Details of DPV and References
DPV NO: 61 October 1971
Family: Betaflexiviridae
Genus: Carlavirus
Species: Carnation latent virus | Acronym: CLV
Carnation latent virus
C. Wetter Botanisches Institut, Universität des Saarlandes, 66 Saarbrücken, Germany
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Kassanis (1954).
An RNA-containing virus with straight to slightly curved filamentous particles c. 650 x 12 nm. Transmissible by inoculation of sap and by aphids in the non-persistent manner.
Main Diseases
Causes few or no symptoms in carnation (Dianthus caryophyllus).
Geographical Distribution
Found occasionally in cultivated carnation plants in UK and continental Europe.
Host Range and Symptomatology
No extensive studies on host range and symptomatology. Difficult to transmit from carnation to other species because of inhibitors in the sap. The virus can infect a number of plants outside the family Caryophyllaceae such as Nicotiana clevelandii, in which it multiplies without causing symptoms (M. Hollings, pers. comm.).
- Diagnostic species
- Chenopodium amaranticolor.
Small chlorotic local lesions in inoculated leaves; systemic mottle. - Chenopodium quinoa. Small yellowish spots in inoculated leaves; systemic
interveinal mottle and leaf dwarfing (Fig. 1).
- Propagation species
- Dianthus barbatus
is a suitable source of virus for purification.- Assay species
- Chenopodium amaranticolor
or C. quinoa.
Strains
None reported.
Transmission by Vectors
Transmissible in the non-persistent manner by the aphid Myzus persicae to carnation, sweet william and sugar beet (Kassanis, 1955).
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
Antisera with titres of 1/32,000 were prepared (Wetter, 1960) using purified virus preparations. The precipitate in precipitin tube tests is flagellar. Double diffusion tests in agar gel can be used, but a high concentration of antigen is necessary (Wetter, 1967). No low-molecular weight antigen has been observed.
Relationships
The virus belongs to the potato virus S group. It is distantly serologically related to the following viruses: potato S (Kassanis, 1955; 1956) potato M (Bagnall, Wetter & Larson, 1959), chrysanthemum B (Hakkaart, Van Slogteren & De Vos, 1962), passiflora latent (Brandes & Wetter, 1963), cactus 2 (Brandes & Wetter, 1963) and red clover vein mosaic (Wetter, 1967).
Stability in Sap
In carnation sap, the thermal inactivation point (10 min) is 60-65°C, dilution end-point 10-3-10-4 and infectivity is retained in carnation sap at 20°C for 2-3 days.
Purification
(Wetter & Paul, 1961). Extract sap from leaf tissue of Dianthus barbatus, filter and add ascorbic acid (to 0.2% w/v) and sodium sulphite (to 0.2% w/v). Shake sap with an equal volume of diethylether. Centrifuge at low speed. Shake clarified aqueous phase with an equal volume of carbon tetrachloride. Centrifuge at low speed and again retain aqueous phase. Adjust to pH 7.0 with 0.5 M phosphate buffer and add n-butanol to 8% (v/v). Keep at 4°C overnight. Shake with an equal volume of carbon tetrachloride and centrifuge at low speed, retaining aqueous phase. Sediment the virus by high speed centrifugation, resuspending it in 0.2 M borate buffer. Dialyse against 0.02 M borate buffer at 4°C. Concentrate by three cycles of differential centrifugation. Infected leaves of Dianthus barbatus may yield 40-60 mg virus per litre of expressed sap.
Properties of Particles
(Paul & Wetter, 1964). Sedimentation coefficient (s20,w): 167 S. No accessory viral components are found by ultracentrifugation but aggregates may occur.
Diffusion coefficient: c. 0.24 x 10-7cm2/sec.
Molecular weight: c. 6 x 107.
Partial specific volume: 0.72 cm3/g.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 2.1.
A260/A280: 1.37; Amax/Amin: 1.23.
Particle Structure
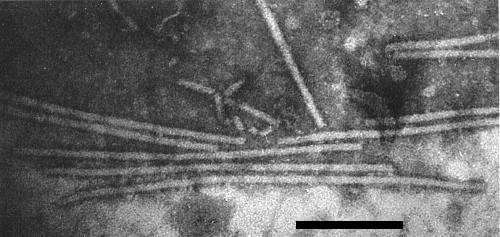
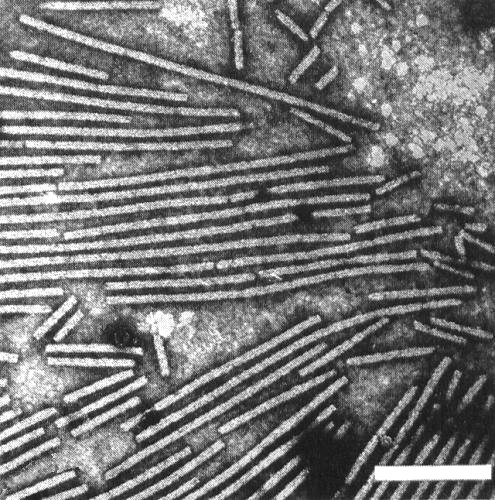
Particles are straight to slightly curved filaments c. 650 x 12 nm (Brandes et al., 1959) with a central channel (Fig. 2, Fig. 3). Particles are helically constructed with a basic helix of pitch 3.3 nm and about 12 subunits per turn (Varma et al., 1968).
Particle Composition
14.6% of particle weight is nitrogen, 0.5% phosphorus. RNA is 6% of particle weight (Paul & Wetter, 1964).
Relations with Cells and Tissues
No reports.
Notes
No differential host has been found in which carnation latent virus can be separated from other carnation viruses. It may be transmitted to healthy Dianthus barbatus plants by the aphid Myzus persicae which is also a vector of carnation vein mottle virus. It is possible to eliminate the latter from sap containing both viruses by heating the inoculum for 10 min at 60°C (Kassanis, 1955).
Acknowledgements
The support of the ‘Deutsche Forschungsgemeinschaft’ is gratefully acknowledged.
Figures
References list for DPV: Carnation latent virus (61)
- Bagnall, Wetter & Larson, Phytopathology 49: 435, 1959.
- Brandes & Wetter, Phytopath. Z. 49: 61, 1963.
- Brandes, Wetter, Bagnall & Larson, Phytopathology 49: 443, 1959.
- Hakkaart, Van Slogteren & De Vos, Tijdschr. PlZiekt. 63: 126, 1962.
- Kassanis, Nature, Lond. 173: 1097, 1954.
- Kassanis, Ann. appl. Biol. 43: 103, 1955.
- Kassanis, J. gen. Microbiol. 15: 620, 1956.
- Paul & Wetter, Phytopath. Z. 49: 401, 1964.
- Varma, Gibbs, Woods & Finch, J. gen. Virol. 2: 107, 1968.
- Wetter, Arch. Mikrobiol. 37: 278, 1960.
- Wetter, Z. Naturf. Ser. B 22: 1008, 1967.
- Wetter & Paul, Phytopath.Z. 43: 207, 1961.