Details of DPV and References
DPV NO: 85 June 1972
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Broccoli necrotic yellows virus | Acronym: BNYV
Broccoli necrotic yellows virus
R. N. Campbell Department of Plant Pathology, University of California, Davis, California 95616, USA
M. T. Lin Department of Plant Pathology, University of California, Davis, California 95616, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Hills & Campbell (1968).
An enveloped RNA-containing virus with bacilliform (275 x 75 nm) particles, often appearing bullet-shaped (266 x 66 nm) with some staining methods. It has a narrow host range and is transmitted by aphids but with difficulty by sap inoculation. Found only in England.
Main Diseases
It is doubtful whether the virus causes an economically important disease. Found occasionally in cauliflower-headed broccoli (late cauliflower) plants showing symptoms of infection with cauliflower mosaic virus (Hills & Campbell, 1968); also found in brussels sprout (Tomlinson & Webb, 1971).
Geographical Distribution
England.
Host Range and Symptomatology
Restricted host range. Brassica oleracea is the only known naturally infected host. Mechanical inoculation to B. oleracea is difficult; infected plants show mild vein-clearing and slight leaf-rolling but later become symptomless (Tomlinson & Webb, 1971). Transmissible by sap inoculation to a few species of Solanaceae including:
-
Diagnostic species
- Datura stramonium.
Chlorotic to necrotic local lesions may appear in about 10 days; systemic mosaic appears in about 18 days (Fig. 1) and often is accompanied by necrosis of fine veins. Occasionally, leaf distortion (Fig. 2) and necrotic rings (Fig. 3) are seen. - Nicotiana glutinosa. Symptom expression erratic. Sometimes local lesions form, occasionally systemic infection results in mosaic symptoms (Fig. 4).
-
Propagation species
- D. stramonium
is suitable for maintaining cultures and as a source of virus for purification.Assay species
- D. stramonium
is the most sensitive host. Local lesions are not dependable, and thus systemic infection is used to assess infectivity.
Strains
No information.
Transmission by Vectors
Myzus persicae and Brevicoryne brassicae did not transmit (Hills & Campbell, 1968; Tomlinson & Webb, 1971), but transmission has now been obtained with B. brassicae (Tomlinson, Webb & Faithfull, 1972).
Transmission through Seed
No seed-transmission was found in D. stramonium (Lin & Campbell, 1972).
Transmission by Dodder
Not tested.
Serology
The virus seems to be poorly immunogenic but a specific antiserum has been obtained (Lin & Campbell, 1972). In double-diffusion tests in agar, virus preparations reacted with antiserum to give three specific bands. The most intense was midway between antigen and antiserum wells; a faint band developed later near the antiserum well and another near the antigen well.
Relationships
Morphologically, broccoli necrotic yellows virus is in the ‘rhabdovirus group’ (sensu Howatson, 1970), which contains at least 11 plant-infecting viruses, as well as viruses infecting vertebrates and invertebrates. Together with lettuce necrotic yellows and plantain A viruses it forms a sub-group of these viruses whose assembly is associated with cytoplasmic membranes and whose shape is longer and narrower than that of viruses in the other sub-group (Hull, 1970). No serological relationships have been reported. Antiserum to broccoli necrotic yellows virus did not react with vesicular stomatitis virus (New Jersey serotype), nor did antisera to lettuce necrotic yellows or barley yellow striate mosaic (Conti, 1969) viruses react with broccoli necrotic yellows or vesicular stomatitis viruses (M. T. Lin, unpublished). Broccoli necrotic yellows virus is similar to lettuce necrotic yellows virus in size and intracellular location but differs in host range, host reaction, geographical distribution and serological activity. These differences may not be of overriding importance, but at present it seems that broccoli necrotic yellows virus is best considered a distinct virus.
Stability in Sap
In Datura stramonium sap, the thermal inactivation point (10 min) is about 50°C, the dilution end-point is between 10-3 and 10-4, and infectivity is retained for 2 days at 4°C or for less than 24 h at 23°C. Infectivity is not stabilized by the addition of 0.1 % mercaptoacetic acid or a combination of 0.1 M glycine and 0.01 M MgCl2. The virus is sensitive to ether and butanol (Lin & Campbell, 1972).
Purification
Lin & Campbell (1972). Grind tissue in 0.2 M potassium phosphate buffer (pH 7.6). Clarify by low-speed centrifugation and charcoal-Celite filtration (McLean & Francki, 1967). Sediment and clarify by one or two cycles of differential centrifugation (25,000 rpm for 20 min in Spinco SW 50.1 rotor and 4000 rpm for 5 min in Sorvall SS-34 rotor), resuspending the pellets obtained at high speed in 0.01 M phosphate buffer (pH 7.6). Purify further by column chromatography on hydroxylapatite and by sucrose density-gradient centrifugation.
Properties of Particles
Sedimentation coefficient (s20,w) is 874 ± 41 S after column chromatography and before density-gradient centrifugation. Buoyant density is 1.183-1.195 g/ml in potassium tartrate (Lin & Campbell, 1972).
Particle Structure
Particles are bacilliform in uranyl acetate (Fig. 5) and measure 275 x 75 nm. In potassium phosphotungstate occasional bacilliform particles (Fig. 6) are seen but most particles are ‘bullet-shaped’ and measure 266 x 66 nm (Fig. 7). The bullet-like shape is regarded as a staining artifact. The particles have an outer envelope 13.5 nm thick with hexagonally arranged subunits; uniformly arranged spikes (8.5 nm long) project beyond the envelope. Within the envelope is a helically coiled filament with a 4.5 nm periodicity; this is thought to be the nucleoprotein. Preparations may contain bag-like rosette structures (Fig. 7) which are thought to be virus membranes remaining after disintegration of the internal structures (Lin & Campbell, 1972).
Particle Composition
Purified virus preparations contain RNA and possibly lipid (Lin & Campbell, 1972). The protein(s) have not been characterized.
Relations with Cells and Tissues
No inclusion bodies are found. Virus particles are enclosed in membrane-bounded sacs in the cytoplasm of parenchyma cells (Fig. 8). These sacs are formed from endoplasmic reticulum; the final stage of virus assembly occurs when the incomplete particles pass through the membrane into the sac lumen (Hills & Campbell, 1968). In virus-containing cells, the mitochondria were swollen and the cristae were scarce (Hills & Campbell, 1968).
Notes
Mechanical transmission to Datura stramonium and symptom expression are erratic in our glasshouses in the summer. This appears to be due to high light intensity and/or high temperatures (21-35°C). To compare broccoli necrotic yellows with other similar viruses see lettuce necrotic yellows virus, potato yellow dwarf virus, and sowthistle yellow vein virus.
Figures
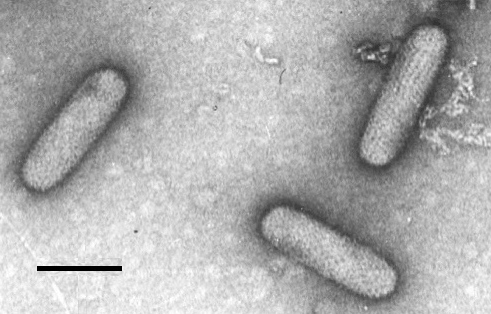
Virus particles from purified preparation in uranyl acetate, showing pattern of outer envelope. Bar represents 100 nm.

Virus particles from purified preparation in 2% potassium phosphotungstate, pH 8.2. Bar represents 100 nm.
References list for DPV: Broccoli necrotic yellows virus (85)
- Conti, Phytopath. Z. 66: 275, 1969.
- Hills & Campbell, J. Ultrastruct. Res. 24: 134, 1968.
- Howatson, Adv. Virus Res. 16: 196, 1970.
- Hull, in Biology of Large RNA Viruses, ed. R. D. Barry & B. W. J. Mahy, Academic Press, N.Y., 1970.
- Lin & Campbell, Virology 48: 30, 1972.
- McLean & Francki, Virology 31: 585, 1967.
- Tomlinson & Webb, Rep. natn. Veg. Res. Stn 1970: 110, 1971.
- Tomlinson, Webb & Faithfull, Ann. appl. Biol. 71: 127, 1972.





