. The virus may be further purified by precipitating host protein with the gammaglobulin portion of host-specific antiserum. Precipitation of host protein with citric acid eliminates the virus.
Properties of Particles
Two kinds of nucleoprotein particles have been reported, with sedimentation coefficients of 88 and 117 S (De Sequeira, 1967). A third type of particle with a sedimentation coefficient intermediate to these becomes discernible after centrifugation for 4-5 hr (Fulton, 1967). The 88 S particles are not infective. Particles are serologically and electrophoretically homogeneous.
A260/A280: c. 1.50.
Particle Structure
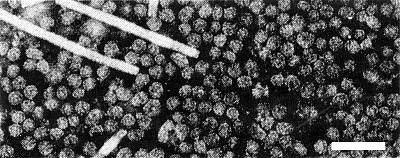
Particles are isometric (Fig. 6). Two sizes have been reported, c. 25 and 29 nm, corresponding to two peaks in density gradient centrifugation (De Sequeira, 1967). Particles disintegrate rapidly in phosphotungstate unless fixed in glutaraldehyde; even fixed particles are easily deformed.
Particle Composition
No information for this virus, but by analogy with prunus necrotic ringspot virus, the particles probably contain l6% RNA.
Relations with Cells and Tissues
Systemic invasion of apple and plum is often slow and erratic (Luckwill, 1954; Posnette & Cropley, 1956; Posnette & Ellenberger, 1957). The virus has been eliminated from cuttings of young plants of both apple and rose by holding them close to 36°C for 3-10 weeks (Posnette & Cropley, 1956; Holmes, 1960).
Notes
Unrelated viruses causing similar symptoms in some of their woody hosts are Tulare apple mosaic and American plum line pattern viruses. The plum line pattern strain of prunus necrotic ringspot virus also causes similar symptoms in plum (Fulton, 1965; Seneviratne & Posnette, 1970). Rose mosaic is not readily differentiated from infection of rose by prunus necrotic ringspot virus, which is common.
Figures
References list for DPV: Apple mosaic virus (83)
- Bock, Ann. appl. Biol. 59: 437, 1967.
- Bradford & Joley, J. agric. Res. 46: 901, 1933.
- Campbell, Rep. agric. hort. Res. Stn Univ. Bristol 1963: 63, 1964.
- Christoff, Phytopath. Z. 7: 521, 1934.
- De Sequeira, Virology 31: 314, 1967.
- Fulton, Phytopathology 42: 413, 1952.
- Fulton, Zast. Bilja 16: 427, 1965.
- Fulton, Phytopathology 57: 1197, 1967.
- Fulton, Phytopathology 58: 635, 1968.
- Gilmer, Phytopathology 46: 127, 1956.
- Gilmer, Phytopathology 48: 432, 1958.
- Gualaccini, Boll. Staz. Patol. veg. Roma 18: 49, 1960.
- Holmes, Pl. Dis. Reptr 44: 46, 1960.
- Hunter, Chamberlain & Atkinson, N. Z. Jl agric. Res. 1: 80, 1958.
- Kristensen & Thomsen, Phytopath. mediterranea 2: 97, 1963.
- Luckwill, Rep. agric. hort. Res. Stn Univ. Bristol 1953: 40, 1954.
- Nagaich & Vashishth, Indian Phytopath. 16: 354, 1963.
- Posnette & Cropley, J. hort. Sci. 31: 119, 1956.
- Posnette & Ellenberger, Ann. appl. Biol. 45: 74, 1957.
- Seneviratne & Posnette, Ann. appl. Biol. 65: 115, 1970.
- White, Pl. Dis. Reptr 13: 33, 1928.