Details of DPV and References
DPV NO: 278 July 1984
Family: Geminiviridae
Genus: Mastrevirus
Species: Tobacco yellow dwarf virus | Acronym: TYDV
Tobacco yellow dwarf virus
J. E. Thomas Department of Primary Industries, Indooroopilly, Queensland, 4068, Australia
J. W. Bowyer Department of Plant Pathology, University of Sydney, New South Wales, 2006, Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Hill (1937) and Ballantyne (1968). Virus first purified by Thomas
& Bowyer (1980).
- Synonym
- (Bean) summer death (virus) (Rev. appl. Mycol. 48: 654)
- A virus with geminate particles, 20 nm x 35 nm. It has a fairly wide host range and is transmitted in the persistent manner by the leafhopper Orosius argentatus, but not by mechanical inoculation. Recorded only from Australia and causes economically significant diseases in bean (Phaseolus vulgaris) and tobacco (Nicotiana tabacum).
Main Diseases
Causes a severe dwarfing disease in tobacco (Hill, 1937) and a lethal, necrotic disease in susceptible bean cultivars (Ballantyne, 1968).
Geographical Distribution
Reported only from Australia, where it occurs in all States.
Host Range and Symptomatology
Not transmitted by mechanical inoculation. Transmitted by the leafhopper vector or by grafting to 30 species in seven dicotyledonous families (Helson, 1950; Hill, 1950; Hill & Mandryk, 1954; Thomas & Bowyer, 1979).
- Diagnostic species
- Datura stramonium.
Strong interveinal chlorosis and down-curling of leaf margins (Fig. 1). - Phaseolus vulgaris (French bean) cvs Spartan Arrow, Bountiful. Plants
inoculated at the cotyledon stage show a reduction in the growth rate of the first
trifoliolate leaf, usually in 7 to 10 days. Down-curling of the trifoliolate leaf
margin, vascular necrosis of the upper stem and epinasty of the primary leaves and
petioles follow rapidly. Ultimately, interveinal chlorosis of the primary leaves,
leaf abscission, axillary shoot formation and finally wilting and collapse of the
plant occur (Fig. 2, Fig. 3). Symptom expression is more rapid in warm conditions
(c. 30°C) and plant death can occur as soon as 8 days after the first
appearance of symptoms.
- Nicotiana tabacum (tobacco). Down-curling of the tips and margins of the youngest leaves, chlorosis and stunting (Fig. 4). Small necrotic spots sometimes occur on the older chlorotic leaves.
- Nicotiana tabacum (tobacco). Down-curling of the tips and margins of the youngest leaves, chlorosis and stunting (Fig. 4). Small necrotic spots sometimes occur on the older chlorotic leaves.
- Propagation species
- Lycopersicon esculentum
(tomato) is useful for maintaining cultures. Datura stramonium is a suitable host for acquisition feeding by the leafhopper vector and as a source of virus for purification.- Assay species
- No local lesion host is known. The virus can be assayed by inoculating seedlings of susceptible cultivars of Phaseolus vulgaris (French bean) with leafhoppers that have acquired virus from test plants or by feeding through membranes.
Strains
Isolates differ in the severity of symptoms produced in Datura stramonium and tobacco.
Transmission by Vectors
Transmitted by the leafhopper Orosius argentatus (Hill, 1941) but not by the leafhoppers Batracomorphus punctatus, Orosius lotophagorum ssp. ryukyuensis (Thomas, 1979) or Austroasca alfalfae (J. E. Thomas, unpublished data). An isolate from Phaseolus vulgaris had a minimum latent period of 24-48 h in O. argentatus; infectivity was not lost after moulting and was retained for at least 21 days after removal from infected plants (Bowyer & Atherton, 1971).
Transmission through Seed
Not detected in Datura stramonium (Thomas, 1979).
Transmission by Dodder
Not transmitted by Cuscuta campestris or eight other species of dodder (Hill & Mandryk, 1954).
Serology
Moderately immunogenic in rabbits. An antiserum with a gel diffusion titre of 1/128 was produced by injecting a total of 230 µg virus. Gel diffusion tests are possible only with concentrated or purified virus preparations. Virus can be detected in sap by immunosorbent electron microscopy and ELISA.
Relationships
A number of isolates from bean and tobacco were shown by ELISA to be
serologically related (J. W. Bowyer & J. E. Thomas, unpublished data). In
gel diffusion serology tests with antiserum to a tobacco isolate, confluent
precipitin lines were formed between bean and tobacco isolates of the virus
(J. E. Thomas, unpublished data). No cross-protection was detected between bean
and tobacco isolates of the virus (Than, 1976).
Tobacco yellow dwarf virus is distantly serologically related to beet curly top
virus, as demonstrated by gel double diffusion tests (Thomas & Bowyer, 1980)
and immunosorbent electron microscopy (B. D. Harrison & I. M. Roberts,
personal communication; J. E. Thomas, unpublished data). Many bean cultivars
selected for resistance to beet curly top virus were also resistant to tobacco
yellow dwarf virus (Ballantyne et al., 1969; Ballantyne, 1970). However,
sugarbeet cultivars susceptible to beet curly top virus could not be infected
by two isolates of tobacco yellow dwarf virus, although some plants of one
susceptible cultivar were symptomlessly infected by a third isolate (J. E.
Thomas, unpublished data).
No serological reaction was detected between tobacco yellow dwarf virus and antiserum to either maize streak or chloris striate mosaic viruses, by gel diffusion serology (Thomas, 1979). Electron microscope grids coated with tobacco yellow dwarf virus antiserum did not trap particles of maize streak, wheat dwarf, cassava latent or tomato golden mosaic viruses (B. D. Harrison & I. M. Roberts, personal communication).
Stability in Sap
Determined by feeding leafhoppers through membranes on virus-containing extracts. The thermal inactivation point of a partially purified preparation from tobacco was between 50°C and 60°C and the dilution end-point in bean sap was between 1/50 and 1/500. Infective virus was recovered from bean tissue stored at -18°C for 11 months and from a purified preparation stored at 4°C for 26 days.
Purification
Modifications of the methods used for beet curly top virus (Mumford, 1974) and maize streak virus (Bock et al., 1974) are suitable (Thomas & Bowyer, 1980). A useful increase in yield is obtained by re-extracting the ‘fibres’ in 0.1 M phosphate buffer (pH 6.0) containing 0.2% (v/v) thioglycerol and 1% (w/v) cellulase and incubating, with shaking, overnight at room temperature. Yields are usually less than 250 µg virus/kg leaf tissue.
Properties of Particles
Geminate particles form a single band in sucrose density gradients.
Sedimentation coefficient (at 20°C in 0.01 M phosphate buffer, pH 7.7): 76 S.
A260/A280: 1.25 (not corrected for light-scattering).
Particle Structure
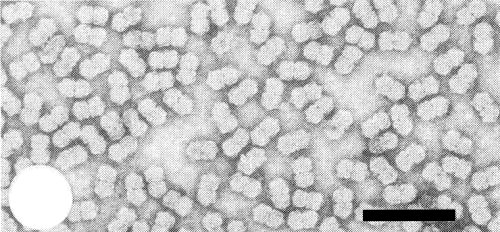
Particles are geminate c. 20 nm x 35 nm (Fig. 5, Fig. 6), probably consisting of two incomplete icosahedra with a T = 1 surface lattice and a total of 22 capsomeres (Thomas, 1979). Particles are stable in 0.5% aqueous uranyl acetate, 2% ammonium molybdate (pH 7.0) or 1% potassium phosphotungstate (pH 7.0).
Particle Composition
Nucleic acid: The type of nucleic acid has not been determined unequivocally. Nucleic acid extracted from a purified preparation with SDS-Pronase had a buoyant density in CsCl of c. 1.72 g/cm3, suggesting the presence of DNA (J. E. Thomas, unpublished data).
Protein: A single protein species of M. Wt c. 27,500, estimated by SDS-polyacrylamide gel electrophoresis (Thomas & Bowyer, 1980).
Relations with Cells and Tissues
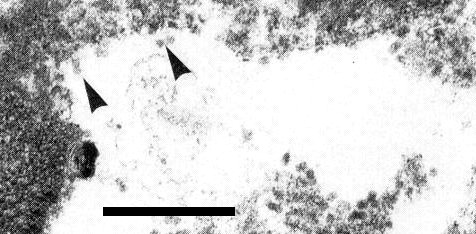
In French bean, cytopathic effects are obvious only in the phloem tissue. Crushed, necrotic sieve tubes become apparent about 8 days after inoculation, coincident with the appearance of macroscopic symptoms in the plant. Virus particles can be detected in some necrotic phloem cells (Fig. 7).
Notes
Tobacco yellow dwarf virus can be distinguished from most other viruses by its geminate particles and leafhopper vector. Two other geminiviruses, tobacco leaf curl virus and beet curly top virus, have many hosts in common with tobacco yellow dwarf virus. Tobacco leaf curl virus, however, is transmitted by the whitefly Bemisia tabaci. Beet curly top virus causes similar symptoms to tobacco yellow dwarf virus in several hosts and is also transmitted by leafhoppers, so the two viruses are best distinguished by serological tests.
Figures
References list for DPV: Tobacco yellow dwarf virus (278)
- Ballantyne, Agric. Gaz. N.S.W. 79: 486, 1968.
- Ballantyne, Pl. Dis. Reptr 54: 903, 1970.
- Ballantyne, Sumeghy & Pulver, Agric. Gaz. N.S.W. 80: 430, 1969.
- Bock, Guthrie & Woods, Ann. appl. Biol. 77: 289, 1974.
- Bowyer & Atherton, Phytopathology 61:1451, 1971.
- Helson, Aust. J. agric. Res. 1:144, 1950.
- Hill, J. Counc. scient. ind. Res. Aust. 10: 228, 1937.
- Hill, J. Counc. scient. ind. Res. Aust. 14: 181, 1941.
- Hill, Aust. J. agric. Res. 1:141, 1950.
- Hill & Mandryk, Aust. J. agric. Res. 5: 617, 1954.
- Mumford, Phytopathology 64:136, 1974.
- Than, M.Agric. Thesis, University of Sydney, 1976.
- Thomas, Ph.D. Thesis, University of Sydney, 1979.
- Thomas & Bowyer, Australas. Pl. Path. 8: 36, 1979.
- Thomas & Bowyer, Phytopathology 70: 214, 1980.