Details of DPV and References
DPV NO: 241 July 1981
Family: Betaflexiviridae
Genus: Carlavirus
Species: Hop mosaic virus | Acronym: HpMV
Hop mosaic virus
D. J. Barbara East Malling Research Station, Maidstone, Kent, England
A. N. Adams East Malling Research Station, Maidstone, Kent, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Disease first described by Salmon (1923). Virus first described by Bock
(1967) and Adams & Barbara (1980).
- A virus with filamentous RNA-containing particles c. 14 x 650 nm frequently occurring in hop (Humulus lupulus) in Europe, Tasmania and North America. Transmitted in the non-persistent manner by aphids and by mechanical inoculation of sap.
Main Diseases
Lethal to Golding-type hop cultivars (e.g. Bramling. Eastwell Golding, Wye Mathon), which are no longer widely grown, but does not cause symptoms in tolerant cultivars. Severe disease problems have occurred in sensitive cultivars grown close to virus-infected tolerant ones (Keyworth, 1943; Eppler, 1980). Cultivars of the Golding type show chlorotic vein-banding, recurving of the leaf margins and stunting, the plant tending to fall away from its support string (Fig. 1); affected plants yield poorly and usually die prematurely. Mild strains of the virus (Fig. 2) (Legg, 1959) may be common and are probably the cause of several diseases described by Blattný & Osvald (1949) and of chlorotic mosaic disease in France (Cairaschi, 1953).
Geographical Distribution
Common in Europe (Keyworth, 1947; Schmidt & Klinkowski, 1965), Tasmania (Cartledge, 1956), and North America (Skotland, 1960).
Host Range and Symptomatology
Experimentally the virus infected 11 out of 44 species in 5 out of 14 families but symptoms have been seen only in sensitive cultivars of hop. The virus has been detected in wild plants of Urtica urens in England (Adams & Barbara, 1980) and in Stellaria media, Chenopodium album, Plantago major and Polygonum aviculare in West Germany (Eppler, 1980).
- Diagnostic species
- Humulus lupulus
(hop) cultivars of the sensitive Golding type react in the same way as field-infected plants when graft inoculated (see Main diseases).- Propagation species
- Nicotiana clevelandii
is suitable for maintaining cultures and as a source of virus for purification.- Assay species
- No local lesion host is known. The virus may be assayed by determining the proportion of Nicotiana clevelandii plants that become infected.
Strains
Most studies have been with virulent isolates. However, isolates causing mild symptoms in hop were reported by Legg (1959) and may be common.
Transmission by Vectors
Transmitted in the non-persistent manner by the aphids Myzus persicae, Macrosiphum euphorbiae and Phorodon humuli (Adams & Barbara, 1980). Experimental transmission between hop plants by P. humuli is inefficient; Paine & Legg (1953) obtained transmission only by spring alatae of this aphid but transmission is also possible by glasshouse-reared alienicolae (A. N. Adams, unpublished data).
Transmission through Seed
None found in hop, Nicotiana clevelandii or Urtica urens.
Serology
Antisera with homologous titres of up to 1/3200 in tube precipitin tests were readily obtained by injecting rabbits intramuscularly with preparations of intact virus particles. Virus degraded by pyrrolidine was poorly immunogenic (Adams & Barbara, 1980). Extracts from infected Nicotiana clevelandii and hop plants react at dilutions in excess of 10-4 in ELISA, which has been used extensively for the detection of the virus in tolerant cultivars (Thresh et al., 1977).
Relationships
A mild strain reported by Legg (1959) protected hop plants against infection with severe isolates; it is not known whether mild and virulent isolates differ serologically. The properties of the virus suggest that it is a member of the carlavirus group: it is serologically distantly related to carnation latent, potato M, cowpea mild mottle and hop latent viruses (Adams & Barbara, 1980).
Stability in Sap
No information.
Purification
The following procedure yields up to 40 mg virus/kg inoculated Nicotiana clevelandii leaves (Adams & Barbara, 1980); 0.05 M borate buffer (pH 8) is used throughout. One month after inoculation, extract leaves at 4°C in buffer (5 ml/g leaf) containing 10 g/l sodium sulphite and centrifuge at 18,000 g for 20 min. To the supernatant fluid add polyethylene glycol (M. Wt 6000) and sodium chloride to 50 and 6 g/l respectively. Keep the mixture at 4°C for 1 h and centrifuge at 18,000 g for 40 min. Resuspend the pellets in 1/15th original volume of extract and subject to one cycle of differential centrifugation. Resuspend pellets overnight, clarify by low speed centrifugation and further purify by sucrose gradient centrifugation.
Properties of Particles
Purified preparations show one component in sucrose density gradient centrifugation.
A260/A280: 1.22 (corrected for light-scattering.)
Particle Structure
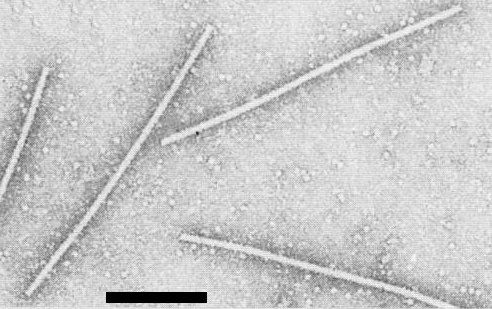
Straight or slightly flexuous filamentous particles c. 14 x 650 nm (Fig. 4).
Particle Composition
Nucleic acid: RNA, single-stranded, c. 5-7% of the particle weight (estimated from the A260/A280 ratio), M. Wt c. 3.0 x 106 (estimated by polyacrylamide gel electrophoresis; Adams & Barbara, 1980).
Protein: c. 93-95% of the particle weight (estimated from the A260/A280 ratio). M. Wt of the single protein species c. 34,000 (estimated by polyacrylamide gel electrophoresis; Adams & Barbara, 1980).
Relations with Cells and Tissues
No information.
Notes
At least two other viruses with similar filamentous particles occur in hop. Hop latent virus, a carlavirus which induces local lesions in Chenopodium murale but does not infect N. clevelandii, occurs in Europe (Schmidt, Schmidt & Eisbein, 1966; Thresh & Ormerod, 1969) and the USA (Probasco & Skotland, 1976a). An unnamed virus which has a wider host range than hop mosaic virus, and infects Chenopodium quinoa systemically, is common in hop in the USA where it was first isolated by Probasco & Skotland (1976b). It has been detected in Europe in introductions from the USA (A. N. Adams, unpublished results). All three viruses can occur together in hop plants that are apparently symptomless but the viruses are distinct both serologically and in host range.
Figures
References list for DPV: Hop mosaic virus (241)
- Adams & Barbara, Ann. appl. Biol. 96: 201, 1980.
- Bock, Rep. E. Malling Res. Stn for 1966: 163, 1967.
- Blattný & Osvald, Ochr. Rost. 22: 5, 1949.
- Cairaschi, Revue Path. vég. Ent. agric. Fr. 32: 175, 1953.
- Cartledge, Tasm. J. Agric. 27: 210, 1956.
- Eppler, Dissertation zum Doktor der Naturwissenschaften der Fakultät für Biologie der Eberhard Karls-Universität Tübingen, 1980.
- Keyworth, J. Inst. Brew. 49: 128, 1943.
- Keyworth, Rep. E. Malling Res. Stn for 1946: 142, 1947.
- Legg, Rep. E. Malling Res. Stn for 1958: 116, 1959.
- Paine & Legg, Nature, Lond. 171: 263, 1953.
- Probasco & Skotland, Can. J. Microbiol. 22: 1160, 1976a.
- Probasco & Skotland, Proc. Am. Phytopath. Soc. 3: 319, 1976b.
- Salmon, J. Minist. Agric. Fish. 29: 927, 1923.
- Schmidt & Klinkowski, Phytopath. Z. 54: 122, 1965.
- Schmidt, Schmidt & Eisbein, Zentbl. Bakt. ParasitKde Abt. II 120: 461, 1966.
- Skotland, Phytopathology 50: 655, 1960.
- Thresh, Adams, Barbara & Clark, Ann. appl. Biol. 87: 57, 1977.
- Thresh & Ormerod, Rep. E. Malling Res. Stn for 1968: 41, 1969.