Details of DPV and References
DPV NO: 207 July 1979
Family: Closteroviridae
Genus: Closterovirus
Species: Beet yellow stunt virus | Acronym: BYSV
Beet yellow stunt virus
J. E. Duffus U.S. Agricultural Research Station, Salinas, California 93915, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Duffus (1972).
A virus with flexuous filamentous particles c. 1400 nm long. Transmitted by several species of aphids in the semi-persistent manner, but not by inoculation with sap. Infects species in the Chenopodiaceae, Compositae and a few other families. Widespread in California. Occasionally destructive on lettuce.
Main Diseases
Causes a yellow, twisting stunt disease of sugar beet (Beta vulgaris) (Fig. 1, Fig. 2) and a severe chlorosis and collapse of lettuce (Lactuca sativa) (Fig. 3). Also occurs commonly in sowthistle (Sonchus oleraceus) where it causes a brilliant red or yellow interveinal coloration.
Geographical Distribution
Widespread and of high incidence in California in sowthistle. Typical symptoms have been observed in sowthistle in Oregon and Washington States, USA, and in England and Scotland, but there is no experimental evidence for the occurrence of the virus in these regions (Duffus, 1972).
Host Range and Symptomatology
Infects species in the dicotyledonous families Chenopodiaceae, Compositae, Geraniaceae, Portulacaceae and Solanaceae. Transmitted readily by aphids, but not by inoculation with sap. Infected plants commonly show interveinal yellowing or reddening of the lower or intermediate leaves; some hosts develop extremely severe symptoms with stunting, necrosis and death.
-
Diagnostic species
- Beta vulgaris
(sugar beet). Initially shows severe twisting, cupping and epinasty of leaves of intermediate age. Petioles are shortened and leaves mottled and yellow. Plants are severely stunted and may collapse and die (Fig. 1, Fig. 2). -
Chenopodium capitatum. Interveinal reddening of the older leaves, similar to
the symptoms induced by mild isolates of beet yellows virus, but with no vein clearing
or vein etching.
- Lactuca sativa (lettuce). Severe stunting and chlorosis. Older leaves collapse prematurely and turn necrotic. Plants infected when young sometimes collapse and die before heading. Virus infection may be rapidly diagnosed in the field by pulling affected plants and cutting the stem and crown tissue longitudinally. The phloem tissue in diseased plants is severely necrotic, and shows distinct brown zones extending into the crown tissue (Fig. 3, Fig. 4).
- Sonchus oleraceus (sowthistle). Intense red or sometimes yellow interveinal coloration.
- Lactuca sativa (lettuce). Severe stunting and chlorosis. Older leaves collapse prematurely and turn necrotic. Plants infected when young sometimes collapse and die before heading. Virus infection may be rapidly diagnosed in the field by pulling affected plants and cutting the stem and crown tissue longitudinally. The phloem tissue in diseased plants is severely necrotic, and shows distinct brown zones extending into the crown tissue (Fig. 3, Fig. 4).
-
Propagation species
- Beta vulgaris, Lactuca sativa
and Sonchus oleraceus are suitable for maintaining cultures. Sonchus oleraceus is readily infected with virus by using the aphid vector, Nasonovia lactucae.Assay species
- Sonchus oleraceus
is suitable in tests for transmission by the aphid Nasonovia lactucae, and Chenopodium capitatum in tests with Myzus persicae.
Strains
None reported.
Transmission by Vectors
Transmitted by aphids in a semi-persistent manner. Nasonovia lactucae is the most efficient vector; it is commonly found on sowthistle, but feeds only transiently on lettuce and rarely on sugarbeet. Transmitted less efficiently by Myzus persicae and Macrosiphum euphorbiae. Most aphids cease to transmit 1 or 2 days after acquisition but a few transmit for up to 4 days; the virus was not transmitted by insects after moulting. Single aphids were capable of acquiring the virus and losing it three successive times.
Transmission through Seed
Not tested directly but there is no evidence for seed transmission from infected lettuce, sowthistle or sugar beet plants.
Transmission by Dodder
Not transmitted by Cuscuta californica.
Serology
The virus is moderately immunogenic. Antisera prepared by intramuscular injections of clarified sap from infected sugar beet reacted in tube precipitin tests with clarified sap from infected lettuce and sowthistle.
Relationships
Resembles the closteroviruses beet yellows (Russell, 1970), carnation necrotic fleck (Inouye, 1974), citrus tristeza (Price, 1970), festuca necrosis (Schmidt et al., 1963) and wheat yellow leaf (Inouye, 1976) viruses in particle morphology, transmission by aphids, and relations with tissues. Duffus (1972) detected no serological relationship to beet yellows virus.
Stability in Sap
Not tested.
Purification
Can be partially purified from clarified sap by differential centrifugation.
Properties of Particles
Unknown.
Particle Structure
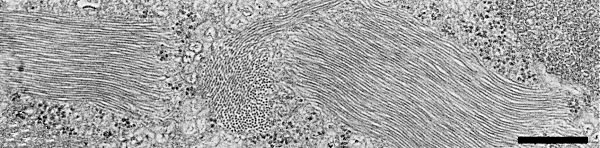
Flexuous filamentous particles c. 1400 nm long and c. 12.5 nm in diameter (Fig. 5, Fig. 6) (J. E. Duffus & L. L. Hoefert, unpublished data).
Particle Composition
Unknown.
Relations with Cells and Tissues
In beet (Hoefert, Esau & Duffus, 1970) and Sonchus oleraceus, virus particles are found only in cells of the vascular tissues, usually in the cytoplasm, and form aggregates of various sizes (Fig. 5). Degenerative changes occur in the chloroplasts of mesophyll and phloem parenchyma and in plastids of sieve elements. Some phloem parenchyma cells undergo complete breakdown.
Notes
Beet yellow stunt virus and beet yellows virus have several characteristics in common. Both are transmitted in a semi-persistent manner by aphids, and both have long, flexuous filamentous particles. The viruses induce yellowing diseases of sugarbeet and Chenopodium capitatum (a major indicator host of beet yellows virus). They differ markedly, however, in host range, especially in the Compositae (sowthistle and lettuce are immune to beet yellows virus). Sowthistle is a widespread natural reservoir of beet yellow stunt virus. Although beet yellows virus can be effectively controlled by adopting a beet-free period at various times during the year (Duffus, 1978), the distribution of beet yellow stunt virus in wild Sonchus is so extensive that its incidence seems unlikely to be affected by cropping changes, except those that affect weed populations. Incidence of beet yellow stunt virus is high in rows adjacent to areas where sowthistle is prevalent, but becomes progressively less with increasing distance from the virus source.
Figures
References list for DPV: Beet yellow stunt virus (207)
- Duffus, Phytopathology 62: 161, 1972.
- Duffus, J. Am. Soc. Sug. Beet Technol. 20: 1, 1978.
- Hoefert, Esau & Duffus, Virology 42: 814, 1970.
- Inouye, CMI/AAB Descriptions of Plant Viruses 136, 3 pp., 1974.
- Inouye, CMI/AAB Descriptions of Plant Viruses 157, 3 pp., 1976.
- Price, CMI/AAB Descriptions of Plant Viruses 33, 3 pp., 1970.
- Russell, CMI/AAB Descriptions of Plant Viruses 13, 3 pp., 1970.
- Schmidt, Richter, Hertzsch & Klinkowski, Phytopath. Z. 47: 66, 1963.