Details of DPV and References
DPV NO: 217 September 1980
Family: Reoviridae
Genus: Fijivirus
Species: Oat sterile dwarf virus | Acronym: OSDV
Oat sterile dwarf virus
G. Boccardo Istituto di Fitovirologia applicata del CNR, Via O. Vigliani 104, 10135 Turin, Italy
R. G. Milne Istituto di Fitovirologia applicata del CNR, Via O. Vigliani 104, 10135 Turin, Italy
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
First described by
Prusa (1958) and
Lindsten (1959).
A virus with polyhedral particles 65-70 nm in diameter, having a double shell of protein containing double-stranded RNA in 10 segments. Host range confined to Gramineae and its planthopper (Delphacidae) vectors. Not mechanically transmissible to plants. Causes mild to severe diseases in oats, barley, ryegrass and Arrhenatherum.
Main Diseases
In oats the virus causes dwarfing, excessive tillering and flower suppression, giving a grass-like appearance, and the leaves become dark green and distorted. Small enations may occur on the backs of the leaves (Lindsten, 1961; Catherall, 1970; Brcák, Králík & Vacke, 1972). Wheat and barley show less severe symptoms but Lolium perenne and L. multiflorum may die. L. perenne and L. multiflorum may show conspicuous enations on the nodes of the flowering stem and spike (Fig. 1) (Huth, 1975). Losses of grain yield in oats may be considerable in Czechoslovakia (Prusa 1958; Brcák, 1979). In Sweden and Finland the disease, formerly severe, has been controlled by altering cultural practice (Lindsten, 1961, 1970, 1974). In Britain, losses appear negligible. In East Germany, losses in yield of forage can be severe in Arrhenatherum elatius (Schumann, 1971), and in West Germany L. multiflorum and A. elatius may be badly affected (Huth, 1975 and unpublished data). Oat sterile dwarf virus can overwinter in diapausing vector nymphs, or in perennial grass hosts such as L. perenne. For reviews, see papers in Maramorosch & Harris (1979), particularly those by Brcák, (1979), Harris (1979), Lindsten (1979) and Shikata (1979).
Geographical Distribution
Reported from Britain, Czechoslovakia, East and West Germany, Sweden, Finland, Norway and Poland. Appears to be absent from the southern areas of Sweden and from Denmark, although the vector occurs there. May be widespread in Europe except in the warmer areas where the vector does not occur (see Milne & Lovisolo, 1977; Brcák, 1979; Lindsten, 1979).
Host Range and Symptomatology
Only species in several tribes of the Gramineae are susceptible. Some hosts are: Arrhenatherum elatius, Avena spp., Cynosurus cristatus, Hordeum spp., Lolium spp., Phalaris canariensis, Poa annua, Secale cereale and Triticum spp. Zea spp. can be infected with difficulty. For a more complete list, see Milne & Lovisolo (1977). The planthopper vectors (see Transmission by Vectors) are presumably hosts but no disease is reported in these insects when they carry the virus. The virus is not sap-transmissible.
-
Diagnostic species
- Avena sativa.
The symptoms on naturally infected plants are described under Main Diseases but experimentally infected glasshouse plants may show more conspicuous enations (Lindsten, 1961; Brcák et al., 1972). - Lolium multiflorum. Infected plants are stunted, dark green, produce extra tillers and, especially in the glasshouse, develop small yellowish enations on the leaf veins.
Propagation species
- Lolium multiflorum
or other susceptible grasses that can be increased vegetatively. The virus may also be propagated in other graminaceous hosts inoculated by means of a vector.Assay species
- There is no direct plant-to-plant assay or local lesion host. Avena sativa and Lolium multiflorum have been used to assay transmission by the vector. L. multiflorum has been used to test planthoppers (Javesella pellucida) from the field as carriers of the virus (Medaiyedu & Plumb, 1976).
Strains
A mild strain has been reported in Sweden in addition to the normal one (Lindsten, 1973 and unpublished data). Lolium enation virus and arrhenatherum blue dwarf virus are serologically closely similar to each other and to oat sterile dwarf virus (Milne & Lesemann, 1978) and the latter two viruses have genome segments of the same number and electrophoretic mobility (Luisoni et al., 1979); all these viruses are transmitted in nature by J. pellucida. The data suggest that all three are closely related strains or forms of the same virus.
Transmission by Vectors
Transmitted by the planthoppers Javesella pellucida, J. obscurella, J. discolor, J. dubia and Dicranotropis hamata (Lindsten, 1959, 1961; Ikäheimo & Raatikainen, 1963; Vacke, 1964; Mühle & Kempiak, 1971; Brcák, 1979; Lindsten, 1979). The most important vector is J. pellucida. The minimum acquisition feeding period is 30-60 min and the latent period is 3-4 weeks, after which the vector can transmit for life. The minimum inoculation feeding period is about 30 min. Virus transmission through 0.2% of the eggs of J. pellucida is reported (Vacke, 1966). These properties and evidence from electron microscopic examination of thin sections (see Relations with Cells and Tissues) suggest that the virus multiplies in the vector.
Transmission through Seed
No transmission (Lindsten, 1961).
Serology
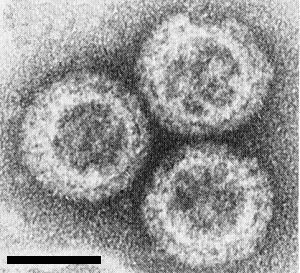
The B-spiked dsRNA-containing inner shell (subviral particle) is a good immunogen but there is no information about the A spikes and the outer shell. Two antisera, prepared by intramuscular injection of rabbits with purified subviral particles (Fig. 5) did not react, or reacted only when undiluted, with extracts of healthy plants, in gel diffusion tests. The first antiserum did not react with dsRNA but reacted with preparations of subviral particles to a dilution of 1/512. The second antiserum reacted with dsRNA to a dilution of 1/8 and with preparations of subviral particles to a dilution of 1/2048. Gel diffusion tests and immunoelectron microscopy have shown that the inner shell and probably the B spikes react, but not the A spikes or the outer shell (Milne & Lesemann, 1978; Luisoni et al., 1979).
Relationships
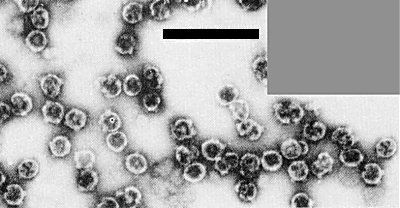
The virus is in the fijivirus group of the family Reoviridae (Fenner, 1976; Matthews, 1979). Fijiviruses differ from other plant reo-like viruses (the phytoreovirus group containing wound tumor and rice dwarf viruses) in the structure of the protein shell, type of vector and genome pattern. Unlike the phytoreoviruses, fijiviruses have spikes on the outer shell, and this outer shell is easily lost to give a spiked inner shell (Fig. 8, Fig. 9); the vectors are planthoppers (Delphacidae), not leafhoppers (Cicadellidae), and there are 10 genome segments, not 12. The viruses multiply in their insect vectors. Within the fijiviruses, the B-spiked subviral particles of oat sterile dwarf virus are not serologically related to those of Fiji disease, maize rough dwarf (Fig. 4), pangola stunt and rice black streaked dwarf viruses, or to those of the possible member rice ragged stunt virus (Milne & Luisoni, 1977a, 1977b; Milne & Lesemann, 1978; Boccardo, Milne & Luisoni, 1979; Luisoni et al., 1979; Milne, Lussoni & Ling, 1979). The natural vector of oat sterile dwarf virus, J. pellucida, is different from those of other fijiviruses, though maize rough dwarf virus can be experimentally transmitted by this species (Harpaz et al., 1965). Laodelphax striatellus, the natural vector of maize rough dwarf virus, does not transmit oat sterile dwarf virus. Oat sterile dwarf virus and maize rough dwarf virus (cereal tillering disease strain) also differ in symptoms and host range (Lindsten et al., 1973; Milne & Lovisolo, 1977). The genome of oat sterile dwarf virus (Fig. 3) resembles that of other fijiviruses but differs in the mobilities of the individual segments (Luisoni et al., 1979).
Stability in Sap
Infectivity not investigated, but see Properties of Particles.
Purification
Only the B-spiked subviral particles have been purified (Fig. 5), the outer shell and A spikes being lost during the process (Luisoni et al., 1979). Crush the roots and stem bases of infected Lolium multiflorum in the presence of 0.4 M phosphate buffer, pH 7, containing 5 mM EDTA and 10 mM sodium sulphite, in the cold. Filter the juice through nylon stocking, shake with an equal volume of Freon 113 (1,1,2-trifluoro-1,2,2-trichloroethane) and recover the subviral particles from the aqueous phase by ultracentrifugation. Purify the particles further by two cycles of differential centrifugation and banding in preformed Cs2SO4 density gradients. Low molarity buffers appropriate for the purification of other fijivirus subviral particles (Milne, Conti & Lisa, 1973; Boccardo et al., 1979) are unsuitable because the particles become aggregated with impurities. Phosphate buffer molarities greater than 0.4 damage the particles. Sucrose, metrizamide and CsCl are not suitable for the density gradients (Luisoni et al., 1979).
Properties of Particles
The particles of the virus are unstable, though somewhat less so than those of Fiji disease and maize rough dwarf viruses. Purified subviral particles are stable. Storage of virus particles for several days in sap at 4°C, or shaking with chloroform causes loss of the A spikes and outer shell, leaving B-spiked subviral particles. Treatment with 0.5 M NaCl or CsCl, 2% phosphotungstate at pH 7, or 10% n-butanol results in loss of the B spikes. Particles are stable when negatively stained in 2% phosphotungstate, pH 3 to 5, or in 2% aqueous uranyl acetate or formate. Glutaraldehyde (0.1-1.0% in 0.01 M phosphate buffer, pH 7) causes swelling of the virus particles when in suspension though not, or less so, when attached to electron microscope support films. Other properties are not investigated.
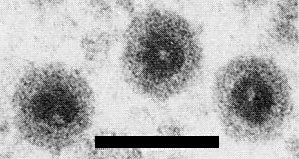
Particle Structure
In uranyl acetate or formate negative stain, the virus particles appear circular or slightly angular (Fig. 8) and 65-70 nm in diameter, and projections (A spikes) like those of other fijiviruses are occasionally observed (Milne et al., 1973, 1974, 1975; Lesemann & Huth, 1975; Hatta & Francki, 1977). The outer shell is easily lost to give a nearly spherical subviral particle 50-55 nm in diameter with 12 symmetrically placed hollow projections or B spikes (Fig. 9). Morphological subunits are discernible in the intact particles but their number and arrangement are unknown. In thin sections, objects corresponding to the virus particles (Fig. 6) and subviral particles are seen; both types of particle contain an electron-dense core (presumably nucleic acid) 45-50 nm in diameter (Lesemann & Huth, 1975; Milne et al., 1974, 1975).
Particle Composition
-
Nucleic acid: The subviral particle contains double-stranded RNA
separating into 10 segments upon polyacrylamide gel electrophoresis in 5% gels
(Fig. 3).
Taking the
maize rough dwarf virus genome
as standard
(Reddy et al., 1975),
the segments have the following estimated M. Wt (x 10-6 daltons):
2.76, 2.48, 2.35, 2.35, 2.08, 1.88, 1.18, 1.17, 1.16 and 1.00
(Luisoni et al., 1979).
Note that bands 3 and 4, though clearly separated
(Fig. 3),
have the same
mobilities as bands 3 and 4 of maize rough dwarf virus, which were both assigned
the same M. Wt of 2.35 x 106 by
Reddy et al. (1975).
The total
RNA M. Wt of 18.39 x 106 is close to those of
Fiji disease,
maize
rough dwarf and
pangola stunt viruses
but the distribution of M. Wt among the
segments is different.
Protein: No information.
Relations with Cells and Tissues
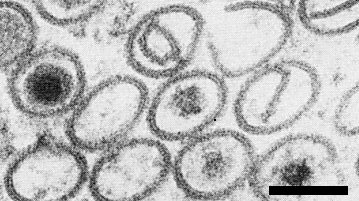
The cytopathology resembles that of other fijiviruses (Milne & Lovisolo, 1977; Maramorosch & Harris, 1979). In plants, virus particles (Fig. 6) are confined to phloem and phloem-derived enations. They occur either isolated or in crystalline aggregates in the cytoplasm and are associated with cytoplasmic ‘viroplasms’ (Fig. 2) and tubes (Fig. 2, Fig. 7, Fig. 11, Fig. 12) (Brcák et al., 1972; Lindsten et al., 1973; Milne et al., 1974; Lesemann & Huth, 1975). The viroplasm, visible as an X-body in the light microscope (Lesemann & Huth, 1975; Kempiak, 1977) contains a central fibrillar region devoid of virus particles, surrounded by a granular, more electron-dense zone in which the subviral particles are formed. These mature into complete virus particles at the edge of the viroplasm. In negatively stained unpurified preparations, kinked filaments (Fig. 10) are seen; they are possibly the viroplasm fibrils (Milne, 1977) or perhaps re-aggregated outer capsid protein (Hatta & Francki, 1977). The tubes, often containing virus particles, probably consist of a protein but they do not react with antisera to the inner capsid or B spikes. The subunits of the tube wall form a square lattice with 4 nm spacing, the same spacing found in maize rough dwarf-associated tubes (R. G. Milne, unpublished data; Milne & Lovisolo, 1977). Tubes may be incompletely closed, forming scrolls (Fig. 7). A similar pathology, though without cell hyperplasia, is found in midgut cells of the infected vector (Brcák, Králík & Vacke, 1970; Brcák 1979).
Notes
Disease in cereals caused by oat sterile dwarf virus has been controlled by avoiding overwintering oat crops, especially as cover crops for undersown leys. Using barley or wheat instead of oat reduces the number of virus-carrying overwintering vector individuals (Lindsten, 1974, 1979).
In Sweden, the cereal tillering disease strain of maize rough dwarf virus causes a disease easily confused in the field with oat sterile dwarf (Lindsten et al., 1973).
Oat blue dwarf virus, from North America (Banttari & Zeyen, 1973), has isometric particles 28-30 nm in diameter and is quite different from oat sterile dwarf virus, although it is transmitted by a leafhopper (Macrosteles fascifrons) and produces rather similar symptoms. A virus similar to oat blue dwarf, in Czechoslovakia, is transmitted by M. laevis (Vacke, 1970) and is also found in Sweden (K. Lindsten, personal communication).
The other viruses isolated from oats are oat mosaic (Hebert & Panizo, 1975) and oat necrotic mottle (Gill, 1976), both of which are potyviruses; oat golden stripe, a soil-borne virus with tubular particles of two modal lengths (MacFarlane & Plumb, 1978); maize rough dwarf (Conti & Milne, 1977); and barley yellow dwarf (Rochow, 1970).
Lolium multiflorum, used to isolate and propagate oat sterile dwarf virus, frequently carries symptomlessly a small isometric seed-borne virus (Plumb & Misari, 1974).
Acknowledgements
We thank H. Paul, W. Huth and D. E. Lesemann, Braunschweig, and K. Lindsten, Uppsala, for kindly reviewing the script and offering unpublished data.
Figures

Lolium perenne naturally infected with the Lolium enation isolate. The arrows indicate characteristic swellings or enations. (Courtesy of W. Huth.)
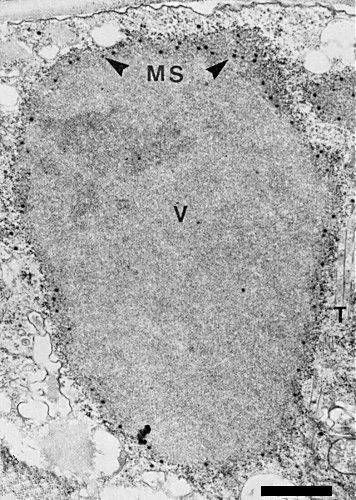
Infected Lolium multiflorum phloem parenchyma, showing viroplasm (V), maturing subviral particles (MS, arrows) and tubes (T). Bar represents 1 µm.
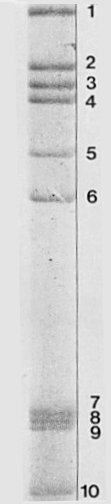
The double-stranded RNA genome segments after electrophoresis in 5% polyacrylamide (phosphate-Tris-EDTA buffer). Ten bands are seen (extremities of the gel not shown).

Subviral particles of oat sterile dwarf virus (right) and maize rough dwarf virus (left) treated with undiluted antiserum to oat sterile dwarf subviral particles. Bar represents 100 nm.
References list for DPV: Oat sterile dwarf virus (217)
- Banttari & Zeyen, CMI/AAB Descriptions of Plant Viruses 123, 4 pp., 1973.
- Boccardo, Milne & Luisoni, J. gen. Virol. 45: 659, 1979.
- Brcák, in Leafhopper Vectors and Plant Disease Agents, ed. K.Maramorosch & K. Harris, 654 pp., New York: Academic Press, 1979.
- Brcák, Králík & Vacke, Proc.1st int. Symp. Pl. Path. New Delhi, 1966, p. 93, 1970.
- Brcák, Králík & Vacke, Biol. Plantarum 14: 302, 1972.
- Catherall, Pl. Path. 19: 75, 1970.
- Conti & Milne, Annls Phytopath. 9: 255, 1977.
- Fenner, Intervirology 7: 1, 1976.
- Gill, CMI/AAB Descriptions of Plant Viruses 169, 4 pp., 1976.
- Harpaz, Vidano, Lovisolo & Conti, Atti Accad. Sci., Torino 99: 885, 1965.
- Harris, in Leafhopper Vectors and Plant Disease Agents, ed. K. Maramorosch & K. Harris, 654 pp., New York: Academic Press, 1979.
- Hatta & Francki, Virology 76: 797, 1977.
- Hebert & Panizo, CMI/AAB Descriptions of Plant Viruses 145, 4 pp., 1975.
- Huth, NachrBl. dt. PflSchutzdienst., Braunschweig 27: 49, 1975.
- Ikäheimo & Raatikainen, Ann. Agric. Fenn. 2: 153, 1963.
- Kempiak, Arch. Phytopath. PflSchutz. 13: 83, 1977.
- Lesemann & Huth, Phytopath. Z. 82: 246, 1975.
- Lindsten, Phytopath Z. 35: 420, 1959.
- Lindsten, K. LantbrHögsk. Annlr 27: 137, 1961.
- Lindsten, Meddn St. VäxtskAnst. 14: 134, 1970.
- Lindsten, Växtskyddsnotiser 37: 55, 1973.
- Lindsten, Mikrobiologija 11: 55, 1974.
- Lindsten, in Leafhopper Vectors and Plant Disease Agents, ed. K.Maramorosch & K. Harris, 654 pp., New York: Academic Press, 1979.
- Lindsten, Gerhardson & Pettersson, Meddn St. VäxtskAnst. 15: 375, 1973.
- Luisoni, Boccardo, Milne & Conti, J. gen. Virol. 45: 651, 1979.
- MacFarlane & Plumb, Abstracts 3rd. int. Congr. Plant Path. München 1978, p. 27, 1978.
- Maramorosch & Harris, eds., Leafhopper Vectors and Plant Disease Agents, 654 pp., New York: Academic Press, 1979.
- Matthews, Intervirology 11: 133, 1979.
- Medaiyedu & Plumb, Rep. Rothamsted exp. Stn 1975, Part 1, p. 260, 1976.
- Milne, Annls Phytopath. 9: 333, 1977.
- Milne & Lesemann, Virology 90: 299, 1978.
- Milne & Lovisolo, Adv. Virus Res. 21: 267, 1977.
- Milne & Luisoni, Virology 80: 12, 1977a.
- Milne & Luisoni, Annls Phytopath. 9: 337, 1977b.
- Milne, Conti & Lisa, Virology 53: 130, 1973.
- Milne, Kempiak, Lovisolo & Mühle, Phytopath. Z. 79: 315, 1974.
- Milne, Lindsten & Conti, Ann. appl. Biol. 79: 371, 1975.
- Milne, Luisoni & Ling, Pl. Dis. Reptr 63: 445, 1979.
- Mühle & Kempiak, Phytopath. Z. 72: 269, 1971.
- Plumb & Misari, Rep. Rothamsted exp. Stn 1973, Part I, p. 37, 1974.
- Prusa, Phytopath. Z. 33: 99, 1958.
- Reddy, Boccardo, Outridge, Teakle & Black, Virology 63: 287, 1975.
- Rochow, CMI/AAB Descriptions of Plant Viruses 32, 4 pp., 1970.
- Schumann, in Krankheiten und Schädlinge der Futtergräser, ed. E. Mühle, p. 94, Leipzig: Hirzel Verlag, 1971.
- Shikata, in Leafhopper Vectors and Plant Disease Agents, ed. K.Maramorosch & K. Harris, 654 pp., New York: Academic Press, 1979.
- Vacke, Proc. 5th Conf. Czech. Plant Virol. Prague, 1962, p. 335, 1964.
- Vacke, Biologia Plantarum 8: 127, 1966.
- Vacke, Ved. Pr. výsk. Ust. rostl. Výroby Praze-Ruzyni 16: 21, 1970.