Details of DPV and References
DPV NO: 93 June 1972
Family: Potyviridae
Genus: Potyvirus
Species: Soybean mosaic virus | Acronym: SMV
Soybean mosaic virus
L. Bos Institute of Phytopathological Research, Wageningen, The Netherlands
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Gardner & Kendrick (1921).
- Selected synonyms
- Soybean virus 1 (Rev. appl. Mycol. 15: 418)
- Soja virus 1 (Rev. appl. Mycol. 17: 52)
- Soja virus 1 (Rev. appl. Mycol. 17: 52)
- A virus with flexuous particles c. 750 nm long, transmitted readily by inoculation of sap, by many aphid species in the non-persistent manner, and through seed of soybean (Glycine max), its only known natural host. The virus is probably common wherever this crop is grown.
Main Diseases
Causes mosaic of soybean.
Geographical Distribution
In most countries where soybeans have been tested for viruses.
Host Range and Symptomatology
Long thought to be restricted to soybean, even when transmitted experimentally, but now known to be transmissible to about 30 plant species, in about 10 of these causing only local reactions or none at all (Quantz, 1961; Galvez, 1963; De Vasconcelos, 1964). Chenopodium quinoa and C. album are the only non-legume hosts. Readily transmissible by inoculation of sap or by aphid vectors to the following:
- Diagnostic species
- Glycine max
(soybean). Most varieties develop transient systemic vein-clearing followed by a rolling or distorting mosaic in the younger leaves with dark green, later puffed areas along the main veins and chlorosis between the dark-coloured areas (Fig. 1); plants are usually slightly stunted, pods are fewer, sometimes malformed, glabrous and seedless. Some varieties develop progressive necrosis (Gardner & Kendrick, 1921). Primary leaves of plants grown from infected seed may show mottling and downward curling (Fig. 2). In Japan only 27 cultivars out of 110 tested (Koshimizu & Iizuka, 1963) were resistant or immune. Symptoms are more severe at 18-20°C than at 27-30°C (Conover, 1948; Walters, 1963). - Phaseolus vulgaris (French bean). Latent local infection in some
cultivars, followed by latent systemic infection in a few of these. Systemic
symptoms, somewhat reminiscent of bean common mosaic virus,
are produced only
in Double White Princess (Quantz, 1961; Galvez, 1963; Debrot & De Rojas,
1967).
- Phaseolus lathyroides. Systemic chlorotic mottling or mosaic and leaf curling (Quantz, 1961; Galvez, 1963).
- Chenopodium album. Local lesions, but virus could not be recovered from them (Galvez, 1963).
- Chenopodium quinoa. Weak and diffuse chlorotic local lesions (Quantz, 1961).
- Phaseolus lathyroides. Systemic chlorotic mottling or mosaic and leaf curling (Quantz, 1961; Galvez, 1963).
- Propagation species
- Glycine max
(soybean). Various cultivars have been used. Lincoln plants were harvested 15 days after inoculation (Galvez, 1963). In Bansei, virus infectivity was highest in inoculated leaves 25 days after inoculation and decreased sharply after 31 days; in systemically infected leaves infectivity was correlated with the intensity of mosaic symptoms and remained high from 3 to at least 6 weeks after inoculation (Quiniones & Dunleavy, 1970).- Assay species
- Glycine max
(soybean). Most cultivars react with characteristic systemic symptoms (Fig. 1). - Phaseolus vulgaris (French bean). Necrotic local lesions develop on
inoculated attached primary leaves of Kentucky Wonder waxpole bean (Ross, 1967).
Eight varieties including Processor and Topcrop, produce necrotic local lesions
when detached leaves are incubated on moist filter paper in closed Petri dishes
under artificial light at 30-32°C (Fig. 3; Quantz, 1961).
- Other local-lesion hosts sometimes used are Cyamopsis tetragonoloba, Dolichos biflorus and D. lablab (Dunleavy, Quiniones & Krass, 1970; Quiniones & Dunleavy, 1970; Ross, 1967).
Strains
Some minor strains have been distinguished on the basis of differences in pathogenicity and virulence towards soybean selections, Lespedeza stipulacea and Japanese soybean cultivars (Quiniones & Dunleavy, 1970; Ross, 1968, 1969).
Transmission by Vectors
Some 16 aphid species, including Acyrthosiphon pisum, Aphis fabae and Myzus persicae, have been reported to transmit the virus in the non-persistent manner (Heinze & Köhler, 1940; Conover, 1948; Koshimizu & Iizuka, 1963; De Vasconcelos, 1964).
Transmission through Seed
Common and probably the most important source of initial crop infection. Up to 30% or sometimes more of the seeds of diseased plants are infected, depending on cultivar and duration of infection before flowering (Gardner & Kendrick, 1921; Kendrick & Gardner, 1924; Koshimizu & Iizuka, 1963). Plants infected after flowering do not produce infected seeds. The virus is present in seed-coat and embryo, and green seeds contain more virus than mature ones (Koshimizu & Iizuka, 1963). Seed-coat mottling is stimulated by virus infection, hut there is no direct correlation between this mottling and the presence of virus in particular seeds (Koshimizu & Iizuka, 1963; Kennedy & Cooper, 1967; Ross, 1970). Distribution of infected seeds in pods and on plants is erratic (De Vasconcelos, 1964).
Transmission by Dodder
Not reported.
Serology
So far little information is available. Antisera prepared by Ross (1967) and Quiniones & Dunleavy (1970) using purified virus had a titre of 1/2048 when tested against purified antigen. The titre of Ross’s antiserum was 1/4096 when tested with virus-containing clarified sap. Microprecipitin tests were used because intact virus particles do not diffuse in agar gel.
Relationships
Working with two strains, Quiniones & Dunleavy (1970) found homologous antiserum titres of 1/2048 and titres of 1/256 after cross absorption with heterologous antigens. Bercks (quoted by Quantz, 1961) found serological relationships between soybean mosaic, bean yellow mosaic and bean common mosaic viruses, but this preliminary finding has not been confirmed.
In particle morphology and size, mode of transmission and other biological and biophysical properties, soybean mosaic virus resembles members of the potato virus Y group (potyviruses), especially those closely related to bean yellow mosaic virus (Bos, 1970b). This is particularly true of the seed-transmitted viruses of bean common mosaic (Bos, 1971), cowpea aphid-borne mosaic (Lovisolo & Conti, 1966) and pea leafroll mosaic (Bos, 1970a), and the incompletely described viruses of adzuki bean (Phaseolus angularis) mosaic (Matsumoto, 1922), asparagus bean (Vigna sesquipedalis) mosaic (Snyder, 1942; lnouye, 1969) and mung bean (Phaseolus aureus) mosaic (Kaiser et al., 1968). They all seem to have a rather limited natural host range, possibly because seed transmission makes alternative hosts superfluous.
In cross-protection tests, Quantz (1961) found that soybean mosaic and bean yellow mosaic viruses produced a striking but varying protection against bean common mosaic virus. Similarly, the number of local lesions produced by soybean mosaic and bean common mosaic viruses in detached Topcrop bean leaves in Petri dishes was considerably decreased when the leaves were systemically infected with bean yellow mosaic virus. Quantz (1961) also found that using detached leaves of 23 bean cultivars, the same cultivars were hypersensitive to both soybean mosaic virus and bean common mosaic virus. However, of ten other varieties systemically susceptible to bean common mosaic virus, only one contracted systemic infection by the soybean virus. Moreover, soybean was found to be only locally susceptible to bean common mosaic virus.
Stability in Sap
The thermal inactivation point (10 min) is usually 55-60°C, but sometimes a few degrees higher; the dilution end-point is usually around 10-3 but values of 10-5-10-6 have also been published; sap commonly loses infectivity after 2-3 days at room temperature but sometimes retains it as long as 4 days. In expressed sap the virus was most stable at pH 6.0 (Galvez, 1963).
Purification
Like other viruses of the potato virus Y group, soybean mosaic virus easily aggregates and sediments during initial low-speed centrifugation. Thus much virus is lost when organic solvents are used to denature colloidal plant constituents (Galvez, 1963; Quiniones & Dunleavy, 1970). For good results, according to Ross (1967), grind each 100 g cooled leaves in 200 ml cold 0.5 M sodium citrate buffer containing 1% mercaptoethanol, and strain the extract through cheesecloth. Slowly add n-butanol (7 ml/100 ml extract) while slowly stirring. Refrigerate overnight. Clarify and sediment by two cycles of differential centrifugation, resuspending the sediments obtained at high speed in borate buffer (0.01 M, pH 8.3). Then further purify by centrifugation in sucrose density gradients.
Properties of Particles
No data.
Particle Structure
Particles are flexuous filaments (Fig. 5) c. 746 nm long (Quantz, 1961; De Vasconcelos 1964), sometimes shorter in purified preparations.
Particle Composition
Nucleic acid content (based on A260/A280) is 6-7% (Ross, 1967).
Relations with Cells and Tissues
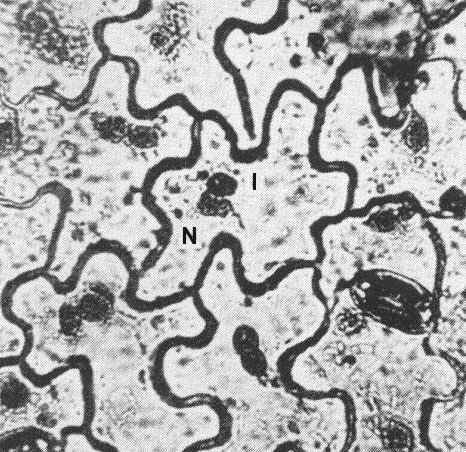
Koshimizu & Iizuka (1963) and Quiniones & Dunleavy (1970) found a single amorphous globular inclusion in each epidermis cell of young soybean plants (Fig. 4). In ultrathin sections of soybean, Dunleavy et al. (1970) found circular membrane-bound and pinwheel micro-inclusions.
Notes
Soybean mosaic virus differs from bean yellow mosaic and bean common mosaic viruses in that (i) it fails to cause systemic symptoms in Phaseolus vulgaris cultivars (excepting Double White Princess), (ii) bean common mosaic virus does not infect soybean systemically, and (iii) bean yellow mosaic virus infects Vicia faba.
Figures

(Right) soybean seedling, cv. Kimusume, grown from infected seed; (left) healthy seedling (after Koshimizu & Iizuka, 1963).

Phaseolus vulgaris, cv. Furore, detached leaf with necrotic local lesions produced at 32°C. (Photo courtesy Dr L. Quantz.)
References list for DPV: Soybean mosaic virus (93)
- Bos, Neth. J. Pl. Path. 76: 8, 1970a.
- Bos, CMI/AAB Descriptions of Plant Viruses 40, 4 pp., 1970b.
- Bos, CMI/AAB Descriptions of Plant Viruses 73, 4 pp., 1971.
- Conover, Phytopathology 38: 724, 1948.
- Debrot & De Rojas, Agronomia trop. 17: 75, 1967.
- De Vasconcelos, Anais. Inst. sup. Agron. Univ. téc. Lisb. 26: 181, 1964.
- Dunleavy, Quiniones & Krass, Phytopathology 60: 883, 1970.
- Galvez, Phytopathology 53: 388, 1963.
- Gardner & Kendrick, J. agric. Res. 22: 111, 1921.
- Heinze & Köhler, Phytopath. Z. 13: 207, 1940.
- Inouye, Rev. Pl. Prot. Res., Tokyo 2: 42, 1969.
- Kaiser, Danesh, Okhovat & Mossahebi, Pl. Dis. Reptr 52: 687, 1968.
- Kendrick & Gardner, J. agric. Res. 27: 91, 1924.
- Kennedy & Cooper, Phytopathology 57: 35, 1967.
- Koshimizu & Iizuka, Bull. Tohoku natn. agric. exp. Stn 27: 1, 1963.
- Lovisolo & Conti, Neth. J. Pl. Path. 72: 265, 1966.
- Matsumoto, Phytopathology 12: 295, 1922.
- Quantz, Phytopath. Z. 43: 79, 1961.
- Quiniones & Dunleavy, Pl. Dis. Reptr 54: 301, 1970.
- Ross, Phytopathology 57: 465, 1967.
- Ross, Pl. Dis. Reptr 52: 344, 1968.
- Ross, Phytopathology 59: 829, 1969.
- Ross, Phytopathology 60: 1798, 1970.
- Snyder, Phytopathology 32: 518, 1942.
- Walters, Pl. Dis. Reptr 47: 726, 1963.