Details of DPV and References
DPV NO: 88 June 1972
Family: Potyviridae
Genus: Potyvirus
Species: Sugarcane mosaic virus | Acronym: SCMV
There is a more recent description of this virus: DPV 342
Sugarcane mosaic virus
T. P. Pirone Department of Plant Pathology, University of Kentucky, Lexington, Kentucky, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Brandes (1919).
Selected synonyms
- Grass mosaic virus (Rev. appl. Mycol. 2: 584)
- Marmor sacchari (Rev. appl. Mycol. 28: 514)
- Saccharum virus 1 (Rev. appl. Mycol. 17: 52)
- Marmor sacchari (Rev. appl. Mycol. 28: 514)
-
Serologically related viruses include:
- Maize dwarf mosaic virus (Rev. appl. Mycol. 44, 3333)
- Abaca mosaic virus (Rev. appl. Mycol. 42: 687)
- Sorghum red stripe virus (Rev. app1. Mycol. 37: 232)
- Maize mosaic virus (Rev. app1. Mycol. 42: I5)
- Abaca mosaic virus (Rev. appl. Mycol. 42: 687)
-
A virus with filamentous flexuous particles about 750 nm long. It infects numerous species in the Gramineae, is transmitted in the non-persistent manner by aphids and is transmissible by sap inoculation.
Main Diseases
Causes mosaic diseases in sugarcane, maize, sorghum and other graminaceous plants.
Geographical Distribution
Occurs in many parts of the world where susceptible species are grown.
Host Range and Symptomatology
Host range is restricted to members of the Gramineae. Strains of the virus have been shown to infect, in addition to sugarcane, varieties of such cultivated crops as maize, sorghum and millet (Brandes & Klaphaak, 1923), wheat, barley, rye and rice (Abbott & Tippett, 1964), as well as numerous other cultivated and wild grasses.
-
Diagnostic species
- Saccharum
spp. (sugarcane). Mosaic and/or striping depending on cultivar and virus strain (Fig. 1, Fig. 2). - Zea mays (maize). Seedlings of most varieties of sweet corn are readily
infected with the
various strains. A mosaic symptom is produced.
- Sorghum bicolor (sorghum). Sart and several other varieties are readily infected with most strains. Mosaic symptoms are produced initially; some strains later cause necrosis and associated leaf reddening (Fig. 6).
-
Propagation species
- Sorghum bicolor
cvs. Sart and Rio are good sources for maintenance and purification of most strains. Zea mays (sweet corn cultivars) yields less virus but is preferable for strains which cause necrosis in sorghum (Gillaspie, 1971).Assay species
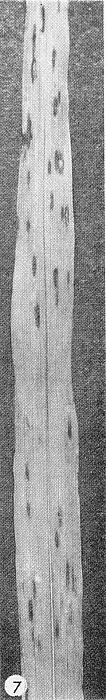
- Strain E of the virus produces necrotic local lesions in Sorghum bicolor cv. Atlas (Fig. 7) (Dean, 1970). Other sorghum varieties such as Sart are good systemic assay hosts.
Strains
The following are serologically related and are considered by some to be strains of sugarcane mosaic virus (see Relationships):
Sugarcane mosaic strains (Brandes, 1919). Cause mosaic in sugarcane. Rarely, if ever, infect Johnson grass (Sorghum halepense). Strains A, B, D, E and F may be differentiated on sugarcane variety C.P.31-294, strain C on Co.281, and strains A and H on C.P.31-588 (Abbott & Tippett, 1966). Strain I may be differentiated on Rio sorghum (Tippett & Abbott, 1968). Other isolates that differ from the strains described from the USA have been obtained from sugarcane in other parts of the world (Abbott & Stokes, 1966).
Maize dwarf mosaic (Williams & Alexander, 1965). Causes mosaic (Fig. 4) and, occasionally, reddening and dwarfing in maize. Strain A infects Johnson grass (Fig. 3) but less readily infects sugarcane, in which it causes slight symptoms or none (Dale & Anzalone, 1965; Gillaspie, 1967). Strain B does not infect Johnson grass or the sugarcane varieties C.P.31-294 and C.P.31-588 (Snazelle, Bancroft & Ullstrup, 1971).
Sorghum red stripe (Grancini, 1957; Lovisolo, 1957). Causes mosaic, followed by red striping and necrosis in sorghum. The virus also infects maize and Johnson grass (Lovisolo, 1957).
European maize mosaic (Panjan, 1960). Has properties similar to maize dwarf mosaic strain A (Tosic & Simova, 1967).
Abaca mosaic virus (Eloja & Tinsley, 1963). Causes a mosaic in abaca (Musa textilis). Its host range includes monocotyledons outside the Gramineae (Eloja & Tinsley, 1963).
Transmission by Vectors
Transmitted by Dactynotus ambrosiae, Hysteroneura setariae, Rhopalosiphum maidis, Toxoptera graminum and a number of other aphid species in the non-persistent manner (Kennedy, Day & Eastop, 1962). The virus is more readily transmitted to or from hosts such as maize and sorghum than to or from sugarcane.
Transmission through Seed
Not reported for the true sugarcane mosaic strains. Transmission of the maize dwarf mosaic strain in maize seed has been reported (Shepherd & Holdeman, 1965).
Transmission by Dodder
Not reported.
Serology
Numerous antisera have been prepared; the titres are usually about 1/256. They give flagellar type precipitates in tests with intact virus in liquid. Immunodiffusion tests may be done in agar gels using purified virus treated with ethanolamine at pH 10.5 as antigen (Bond & Pirone, 1971).
Relationships
Sugarcane mosaic virus belongs to the potato virus Y (potyvirus) group and may be distantly related to several other viruses in this group (Brandes & Bercks, 1965).
Maize dwarf mosaic virus, strains A and B, are serologically related to each other (Gordon & Williams, 1970; Snazelle et al., 1971), and to sugarcane mosaic virus strains (Shepherd, 1965; Wagner & Dale, 1966; Gordon & Williams, 1970; Bond & Pirone, 1971; Snazelle et al., 1971).
Viruses isolated from sorghum in South Africa were closely serologically related to sugarcane mosaic (Von Wechmar & Hahn, 1967). Viruses from maize and Johnson grass in Australia were distantly related to sugarcane mosaic (Taylor & Pares, 1968). Particles of the viruses from South Africa and Australia had greater modal lengths than the sugarcane mosaic isolates used for serological comparisons.
Sorghum red stripe and abaca mosaic viruses are also serologically related to sugarcane mosaic virus (Dijkstra & Grancini, 1960; Eloja & Tinsley, 1963).
Stability in Sap
In sorghum sap, the thermal inactivation point is about 56°C; dilution end-point is 10-2 to 10-4 , depending on the strain. Longevity in vitro is 1-2 days at 20°C. Similar values have been obtained for the maize dwarf mosaic virus isolates (Williams & Alexander, 1965; Shepherd, 1965).
Purification
Bond & Pirone (1971). This procedure may be used for sugarcane mosaic virus strains and also for maize mosaic virus, strain A. Homogenize each 300 g of minced tissue in 300 ml water containing 0.3% ascorbic acid, 0.01 M sodium diethyldithiocarbamate and 0.3% 2-mercaptoethanol. Use each 300 ml of extracting fluid to extract three successive 100 g amounts of leaf material, removing the pulp and retaining the liquid each time. Strain through cheesecloth and homogenize the filtrate with an equal volume of chloroform. Centrifuge at low speed and then at high speed, resuspend the high speed pellets in 0.1 M borate, 0.01 M EDTA, pH 8.2. The pellets should be pooled and resuspended in a minimum volume, because over 90% of the infectivity is lost if a second high speed centrifugation is done. After a second low speed centrifugation, float the supernatant fluid on 10-40% sucrose gradients in 0.1 M borate, 0.01 M EDTA, pH 8.2, and centrifuge for 2 h at 90,000 g. The virus forms a single light-scattering zone.
A very similar procedure, in which the sucrose for density gradients was dissolved in 0.01 M sodium citrate, was used for purifying maize dwarf mosaic virus strains A and B as well as sugarcane mosaic virus strains (Snazelle et al., 1971).
Preparations may be further purified by zone electrophoresis in sucrose gradients (Von Wechmar & Hahn, 1967) or by equilibrium centrifugation in sucrose gradients (Gillaspie, 1971).
Properties of Particles
A sedimentation coefficient of 176±5 S has been calculated for strain B of sugarcane
mosaic virus; the buoyant density in CsCl for strain D is 1.3327
(Tosic & Ford, 1972).
Sedimentation coefficients (svedbergs) of 168±6 (Shepherd, 1965), 155±3 (Bancroft et al., 1966), 148±2 (Sehgal, 1968) and 160-162 (Jones & Tolin, l972), and a buoyant density in CsCl of 1.3245 (Sehgal & Jean, 1970) have been reported for maize dwarf mosaic virus.
Particle Structure
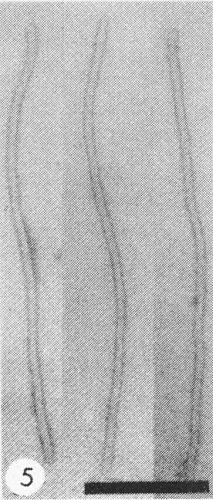
Particles are flexuous filaments about 750 nm long and 13 nm in diameter (Fig. 5).
Particle Composition
Not determined.
Relations with Cells and Tissues
These are similar in maize infected with sugarcane mosaic virus strain H or with maize dwarf mosaic virus strains A or B. Pinwheel inclusions are found in systemically infected tissues. Membrane bound micro-inclusion bodies occur between the plasmalemma and cell wall. Chloroplasts may be swollen (Krass & Ford, 1969).
Notes
There is a high degree of correlation between the presence of maize dwarf mosaic virus strain A in maize fields and the presence of Johnson grass, a perennating source of the virus. Ecological aspects of maize dwarf mosaic virus strain B are not well understood, because no overwintering host has been found. There is still some confusion about the severity of symptoms caused in field-grown maize in USA by maize dwarf mosaic virus strain A. Johnson grass is also a source of the causal agent of corn stunt, and double infections by this and maize dwarf mosaic virus strain A doubtless often occur. Maize grown adjacent to sugarcane may become infected with sugarcane mosaic virus strains, but is seldom severely damaged.
Figures

Sugarcane variety C.P.31-294 systemically infected by sugarcane mosaic virus strain B. (Courtesy A. G. Gillaspie.)

Sugarcane variety C.P.31-294 systemically infected by sugarcane mosaic virus strain D. (Courtesy A. G. Gillaspie.)
References list for DPV: Sugarcane mosaic virus (88)
- Abbott & Stokes, Sug. Azúc 61: 27, 1966.
- Abbott & Tippett, Pl. Dis. Reptr 48: 443, 1964.
- Abbott & Tippett, Tech. Bull. U.S. Dep. Agric. 1340, 25 pp., 1966.
- Bancroft, Ullstrup, Messieha, Bracker & Snazelle, Phytopathology 56: 474, 1966.
- Bond & Pirone, Phytopath. Z. 71: 56, 1971.
- Brandes, Tech. Bull. U.S. Dep. Agric. 829, 26 pp., 1919.
- Brandes & Bercks, Adv. Virus Res. 11: 1, 1965.
- Brandes & Klaphaak, J. agric. Res. 24: 247, 1923.
- Dale & Anzalone, Pl. Dis. Reptr 49: 757, 1965.
- Dean, Phytopathology 60: 569, 1970.
- Dijkstra & Grancini, Tijdschr. PlZiekt. 66: 295, 1960.
- Eloja & Tinsley, Ann. appl. Biol. 51: 253, 1963.
- Gillaspie, Pl. Dis. Reptr 51: 761, 1967.
- Gillaspie, Proc. Int. Soc. Sug. Cane Technol. (in press), 1971.
- Gordon & Williams, Phytopathology 60: 1293, 1970.
- Grancini, Maidica 2: 83, 1957.
- Jones & Tolin, Phytopathology 62: (in press), 1972.
- Kennedy, Day & Eastop, A conspectus of aphids as vectors of plant viruses, London, Commonwealth Institute of Entomology, 1962.
- Krass & Ford, Phytopathology 59: 431, 1969.
- Lovisolo, Boll. Staz. Patol. veg. Roma (1956): 14: 261, 1957.
- Panjan, Zast. Bilja 62: 3, 1960.
- Sehgal, Phytopath. Z. 62: 232, 1968.
- Sehgal & Jean, Phytopathology 60: 189, 1970.
- Shepherd, Phytopathology 55: 1250, 1965.
- Shepherd & Holdeman, Pl. Dis. Reptr 49: 468, 1965.
- Snazelle, Bancroft & Ullstrup, Phytopathology 61: 1059, 1971.
- Taylor & Pares, Aust. J. agric. Res. 19: 767, 1968.
- Tippett & Abbott, Pl. Dis. Reptr 52: 449, 1968.
- Tosic & Ford, Phytopathology 62 (in press), 1972.
- Tosic & Simova, Arch. poljopr. Nauke Teh. 20: 92, 1967.
- Von Wechmar & Hahn, S. Afr. J. agric. Sci. 10: 241, 1967.
- Wagner & Dale, Phytopathology 56: 1422, 1966.
- Williams & Alexander, Phytopathology 55: 802, 1965.