Details of DPV and References
DPV NO: 96 October 1972
Family: Betaflexiviridae
Genus: Carlavirus
Species: Lily symptomless virus | Acronym: LSLV
Lily symptomless virus
T. C. Allen Department of Botany and Plant Pathology, Oregon State University, Corvallis, Oregon 97331, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Brierley & Smith (1944) and Allen & McWhorter (1966).
- Selected synonyms
- Lily curl stripe virus (Rev. appl. Mycol. 46, 12v)
- Lily virus (Allen, 1971)
- Marmor mite (Rev. appl. Mycol. 28: 514)
- Lily virus (Allen, 1971)
- A virus with elongated slightly flexuous filaments c. 640 nm long and 18 nm in diameter, restricted to members of the Liliaceae. Probably transmitted by Myzus persicae in a non-persistent manner; also transmissible by leaf unions and by inoculation of sap. Widely distributed in commercial stocks of lilies.
Main Diseases
Causes curl-stripe (and basal stripe) in lilies under some environmental conditions. Causes necrotic fleck disease in lilies when associated with cucumber mosaic virus. Causes brown rings in bulbs of Lilium mid-century hybrids and streak mottle on leaves of L. speciosum varieties when associated with tulip breaking virus (Asjes, de Vos & van Slogteren, 1973).
Geographical Distribution
Probably occurs wherever lilies are grown.
Host Range and Symptomatology
Host range is restricted to the Liliaceae. All host species are infected systemically but they differ in susceptibility. Sap-transmissible. Also transmissible by leaf unions (McWhorter & Allen, 1964).
- Diagnostic species
- Lilium longiflorum
seedlings previously infected with cucumber mosaic virus develop necrotic flecks. Characteristic flecks of various sizes occur about 3 weeks after inoculation. They are parallel to the leaf veins, and are chlorotic at first, later becoming grey-brown and necrotic (Brierley & Smith, 1944) (Fig. 1). - L. longiflorum seedlings inoculated with lily symptomless virus alone
and grown at temperatures less than 15.5°C develop ‘curl-stripe’
(characteristic white striping and extreme twisting of leaves) about 60 to 90
days after inoculation (McWhorter & Allen, 1967) (Fig. 2).
- Propagation species
- L. longiflorum
‘Ace’ is suitable for maintaining cultures and is a good source of virus for purification.- Assay species
- L. longiflorum
previously inoculated with cucumber mosaic virus, Tulipa gesneriana cvs. Clara Butt and Rose Copland. Dark streaks that are more distinct on the outside than on the inside occur along the veins of the petals of Rose Copland. This differs from the typical colour breaking incited by tulip breaking virus (van Slogteren, personal communication). All are systemic assay hosts.
Strains
None reported.
Transmission by Vectors
A virus with particles about 650 nm long, which was probably lily symptomless virus, was transmitted in the non-persistent manner by the aphid Myzus persicae (Mowat & Stefanac, 1972). However, Brierley & Smith (1944) reported persistent transmission by Aphis gossypii (see Notes).
Transmission by Dodder
No information.
Serology
Antiserum from a rabbit injected intravenously with clarified sap from infected lilies had a titre of 1/1280 in tube precipitin tests (van Slogteren & de Vos, 1966). A rabbit receiving three intravenous injections at weekly intervals followed by two intramuscular injections of partially purified virus from lilies with curl-stripe symptoms developed an antiserum titre of 1/1024 (T. C. Allen, unpublished). Tube or micro-precipitin tests in which antisera form flocculent precipitates, are useful with clarified sap or partially purified virus.
Relationships
Properties and particle morphology place the virus in the carlavirus group. The virus reacted positively with antisera prepared against chrysanthemum B, potato M, potato S, carnation latent and passiflora latent viruses, Antiserum to lily symptomless virus reacted with partially purified (about 10 times concentrated) suspensions of chrysanthemum B, potato M and potato S viruses (van Slogteren & de Vos, personal communication).
Stability in Sap
No information.
Purification
Two methods have been described:
1. (Civerolo, Semancik & Weathers, 1968). Homogenize frozen leaf tissue 1:2 (w/v) in 0.25 M potassium phosphate buffer, pH 7.5, containing 0.002 M MgSO4, 0.1% thioglycollic acid, and 10-15 ml 1% bentonite solution/100 ml of buffer solution. Express homogenate through cheesecloth and centrifuge at 2000 g for 5 min. Add additional 1% bentonite solution at 1-5 ml/100 ml extract and clarify the suspension by low speed centrifugation until the supernatant fluid is straw- coloured. Centrifuge the supernatant fluid, first at 9600 g for 10 min then at 94,000 g for 2 hr. Resuspend the final pellets in 0.1 M phosphate buffer, pH 7.0. Perform all operations at 4°C. When centrifuged in sucrose density gradients, the virus gives a single light-scattering band.
2. (C. J. Asjes, unpublished). Homogenize leaves in 0.067 M phosphate buffer, pH 7.2, +0.1% thioglycollic acid, squeeze through cheesecloth, and freeze the extract at -20°C for one day or longer. Thaw overnight, add an equal volume of chloroform. Stir occasionally for 30 min and centrifuge for 10 min at 1000 g. The resulting extract is ultracentrifuged for 2 hr at 90,000 g. The pellet is resuspended in phosphate buffer and centrifuged for 10 min at 1000 g.
Properties of Particles
Sedimentation coefficient (s20,w) at infinite dilution: about 172 S.
A260/A280: 1.20-1.43 (Civerolo et al., 1968).
Particle Structure
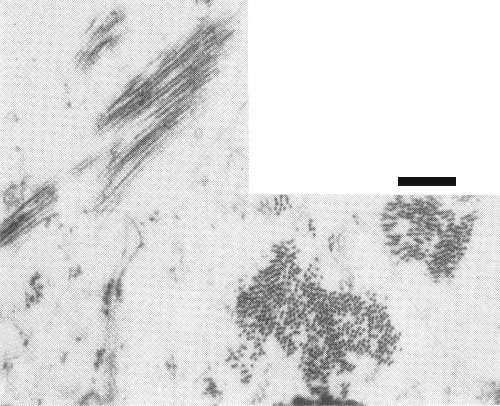
Particles are elongated, slightly flexuous rods, c. 640 nm long and 17-18 nm in diameter (Fig. 3, Fig. 4). Particle centre is densely stained (Fig. 5). When mounted in 2% sodium phosphotungstate or 1% uranyl acetate for electron microscopy, the particles contrast well; the electron microscope is therefore a useful aid in diagnosing infection with this virus (Civerolo et al., 1968; Lyons & Allen, 1969).
Particle Composition
Nucleic acid is about 8.3% of particle weight (Civerolo, 1967).
Relations with Cells and Tissues
All tissues are infected except, possibly, meristematic regions. Leaf tissues tend to yield more particles than root or stem tissue. Thin section studies have shown that the virus-like particles associated with the disease occur in the cytoplasm (Fig. 6). No pinwheels or inclusion bodies have been observed (Allen & Lyons, 1969; Lyons & Allen, 1969).
Notes
The name lily symptomless virus is perhaps unfortunate because other lily viruses can sometimes infect lilies symptomlessly and lily symptomless virus itself causes symptoms in lilies under some environmental conditions. The 650 nm virus described here is thought to be the lily symptomless virus described by Brierley & Smith (1944) because it causes necrotic fleck disease in Lilium longiflorum in mixed infections with cucumber mosaic virus. The main cause for doubt is that, whereas the virus described by Brierley & Smith was transmitted in a persistent manner by Aphis gossypii, evidence (Mowat & Stefanac, 1972) suggests that the 650 nm virus, like other members of the carlavirus group, is transmitted in the non-persistent manner by aphids. However, at present, it seems best to retain the name lily symptomless virus.
Another virus, lily mottle virus (= tulip breaking virus), often occurs in necrotic fleck-diseased L. longiflorum but apparently is not involved in the disease (Brierley & Smith, 1944). In sections of plants showing mottle symptoms as well as necrotic fleck, 750 nm virus particles and pinwheel inclusions characteristic of tulip breaking virus can be found (Allen, 1971).
Figures

Necrotic fleck symptoms in Lilium longiflorum caused by mixed infection with lily symptomless virus and cucumber mosaic virus (courtesy F. P. McWhorter).

Curl-stripe symptoms in L. longiflorum caused by infection with lily symptomless virus alone (courtesy F. P. McWhorter).

Metal-shadowed preparation of virus particles extracted from leaves of Ace lily with curl-stripe symptoms. Bar represents 200 nm.

Virus particle in sodium phosphotungstate-treated preparation from leaves of Ace lily with necrotic fleck disease. Bar represents 100 nm.
References list for DPV: Lily symptomless virus (96)
- Allen, Lily Yb., N. Am. Lily Soc. 24: 29, 1971.
- Allen & McWhorter, Phytopathology 56: 869, 1966.
- Allen & Lyons, Phytopathology 59: 1318, 1969.
- Brierley & Smith, Phytopathology 34: 529, 1944.
- Asjes, de Vos & van Slogteren, Neth. J. Pl. Path. 79: 23, 1973.
- Civerolo, Doctoral Dissertation, Univ. Calif., Riverside, 1967.
- Civerolo, Semancik & Weathers, Phytopathology 58: 1481, 1968.
- Lyons & Allen, J. Ultrastruct. Res. 27: 198, 1969.
- McWhorter & Allen, Nature, Lond. 204: 604, 1964.
- McWhorter & Allen, in Easter Lilies, p. 111, Eds. D. C. Kiplinger & R. W. Langhans, The New York and Ohio Lily Schools, 1967.
- Mowat & Stefanac, Lily Yb., N. Am. Lily Soc. 33: 251, 1972.
- van Slogteren & de Vos, in Viruses of Plants, p. 320, Eds. A. B. R. Beemster & Jeanne Dijkstra, North-Holland, Amsterdam, 1966.